Background
Intravascular lymphoma (IVL) is an unusual form of extranodal non-Hodgkin’s lymphoma that selectively invades the lumina of small blood vessels, especially capillaries [1]. IVL with central nervous system (CNS) manifestation is an extremely rare condition as IVL is usually found with systemic lesions and is commonly diagnosed by autopsy attributed to its myriad clinical presentation and non-specific laboratory findings [1]. Nevertheless, recent data has reverted this trend by reporting antemortem diagnosis in approximately 80% of patients [2]. This condition suggests that the natural history of intravascular large B-cell lymphoma (IVLBCL) has been substantially enhanced by advancements in early clinical identification and treatment [2]. Neurological manifestations comprise seizure, dementia, sensory or motor deficits, altered sensorium, ataxia, vertigo, and transient visual loss. Syncope may also be one of the earliest signs of CNS IVLBCL as reported by Awad et al. [3] in 2021.
This case illustrates a 49-year-old Thai lady who had IVLBCL with CNS involvement manifesting as a behavioral abnormality, cognitive impairment, and seizures. It is also important to highlight the role of a high index of diagnostic suspicion in order to expedite diagnostic investigations.
Case Presentation
This case involves a 49-year-old lady of Thai nationality from a middle-class socioeconomic background who presented with a one-day history of abnormal behaviors and forgetfulness. At the onset of the disease, she developed impaired recent memory (forgot where and who she is). She had repetitive actions such as opening and closing the car doors without any purpose. A few hours later, she experienced three episodes of generalized tonic-clonic seizures and was subsequently brought to the hospital to seek medical attention. There was no history of fever, headache, and vomiting. Her family members did not know of any history of focal neurological deficit, myoclonus, or prior comorbidity. She was restless and her Glasgow Coma Scale remained eye-opening at 4, verbal 2, and motor 5 postictally. General physical examination was unremarkable. A neurological examination revealed that she was confused and uncooperative for a higher mental function examination. Overall cranial nerves examination was normal. Power, tone, and deep tendon reflexes of upper and lower limbs were normal. Frontal release signs, including bilateral grasp and snout reflexes, were elicited. Extrapyramidal, cerebellar, and autonomic examinations were unable to be carried out without the patient’s cooperation. An urgent lumbar puncture was done to rule out meningoencephalitis after a normal computed tomography brain finding. Cerebrospinal fluid (CSF) opening pressure was 16 cmH2O while her CSF analysis was aseptic with minimally raised protein level and normal glucose ratio (Table 1). CSF venereal disease research laboratory and viral studies were negative. Antinuclear antibody (ANA) was found to be positive 1:320 (atypical speckled pattern). However, she did not meet the criteria for systemic lupus erythematosus. The remaining autoimmune workup was negative, and she has no clinical signs of autoimmune diseases. Her hepatitis B, hepatitis C, HIV, and paraneoplastic screen were negative. She underwent an electroencephalogram (EEG) as her seizures remained poorly controlled. The EEG showed cortical dysfunction with occasional sharp waves at the right frontoparietal region. Magnetic resonance imaging (MRI) of the brain (Figure 1) revealed non-enhanced biparietal lobe hyperintensities and focal hyperintensities over the right corpus callosum and adjacent right frontal horns associated with microhemorrhages which may suggest encephalitis or ischemia. She was unable to recognize her family members including her husband. She remained disorientated for a few weeks and subsequently slowly regained her alertness upon discharge. She was discharged with antiseizure medications (T. Levetiracetam 1.5 g BD, C. Phenytoin 300 mg ON, and T. Sodium Valproate 600 mg BD) after having completed treatment for presumed viral encephalitis.
| Lab parameters | Reference range | First admission in May 2022 | Readmission in September 2022 |
|---|---|---|---|
| Blood investigations | |||
| Haemoglobin | 12-18g/dl | 12 | 5.5 |
| Platelets | 150 × 103- 450 × 103 /µl | 200 | 70 |
| White cell counts | 4 × 103- 9 × 103 /µl | 10 | 8.4 |
| Sodium | 135-145 mmol/l | 135 | 130 |
| Potassium | 3.5-5.1mmol/l | 4 | 3.3 |
| Urea | 2.8-7.2 mmol/l | 5 | 4.3 |
| Creatinine | 59-104 µmol/l | 60 | 49 |
| Corrected calcium | 2.2-2.65 mmol/l | 2.32 | 2.42 |
| Total protein | 66-83 g/l | 70 | 71 |
| Albumin | 35-52 g/l | 30 | 19 |
| Globulin | 28-36 g/l | 40 | 57 |
| Total bilirubin | 5-21 µmol/l | 15 | 16.6 |
| Alanine transferase | 0-50 U/l | 20 | 18 |
| Alkaline phosphatase | 30-120 U/l | 100 | 107 |
| LDH | 105-333 U/l | 400 | 1,185 |
| Magnesium | 0.73-1.06 mmol/l | 0.75 | 0.69 |
| Inorganic phosphate | 0.81-1.45 mmol/l | 1 | 0.93 |
| Erythrocyte sedimentation rate | 0-20 mm/hour | 60 | 78 |
| C-reactive protein | <5mg/l | 50 | 191 |
| Blood culture | No growth | No growth | No growth |
| HIV/VDRL/hepatitis B/hepatitis C | Non-reactive | Non-reactive | Non-reactive |
| ANA | Negative | Positive 1:320 (atypical speckled) | Negative |
| Complement 3 | 0.1-0.4g/l | 0.248 | |
| Complement 4 | 0.9-1.8g/l | 1.191 | |
| Extractable nuclear antibodies Anti-Ds DNA Anti- Jo 1 Anti-Scl 70 Anti-smith Anti-RNP Anti-Ro Anti-La | Negative | All negative | |
| P-ANCA C-ANCA | Negative | Both negative | |
| Paraneoplastic panel (anti-YO, anti-HU, anti-Ri, anti-Ma2, anti-Tr, anti-GAD) | Negative | All negative | |
| Coombs test | Negative | Negative | |
| PBF | Normal | Bicytopenia. No blasts. Hypochromic microcytic anemia possible iron deficiency anemia. | |
| B12 | 145-569 pmol/l | 677 | |
| Folate | 10.4- 78.9 nmol.l | >42.9 | |
| Thyroid function test TSH T4 | 0.35-4.94mIU/l 9.01-19.05pmol/l | 2.03 13.02 | |
| Iron study Ferritin Iron Total iron binding capacity (TIBC) Transferrin saturation | 13-150 µmol/l 5.8-34.5 µmol/l 23-70 µmol/l 16%-45% | 1,161 3.2 23 14 | |
| Bone Marrow Aspirations (BMA) | Marrow tissues with diffuse infiltration by abnormal lymphoid cells with only sinusoidal involvement of the interstitium and they are positive for CD79a, CD20, MUM-1, CD138, BcL2, BcL6, Ki67 > 90% and CD5 which are consistent with High Grade Mature B Cell Lymphoma GCB Type. | ||
| Cerebrospinal fluid Cell count Cryptococcal antigen Glucose Protein Globulin Appearance Acid fast bacilli (AFB) Mycobacterium culture Culture Japanese encephalitis PCR Herpes simplex virus PCR varicellar zoster virus PCR Flavivirus PCR Cytology | 0 Negative 2.2-3.9mmol/l 0.15-0.45g/l Negative Clear No AFB No growth No growth Not detected Not detected Not detected Not detected | 0 Negative 3 0.64 Positive Clear No AFB No growth No Growth Not detected Not detected Not detected Not detected Occasional lymphocytes seen, no malignant cells. |
A month later, she was readmitted to the intensive care unit for status epilepticus. Her altered sensorium recurred. We noticed that she had new onset of bicytopenia (hemoglobin 5.5g/dl, white cell counts 8.4 × 103/µl, platelets 70 × 103/µl) with lactate dehydrogenase (LDH) of 1185 IU/l. Physical examination did not reveal any palpable lymphadenopathy or hepatosplenomegaly. An urgent peripheral blood film (PBF) did not find any blasts. We then proceeded with bone marrow aspirations and trephine biopsy (Figure 2), and indeed it showed marrow tissues with diffuse infiltration by abnormal lymphoid cells with only sinusoidal involvement of the interstitium and they are positive for CD79a, CD20, MUM-1, CD138, BcL2, BcL6, Ki67 > 90%, and CD5 which are consistent with high-grade mature B cell lymphoma germinal center B-cell Type. In view of only sinusoidal involvement in the trephine biopsy, IVLBCL is highly suggested. Contrast-enhanced computed tomography Thorax, abdomen, and pelvis were arranged and were negative for distant metastasis or lymphadenopathy. She was then referred to the hematology team and started on Rituximab cyclophosphamide, doxorubicin, vincristine, and prednisolone (R-CHOP) therapy. Of late, her condition is stable without seizures, and her confusion is improving.
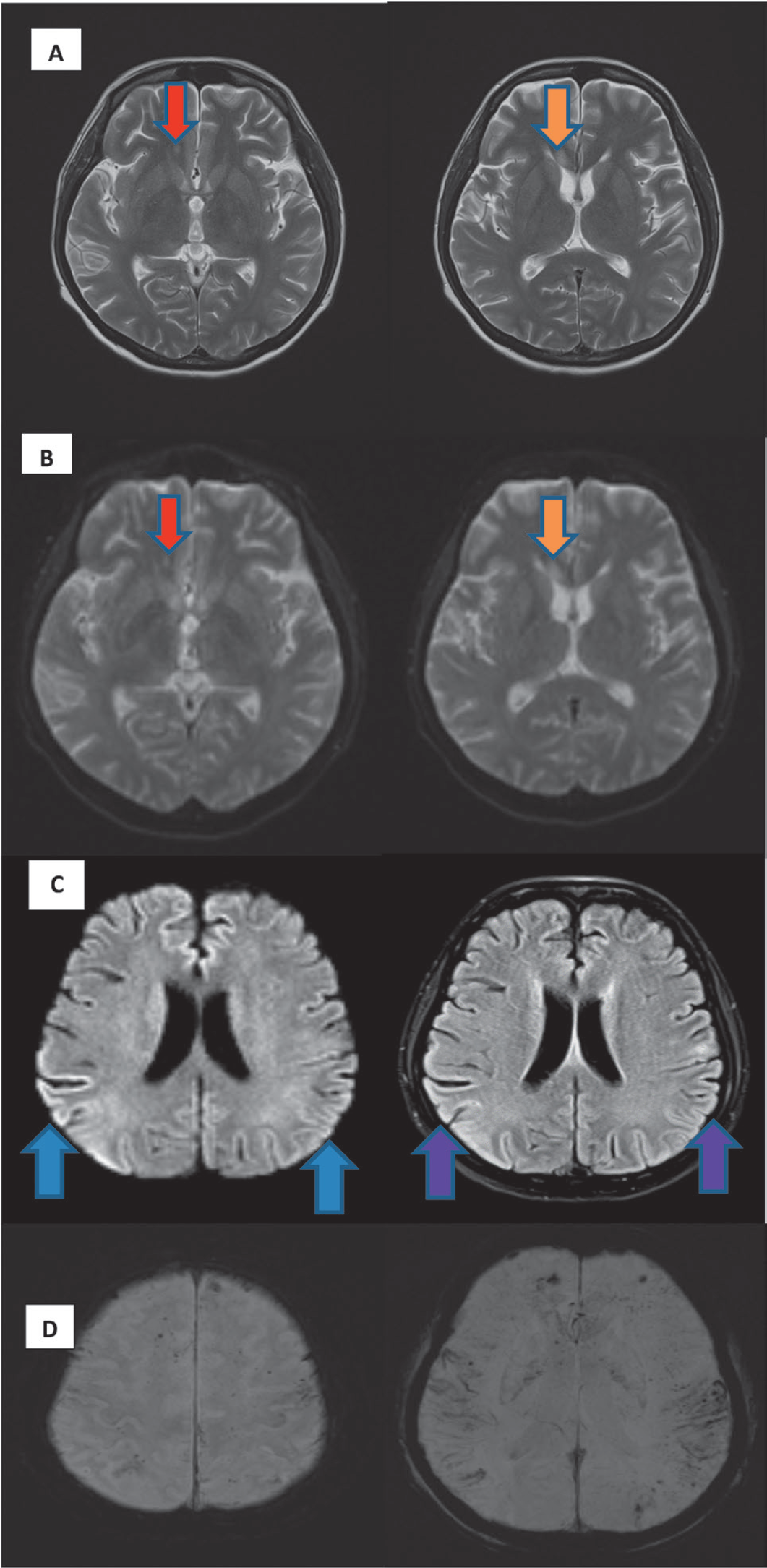
(A) MRI brain T2 weighted showed high signal in the right frontal white matter (red arrow) and right genu of corpus callosum (orange arrow). (B) These lesions at right frontal white matter (red arrow) and right genu of corpus callosum (orange arrow) also demonstrated high signal in DWI acquisitions. (C) MRI brain showed high signal in the gyri at bilateral parietal lobes (blue and purple arrows) in DWI (left) and FLAIR (right) acquisitions. (D) MRI brain susceptible weighted imaging acquisitions showed multiple blooming artifacts in keeping with microhemorrhages.
Discussion
IVLBCL is a rare clinical entity with heterogeneous clinical manifestations. There is selective growth of cancerous and atypical cells in the small and intermediate-sized vessels in various organs such as the CNS, lungs, skin, adrenal gland, liver, kidney, spleen, bone marrow, thyroid, pituitary, and gastrointestinal tract [4]. Diagnosis is made by organ biopsy and random skin biopsy from the uninvolved site in patients who presented with pyrexia of unknown origin [4]; 7 out of 32 patients with pyrexia of unknown origin who had random skin biopsies from unaffected sites were diagnosed to have IVL, according to Sitthinamsuwan et al. [5]. One-quarter of patients experienced early neurological manifestation in the form of altered sensorium, focal neurological deficit, dementia, and seizure. Our patient here had an initial neurological presentation which has led us to the investigation of neurological diagnosis. However, progressive reduction in blood cell count and raised LDH level as seen in our case report are telltale signs of IVLBCL [4].
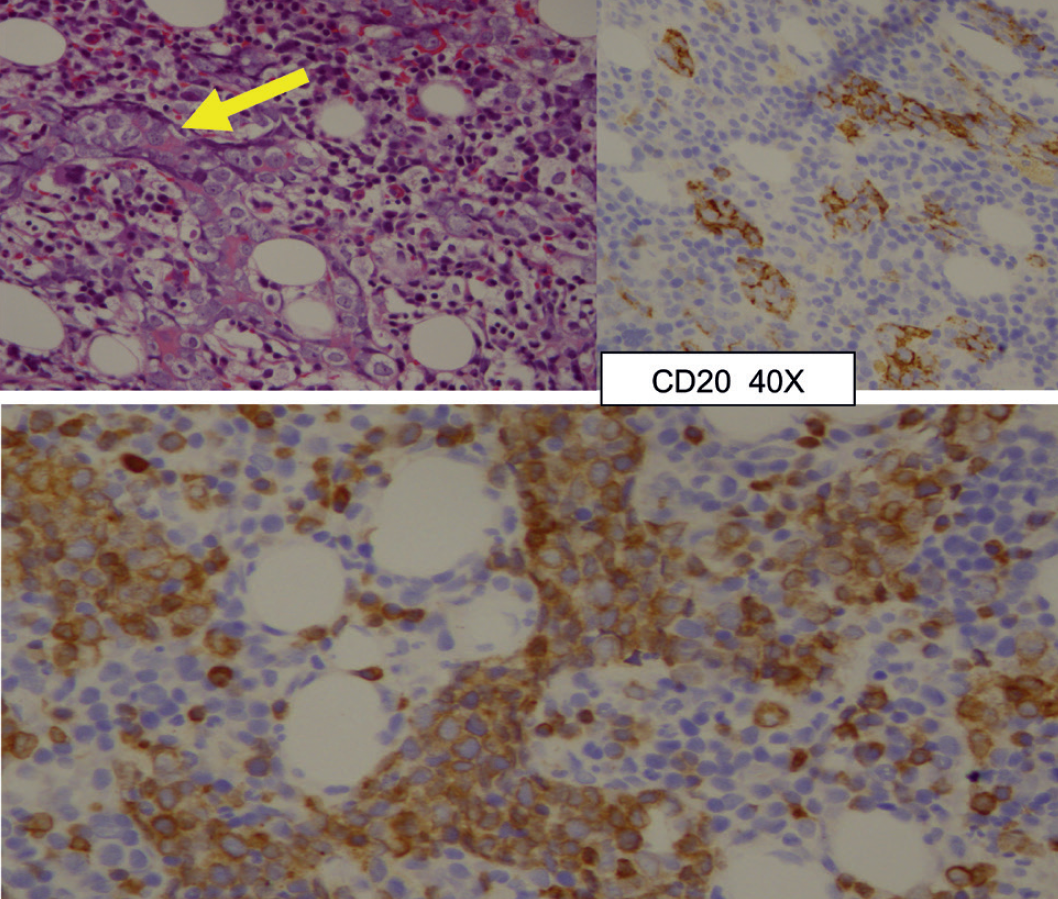
Marrow tissues show diffuse infiltrations by abnormal large lymphoid cells within sinusoid (yellow arrow) which are arranged in chords and aggregates, display moderate amount of cytoplasm, round nucleus, open chromatin pattern, and prominent nucleoli and they showed CD20 and CD79a positivity.
Current literature shows that IVLBCL tends to affect elderly patients aged 60-70 years old, with a male-to-female ratio of 1:2. It has been suggested that the incidence of IVLBCL is increased in Asian populations [6]. Our patient here is a middle-aged Thai lady who came in with seizures and altered sensorium. Initial diagnoses considered are usually stroke, encephalomyelitis, vasculitis, and multiple sclerosis. Therefore, many IVLBCLs are diagnosed at autopsy. MRI has been shown to be the best modality despite its low level of sensitivity and specificity. Hyperintense cortical or subcortical lesions on T2-weighted and fluid-attenuated inversion recovery imaging suggest small vessel ischemia, demyelination, or vasculitic changes [6]. These are usually reported MRI brain findings before IVLBCL is highly suspected as seen in our case. In a series of IVLBCL patients with CNS disease from Massachusetts General Hospital, it was found that six out of seven patients demonstrated CSF lymphocytosis, while only one of them had malignant cells in CSF cytology. Bone marrow infiltration has been reported in 38% of cases, and 5% of cases had circulating neoplastic cells on the peripheral blood smear [7]. Our patient has IVLBCL of CNS with bone marrow infiltration without showing abnormal blasts on PBF.
The clinical course of IVLBCL has been shown to be hostile and the prognoses of most cases are poor [8]. Data in the literature indicate that anthracycline-based regimens, such as Rituximab and R-CHOP, are the treatment of choice [8]. It also has been found that patients with IVLBCL within the CNS are unlikely to respond to R-CHOP attributed to its poor penetration of the blood-brain barrier, and they should be treated with regimens similar to those used in primary CNS lymphoma in which intrathecal or high-dose systemic methotrexate should be included [9]. Without treatment, the 1-year mortality rate is approaching 80%, while its average survival rate is about 1 year [10].
Conclusion
The unpredictable clinical manifestation and the lack of diagnostic protocols available have resulted in the postmortem diagnosis of IVLBCL. Most of the antemortem diagnoses of IVLBCL are made incidentally in biopsies performed for different reasons. We should always have a low threshold to biopsy abnormal brain or skin lesions in order to speed up the diagnosis process.

