INTRODUCTION
Stroke is one of the top causes of disability across the globe. Most stroke survivors require specialized rehabilitation after sustaining a stroke ( Rafiei et al., 2019; Koch et al., 2021; Rajashekar et al., 2021). Medical intervention and specific therapy are used during inpatient stroke rehabilitation to assist patients in regaining maximal functionality ( Kim et al., 2022). Stroke survivors may be left with physical, mental, and emotional impairments that affect their routine activities. They may require a long time to recover and return to normal life ( Kim et al., 2021). In addition, they lack continuity of treatment, health and social care service deficits, and service choices outside of healthcare ( Bonkhoff and Grefkes, 2022). A family member or companion is necessary to perform their regular activities. Clinicians utilize clinical outcome measures or standardized assessment tools, including the functional independence measure, for determining a patient’s degree of impairment and the Ten-Meter Walk Test to track improvement possibilities ( Harari et al., 2020; Thakkar et al., 2020; Sohn et al., 2021). Understanding the factors determining these outcomes may help physicians to streamline the treatment plan and efficiently deploy rehabilitation resources. Clinicians utilize the findings of these standardized tests to make decisions about their patient’s functional skills ( Jiang et al., 2021). Early estimation of a patient’s discharge score enables medical professionals to establish realistic rehabilitation plans.
Acute ischemic stroke (AIS) risk increases globally, presenting severe concerns for healthcare systems ( Sale et al., 2018). In recent years, the healthcare industry has made significant progress toward a data-driven approach to advance precision medicine to a greater level ( Shah et al., 1989; Lee et al., 2016; Iwamoto et al., 2022). The systematic development and utilization of risk prediction tools may enhance the implementation of preventative measures. However, risk prediction methods have numerous shortcomings. Artificial intelligence (AI)-based applications may strengthen risk assessment and the functional recovery of stroke patients (SPs) ( Lin et al., 2018). Machine learning (ML) techniques employ computational algorithms to establish associations between the factors and parameters for predicting the functional outcome of AIS ( Lin et al., 2021). Most ML-based algorithms are employed for classification and regression. Classification tasks divide input data into groups based on the labels, whereas regression tasks attempt to estimate a numerical value for the predicted output. Despite many similarities, ML differs from standard statistical methods ( Campagnini et al., 2022). Traditional statistical methods focus on a fixed model and treat data as normally distributed. On the other hand, ML is based on empirical evidence ( Campagnini et al., 2022). ML-based methods have produced a significant improvement in cardiovascular disease and stroke functional outcome prediction.
Researchers and practitioners in the field of ML face challenges with data preparation due to missing information ( Chen et al., 2022; Liao et al., 2022). In a wide variety of applications and industries, including data mining and the scientific disciplines, there are varying degrees of missing information. The lack of data may weaken the reliability of an analysis and lead to inaccurate inferences. Therefore, it is crucial to deal with missing data effectively. Deep neural networks (DNNs) are an effective tool for uncovering unseen nonlinear temporal patterns in time series data ( Morris et al., 2001; George et al., 2017b; Ohura et al., 2017; Chang et al., 2021; Katsuki et al., 2021). Recently, there has been a lot of interest in using deep learning (DL) models for data infilling alongside massive volumes of acquired sensor data ( George et al., 2017a; Rafiei et al., 2019; Razak et al., 2022). Recently, data-driven models gained popularity for imputing missing data for the reason that they do not presume stationary or linear data and need less domain expertise. Tan et al. (2020) suggested a model for time series data imputation using a bidirectional recurrent neural network. During model training, their method estimates and adjusts the imputed values. In the study ( Ma et al., 2018), the authors developed a multi-directional recurrent neural network to interpolate and impute across data streams. Attention approaches have proved effective in machine translation and computer vision ( Choi et al., 2016; Suo et al., 2017; Heo et al., 2018; Mallick et al., 2021). However, the time intervals in their data or the lack of a time attribute make them inappropriate for electronic health record (EHR) information. EHR data attention has been studied in many studies.
The earlier identification of SPs’ functional outcomes assists clinicians in facilitating an effective rehabilitation environment. The heterogeneity of the stroke impacts among the patients plays a significant role in predicting the functional consequences of SP. The recent prediction models offer a supportive rehabilitation framework for the patients. The existing studies demand a huge computation cost for generating an outcome. In addition, there is a lack of large datasets for training the ML techniques in order to produce an optimal outcome. Due to the considerable computation cost, rehabilitation centers face challenges in implementing the prediction frameworks. In addition, the limitations of DL methods reduce the performance of the frameworks. Thus, there is a demand for lightweight DL-based prediction frameworks for identifying the functional outcomes of SP.
In this study, the researchers propose a framework for identifying the functional outcomes of SPs. The proposed framework employs an attention-based Bi-directional Gated Recurrent Unit (A-Bi-GRU) and a shallow-convolutional neural network (S-CNN) to derive insight from the SP data. The contributions of the study are
An A-Bi-GRU-based data imputation technique to address the data irregularities in SP EHR data,
A functional outcome prediction model using the S-CNN technique,
Performance evaluation of the proposed framework using the benchmark metrics by comparing the existing state-of-the-art methods.
The remaining part of the study is structured as follows:
The following section outlines the importance of predicting the functional outcomes of SPs in rehabilitation services. The process of data acquisition and methodology of the proposed framework is explained in the Materials and Methods section. The Results section describes the experimentation and results. The outcome of the comparative analysis and features of the proposed framework are discussed in the Discussion section. Finally, the Conclusion section highlights the study’s contribution and future directions.
RELATED STUDIES
Supervised learning techniques employ data with ground-truth labeling ( Rafiei et al., 2019; Koch et al., 2021; Rajashekar et al., 2021). In the learning process, classification and regression are often carried out in tandem with the simultaneous provision of input and output values ( Kim et al., 2022). In reinforcement learning, a predefined agent considers its present conditions before choosing an action, or series of activities, from a set of options that maximizes rewards in a specific environment. In contrast to supervised learning, reinforcement learning requires only a training set consisting of input and output pairs or stated proper and improper behaviors ( Kim et al., 2021).
Stroke prognosis models demand enormous computational resources that challenge the healthcare centers in implementing the prediction. Kim et al. (2021) developed a model to predict upper and lower extremity function using EHR data. The model yielded area under receiver operating characteristic (AU-ROC) curve in the range of 0.736 to 0.836. The DNN performed better among the random forest (RF), logistic regression (LR), and DNN model generation approaches. Bonkhoff and Grefkes (2022) employed 2604 patients’ data and used 38 variables, including age, sex, time from the onset at admission, blood pressure, and previous medical history for predicting motor outcomes. Motor outcomes were categorized as “favorable” or “poor” based on the modified Rankin Scale scores.
DL is a class of ML algorithms that attempts to generalize to more significant abstractions by employing a series of nonlinear transformations ( Shah et al., 1989; Sale et al., 2018; Iwamoto et al., 2022). DL techniques can handle newly acquired information in computer vision, speech recognition, natural language processing, and voice/signal processing. DL-based models can uncover variables’ relationships and locate meaningful information in time series, clinical, and imaging data ( Lee et al., 2016; Lin et al., 2018, 2021). These models are beneficial for analyzing massive data and discovering meaningful information in image data, while traditional ML may be more suited for smaller datasets. SP clinical data and images are generated in enormous amounts. Thus, DL-based models can assist stroke rehabilitation by processing clinical data ( Campagnini et al., 2022).
Lin et al. (2018) recruited 313 SPs and gathered their clinical data related to the impact of stroke in the early stages ( Lin et al., 2018). They employed input variables, including the Barthel Index, Berg balance scale, Fugl–Meyer evaluation, 6-minute walk test, mini-mental state examination, aphasia test, and dysphagia scale. RF, LR, and Support Vector Machine (SVM) achieved an AU-ROC curve of 0.792, 0.762, and 0.774, respectively. Modeling and predicting functional outcomes after stroke using a binary (positive or negative) classification or particular score outcomes is a common use of traditional statistical tools like linear regression ( Lin et al., 2018). Based on conventional scales and pertinent clinical data, these models neglect to include crucial aspects that hinder patient-specific rehabilitation routes, such as family or community support and cultural level ( Lin et al., 2021).
Most research has focused on forecasting the prognosis following a stroke using magnetic resonance imaging or EHR data. Researchers intend to build applications to improve SPs’ diagnosis, treatment, and rehabilitation. Reliable risk prediction algorithms have been developed as resources for avoiding strokes ( Chen et al., 2022). Stroke prevention is an essential component in the overall effort to lower the worldwide burden of this condition. Strategically adopting and developing AI-driven prediction tools can significantly contribute to an effective rehabilitation environment ( Liao et al., 2022). On the other hand, ML algorithms and other forms of AI-based correlation modeling may examine heterogeneous data, map nonlinearities between several input and output variables, and uncover patterns across a wide range of clinical outcomes ( Ohura et al., 2017).
The existing studies ( Morris et al., 2001; George et al., 2017a, b; Ma et al., 2018; Rafiei et al., 2019; Tan et al., 2020; Chang et al., 2021; Katsuki et al., 2021; Razak et al., 2022) applied Artificial Neural Network (ANN), DNN, K-Nearest Neighbor, Adaptive Boosting (AdaBoost), and SVM, LR for developing the functional outcome prediction. In addition, multiple regression (R 2) is applied to find the relationship between input variables related to SPs.
In order to improve the efficiency of the classification techniques, Choi et al. (2016) developed an attention network to identify essential visits and critical variables ( Choi et al., 2016). In addition, they proposed a graph-based attention model to learn robust EHR data representations. A similar knowledge-based attention approach for embedding nodes in the knowledge graph was introduced by Ma et al. (2018) and Suo et al. (2017). Heo et al. (2018) employed the attention mechanism to address the issue of poor measurement quality. To a certain degree, the attention processes can potentially improve the performance and interpretability of classification models ( Heo et al., 2018). Mallick et al. (2021) introduced three attention techniques to monitor health problems and assess links between present status and historical state using the RNN technique ( Mallick et al., 2021). However, the irregularly sampled EHR data present a significant challenge for attention-based methods because they are typically developed for regular time series data.
Recently, GRU-based attention mechanisms have been used to overcome the challenges of time irregularities and missing data imputation. In order to better impute missing data as the average decay of prior input values, GRU augments traditional GRU with observed recordings and corresponding timestamps. However, GRU uses the empirical mean value and the previous observation to impute missing values. This technique is incapable of capturing information on the global structure of sequence data. Additionally, it fails to consider degrees of reliability, especially records that are more likely to be inaccurate than actual ones. Therefore, it offers equal weight to both observed and imputed data, severely degrading its performance.
Furthermore, predictors of quality of life (QOL) have been the subject of a few research studies ( Lee et al., 2016). Multiple predictors, such as age, gender, vascular risk factors, clinical scales, neuropsychological testing, and lesion characteristics, were evaluated in these studies. However, due to the diversity of these studies, it was challenging to identify common predictors of QOL outcomes ( Lee et al., 2016). QOL after stroke is predicted by both physical and psychological aspects. QOL improvements during stroke rehabilitation are crucial. However, which patients would reap the most benefits from a particular therapy is not well understood, and statistical methods may not be the best way to uncover these relationships and predictions. Predictors of QOL performance following recovery were rarely explored. There is a need for more research to clarify the predictive capacity of various QOL indicators in SPs. The algorithms’ complexity also hampered the practical use of outcome predictors. To enhance the efficacy of rehabilitation treatment, it may be possible to lessen the complexity of clinical implementation by developing a predictive algorithm based on current and straightforward algorithms.
The findings of the state-of-the-art ML-based prediction model highlighted the importance of AI techniques in predicting the functional recovery of SPs. However, lack of computational efficiency, missing data, healthcare data privacy policies, and unique symptoms of stroke impact are the common factors influencing the performance of functional outcome prediction models. In addition, there is a demand for DL-based classification techniques to offer effective services for predicting functional recovery ( Table 1).
Characteristics of the existing literature.
| Authors | Methodology | Dataset | Results |
|---|---|---|---|
| Campagnini et al. (2022) | Cross-validated multiple classification techniques and computed weight voting of classifiers | 278 patients’ data were included in the study | RF-based classifiers outperformed SVM, LR, and KNN classifiers |
| Chen et al. (2022) | Employed RF, ANN, RF, and SVM models for predicting readmission possibilities for SP | 1476 patients were enrolled in the study | ANN outperformed the existing classifiers by achieving an accuracy of 93% |
| Liao et al. (2022) | Applied multiple classification techniques for predicting QOL | 132 AIS individuals were included in the study | RF obtained an accuracy, precision, and F1-score of 85, 88, and 85% |
| Kim et al. (2022) | Applied DNN, LR, and RF for motor function prediction | 833 patients were included in the study | DNN outperformed RF and LR classifiers |
| Iwamoto et al. (2022) | Developed ML-based classifier for functional recovery prediction | 71 patients participated in the study | The classifier obtained an accuracy of 81.7% with an AU-ROC of 0.89 |
| Kim et al. (2021) | Predicted motor function outcome using RF and DNN | 1056 SPs data were used for evaluating the classifier’s performance | DNN and RF achieved an accuracy of 82.2 and 80.2%, respectively |
| Sohn et al. (2021) | Applied ANN and SVM classifiers for predicting disease prognosis | 181 SPs participated in evaluating the model’s performance | ANN achieved a better accuracy and sensitivity of 92 and 90% |
| Jiang et al. (2021) | Developed an ML-based technique for functional outcome prediction | 1035 patients were included in the study | Achieved a reasonable R 2 = 0.32 |
| Thakkar et al. (2020) | Developed a motor prediction model using KNN and ANN | 239 patients aged between 11 and 54 years participated in the study | KNN outperformed ANN by obtaining an accuracy of 85.4% |
| Harari et al. (2020) | Developed a prediction model using Lasso regression | Evaluated the model’s performance with 50 stroke survivors | Achieved a superior R 2 value of 77% |
| Lin et al. (2018) | Employed classification and regression for predicting patients’ status | 313 SPs participated in the study | RF achieved an effective AU-ROC of 0.79 |
| Sale et al. (2018) | Functional outcome prediction mode using SVM | 55 AIS patients participated in this study | Predicted the functional outcome with an average accuracy of 90% |
Abbreviations: AIS, acute ischemic stroke; ANN, artificial neural network; AU-ROC, area under receiver operating characteristic curve; DNN, deep neural network; KNN, K-nearest neighbor; LR, logistic regression; ML, machine learning; RF, random forest; SP, stroke patient; SVM, support vector machine; QOL, quality of life.
MATERIALS AND METHODS
The researchers propose a prediction model based on A-Bi-GRU and CNN techniques. They obtained the institutional review board (IRB) approval (IRB23-031) from AlMaarefa University, Saudi Arabia. They followed the data collection procedures following the ethical standards of AlMaarefa University. Three rehabilitation units across India were requested to provide SP data. Data were gathered between December 2018 and July 2022. Table 2 presents the inclusion and exclusion criteria for collecting the patient’s data.
Inclusion and exclusion criteria.
| Inclusion | Exclusion |
|---|---|
| 1. AIS patients aged >18 years.
2. Patients admitted to the rehabilitation centers between December 2018 and July 2022. 3. Patients received at least 5 and 30 h of rehabilitation daily and weekly | 1. Patients with typical complications such as cancer, visual impairment, psychiatric disorders, and renal failure.
2. Patients with exceptional social circumstances that affect rehabilitation. 3. Patients who experience accidental deterioration prior to outcome assessment have fewer rehabilitation opportunities due to additional treatment for uncommon complications. |
Abbreviation: AIS, Acute ischemic stroke.
Upon admission and the day of discharge, every patient was given a functional assessment to categorize their level of disability. Clinical assessment and subsequent interviews were performed to collect the data. The examiner was a highly qualified medical professional specializing in physical medicine and rehabilitation. During their time at the rehabilitation center, each test subject participated in daily therapy, which may have consisted of physiotherapy, occupational therapy, or speech therapy, based on the requirements of the individual. The modified Barthel Index (MBI) was adopted to determine the outcomes of all patients after they were discharged to their homes. Figure 1 outlines the stroke severity level based on MBI.
Biomarkers
Data from 432 SPs were collected from the rehabilitation centers. The researchers included 356 SPs in the study based on the inclusion criteria. Figure 2 outlines the critical biomarkers of SPs. Nine biomarkers, including age, gender, the existence of bedsores and bladder catheter, communication disabilities, comorbid conditions, Alzheimer’s complications, cognitive disability, and pain, were extracted from the SP data.
Figure 3 highlights the proposed framework for identifying the functional outcomes of SPs. The researchers apply A-Bi-GRU to overcome the data irregularities in SP EHR data. S-CNN is used to classify SPs into five significant classifications, as shown in Figure 3. Based on the study ( Tan et al., 2020), the data irregularities are addressed at the feature level to improve the prediction accuracy.
Data imputation
SP EHR presents essential items that represent the SP’s health status. However, healthcare centers face challenges in offering a complete set of SP data. In order to overcome the challenge, the researchers propose a data imputation technique using A-Bi-GRU. The method supports the prediction model for classifying the functional outcomes of SPs. Equation (1) presents the mathematical expression of SPs using SP EHR and demographic data.
where P ( t) is the SP, P1(t), …..Pn(t) is the SP EHR data at time t, n is the total number of patients, and d 1, …… d u is the unique (u) demographic data at time t.
The stroke progression model using the loss function L( ) is represented in Equation (2).
where P is the SP, Δ t is the stroke severity level at different time intervals, and out is the functional outcome of SP.
Equation (3) presents the covariance matrix for showing the health status of an SP at different periods.
where M(TMn,TMn) is the covariance matrix between the specific period, λ 2 I N is the unit matrix, and Cov[ P *] is the covariance matrix using Gaussian distribution.
However, imputed values are not reliable compared to the actual values. Thus, the unreliability score is computed to determine the reliability of the imputed values. Equation (4) is the mathematical expression for calculating the unreliability score.
where M t ( P *) is the unreliability score matrix. SP data at different time intervals and unreliability scores are passed as inputs to A-Bi-GRU. Equation (5) represents the decay functions’ mathematical expressions for adjusting the A-Bi-GRU cells’ hidden status.
where D c is the decay function at Δ t.
Equations (6), ( 7), and ( 8) show the forward and backward gates and the complete A-Bi-GRU expressions (without attention mechanism) for imputing the missing data.
where ←Ht−1, →Ht−1 , and H t –1 are the gates of A-Bi-GRU and DcΔt is the decay function at a specific interval.
Equation (9) generates the unreliability aware weights using the outcome of A-Bi-GRU.
where Con t is the conversion function for the specific time interval. In order to improve the performance of the functional outcome computation, the unreliability aware weights are converted using the attention mechanism. Equation (10) presents the attention mechanism for data imputation.
where a t is the attention mechanism for computing the missing value; the sigmoid function is used to make binary decisions using the unreliability aware weights.
Functional outcome prediction
An S-CNN requires a single hidden layer to generate results for complex problems. The architecture of S-CNN supports the developers to build a lightweight application for extracting the patterns from the images. Moreover, it demands limited resources compared to DNN techniques. The researchers built the S-CNN with a single hidden, two ReLu, and one fully connected layer with a softmax function to predict the functional outcomes. The hidden layer applies Equation (11) to predict the functional outcomes.
where f out is the functional outcome and a t and awt are the outcomes and weight-adjusted outcomes of A-Bi-GRU, respectively.
A softmax function is used for generating the multi-class classification based on MBI. Equation (12) presents the softmax function to predict the functional outcome.
where f out and b are the functional outcome matrix and vectors, accordingly. Finally, Equation (13) shows the cross-entropy for computing the classification loss.
where N M is the number of iterations in the Mth sample, and f act and f out are actual and predicted functional outcomes, respectively.
Performance evaluation
In order to evaluate the performance of the proposed method, the researchers employ the benchmark metrics, including accuracy, precision, recall, F1-score, specificity, and sensitivity. Cross-entropy is used to compute the computation loss. In addition, AU-ROC and Area Under Precision Recall Curve (AU-PRC) are generated to determine the classification accuracy of the proposed framework.
RESULTS
The researchers developed the proposed framework in Python 3.10.0, Windows 10 Professional, 8GB RAM, and i7 processor. In addition, Keras (2023) and Tensorflow (2023) libraries are used for building the A-Bi-GRU and S-CNN models. The researchers followed fivefold cross-validation for evaluating the performance of the proposed framework. A total of 35 epochs was used for training the proposed framework. The researchers employed the baseline models, including RF, SVM, AdaBoost model (ABM), and LR, to evaluate the proposed framework’s performance. The proposed framework achieved a superior outcome at the 35th epoch. However, the researchers extended the training process up to 42 epochs. There is no significant change in the outcome. Thus, the parameters of the 35th epoch were fixed for executing the results.
Table 3 presents the performance evaluation findings of the proposed framework. The findings show the significance of the proposed framework. The proposed framework achieved a better accuracy for the “fully independent” class. It obtained an average accuracy, recall, precision, F1-measure, specificity, and sensitivity of 98.18, 97.48, 98, 97.74, 96.74, and 97.24, respectively. The proposed data imputation assisted the proposed framework by providing the effective missing values. Figure 4 outlines the performance of the proposed framework.
Performance evaluation outcome.
| Functional outcomes/metrics | Accuracy | Recall | Precision | F1-measure | Specificity | Sensitivity |
|---|---|---|---|---|---|---|
| Unable to perform tasks | 98.6 | 97.4 | 98.1 | 97.75 | 96.8 | 97.9 |
| Attempts task but unsafe | 98.9 | 97.6 | 98.4 | 98 | 95.7 | 96.5 |
| Moderate help required | 97.4 | 97.1 | 97.6 | 97.35 | 96.4 | 95.8 |
| Minimal help required | 97.8 | 97.8 | 98.3 | 98.05 | 97.3 | 98.4 |
| Fully independent | 98.2 | 97.5 | 97.6 | 97.55 | 97.5 | 97.6 |
| Average | 98.18 | 97.48 | 98 | 97.74 | 96.74 | 97.24 |
Table 4 highlights the findings of the comparative analysis. It shows the average accuracy, recall, F1-measure, specificity, and sensitivity during fivefold cross-validation. It is evident that the proposed framework outperformed the existing classifiers. The proposed S-CNN predicted the functional outcome with high accuracy. In addition, the proposed data imputation technique has improved the performance of the classifiers, significantly. Figure 5 presents the performance of the functional outcome predictors. The proposed framework achieved superior accuracy, recall, precision, F1-measure, specificity, and sensitivity of 98.18, 97.48, 98, 97.74, 96.74, and 97.24.
Comparison analysis outcome.
| Functional outcomes/metrics | Accuracy | Recall | Precision | F1-measure | Specificity | Sensitivity |
|---|---|---|---|---|---|---|
| RF | 96.3 | 96.1 | 95.4 | 95.75 | 96.2 | 95.8 |
| SVM | 94.8 | 95.3 | 96.3 | 95.8 | 95.3 | 97.6 |
| AdaBoost | 96.1 | 96.5 | 96.5 | 96.5 | 94.7 | 96.4 |
| PSO | 96.8 | 96.7 | 95.4 | 96.05 | 96.5 | 95.7 |
| Proposed framework | 98.18 | 97.48 | 98 | 97.74 | 96.74 | 97.24 |
Abbreviations: AdaBoost, adaptive boosting; PSO, particle swarm optimization; RF, random forest; SVM, support vector machine.
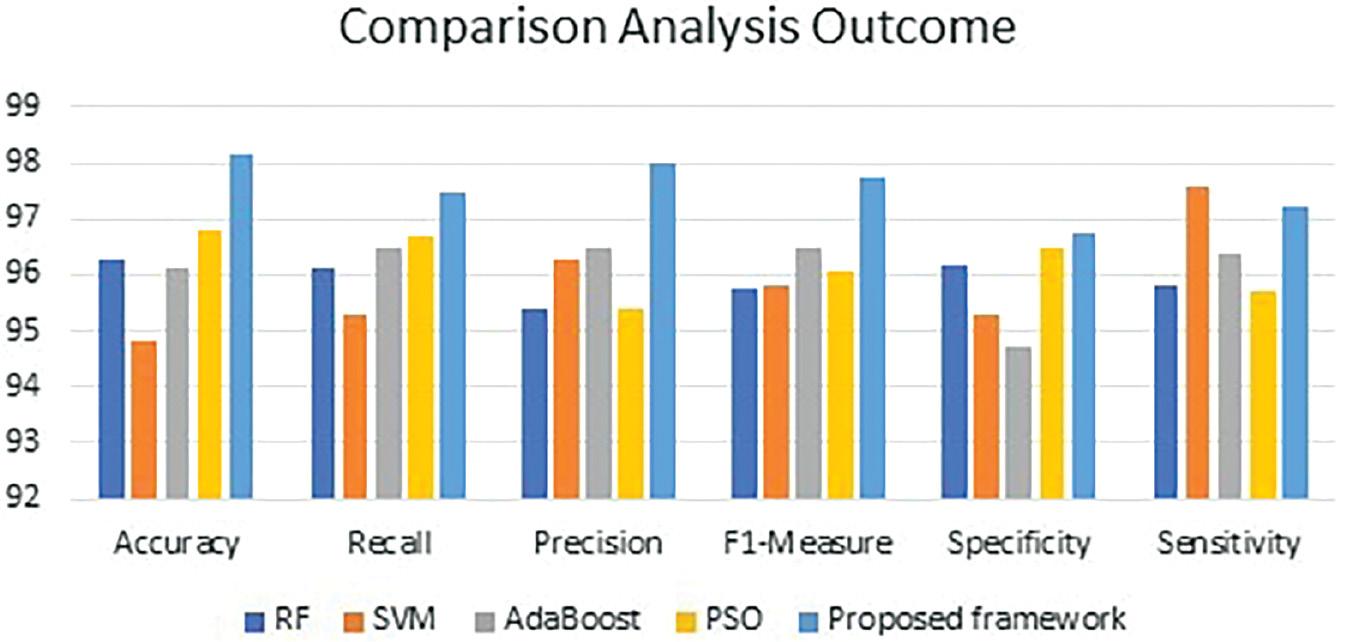
Comparative analysis outcome. Abbreviations: RF, random forest; SVM, support vector machine.
Table 5 presents the models’ computation loss for each MBI classification. The data imputation technique enhanced the classifier to achieve an optimal outcome with less computation loss. The proposed model obtained a lower loss of 12.4 for the “Unable to perform tasks” classification whereas produced a maximum loss of 13.4 for the “Moderate help required” classification.
Computation loss of the prediction models.
| Functional outcomes/metrics | Unable to perform tasks | Attempts task but unsafe | Moderate help required | Minimal help required | Fully independent | Average |
|---|---|---|---|---|---|---|
| RF | 17.2 | 18.5 | 19.5 | 15.9 | 16.4 | 17.5 |
| SVM | 21.3 | 23.6 | 21.8 | 21.5 | 19.4 | 21.52 |
| AdaBoost | 18.6 | 19.6 | 19.7 | 18.6 | 17.5 | 18.8 |
| PSO | 16.4 | 15.9 | 15.4 | 15.7 | 16.2 | 15.92 |
| Proposed framework | 12.4 | 13.8 | 13.4 | 12.9 | 12.7 | 13.04 |
Abbreviations: AdaBoost, adaptive boosting; PSO, particle swarm optimization; RF, random forest; SVM, support vector machine.
Table 6 outlines the computational costs of the prediction models for generating the results. RF generated the results in a computation time of <0.156 seconds. However, it consumed a memory of 14.5 MB. On the other hand, the proposed framework comprises fewer hidden layers achieving better results in 0.161 seconds with a memory size of 12.4 MB.
Computational costs.
| Functional outcomes/metrics | Average computation time (seconds) | Parameters | Memory size (MB) |
|---|---|---|---|
| RF | 0.156 | 12 M | 14.5 |
| SVM | 0.415 | 23 M | 18.6 |
| AdaBoost | 0.368 | 16 M | 24.3 |
| PSO | 0.512 | 19 M | 16.2 |
| Proposed framework | 0.161 | 9 M | 12.4 |
Abbreviations: AdaBoost, adaptive boosting; PSO, particle swarm optimization; RF, random forest; SVM, support vector machine.
Figure 6 presents the AU-ROC and AU-PRC of each prediction model. The proposed framework achieved AU-ROC and AU-PRC of 0.95 and 0.86, respectively. In contrast, RF, SVM, AdaBoost, and particle swarm optimization (PSO) obtained AU-ROC and AU-PRC of 0.81 and 0.87, 0.85 and 0.88, 0.93 and 0.86, and 0.92 and 0.86, respectively.
DISCUSSIONS
There is a demand for an automated tool for rehabilitation centers to derive insights from the SP data to treat them effectively. In this study, the researchers developed a DL-based framework to predict functional recovery for SPs. The researchers employed A-Bi-GRU and S-CNN for building the prediction framework. They obtained the dataset of 356 AIS patients from three rehabilitation centers across India. The findings present a solution for the rehabilitation center to improve its services. In addition, it facilitates an efficient technique for imputing the missing data in larger datasets.
Iwamoto et al. (2022) developed a clinical rule for determining the functional improvement of SPs. Similarly, the proposed study developed an integrated framework for data imputation and functional outcome prediction. In the study ( Ohura et al., 2017), the authors applied MBI for stroke impact analysis. Likewise, the proposed study determined the impact of the stroke using MBI. Campagnini et al. (2022) used the Barthel Index for predicting the functional outcome of SPs. In contrast, the proposed framework utilized MBI for determining the functional recovery. It outperformed the balanced voting technique of Campagnini et al. (2022) by achieving an average accuracy of 98.18. Chen et al. (2022) utilized 1476 patients’ data to predict the possibility of readmission for SPs. The performance of their model was limited compared to the proposed framework.
Most studies ( Shah et al., 1989; Lee et al., 2016; Sale et al., 2018; Thakkar et al., 2020; Jiang et al., 2021; Sohn et al., 2021; Iwamoto et al., 2022) applied RF to identify the functional recovery and possibilities of readmission of SPs. RF and ANN achieved an effective outcome due to their internal structure that supports decision-making. In contrast, the proposed framework applied an attention mechanism for determining the missing value. The unreliability score supported the data imputation technique to replace a reliable value in the missing value position.
The proposed framework offers an effective approach to address the missing data. Using this approach, the researchers can employ a larger dataset for developing DL-based techniques. It obtained a superior outcome in a smaller dataset. However, it can be implemented in real-time application. The proposed data imputation technique can support the proposed functional outcome prediction to deal with a complex dataset. Early acute stroke detection reduces mortality and disability. In the stroke medical care paradigm, the proposed framework may aid in healthcare data classification, Alberta stroke program early computer tomography Score grading, and prognostication. In addition, the proposed framework may assist neurologists in the identification of individuals who are suffering from AIS owing to major vessel occlusions.
Protection requirements for personally identifiable information may be a barrier to data exchange. For instance, the European Union’s General Data Protection Regulation (GDPR) comprises a complete set of restrictions for the collecting, storage, and use of personal information that will have various effects on the use of AI in healthcare. Healthcare centers need to obtain permission from individuals before collecting personal data under the GDPR. It additionally provides individuals with the right to monitor their data. The goal of these rules is to ensure the privacy of patients’ information; however, they place constraints on how medical professionals and researchers can employ this information. The use of natural language processing to automatically extract information on lifestyle modification evaluation using EHR is a potential area for further study. These types of studies may present therapeutic practice and increase the knowledge of functional outcomes to support healthcare practitioners. Knowledge gained from these kinds of research studies may be used to enhance the quality of treatment.
The findings revealed the significance of the proposed study in predicting the functional recovery of SPs. However, there is a demand for future studies to improve the performance of the proposed system. The limitations of S-CNN may influence the outcome of the proposed study. Furthermore, a larger dataset is required to evaluate the effectiveness of the suggested framework.
CONCLUSION
In this study, the authors proposed a framework for predicting the functional outcomes of stroke survivors in rehabilitation centers. An attention-based bidirectional long-short-term memory and S-CNN are used for implementing the prediction model. The authors employed 356 stroke survivors’ data in order to evaluate the performance of the proposed model. Benchmark metrics and baseline models are utilized for comparing the proposed model’s performance. The findings highlighted the significant role of the proposed framework in predicting the functional recovery for stroke survivors. The proposed functional outcome prediction model outperformed the state-of-the-art prediction techniques. The attention mechanism assisted the data imputation technique for finding reliable data. In addition, S-CNN predicted the optimal functional outcome using MBI. The structure of the S-CNN model has significantly reduced the computation resources. Furthermore, the data privacy policies of healthcare centers affect the researchers’ efforts in developing DL-based prediction models. However, the recent techniques offer an opportunity to generate artificial data for training the AI-based applications. The proposed framework can assist the researchers in extending their research in stroke rehabilitation techniques.






