Background
Multiple myeloma (MM) is typically characterized by the neoplastic proliferation of monoclonal plasma cells, nearly 17% of hematological malignancies [1]. MM is suggested by bone pain and lytic lesions on skeletal films or other imaging modalities, along with clinical presentations such as anemia, hypercalcemia, and acute renal failure. Increased clonal plasma cells in the bone marrow can result in extensive skeletal destruction with osteolytic lesions, osteopenia, and pathological fractures. The frequency of bone lesions was found in 20%-25% at the diagnosis in one large cohort [2]. In addition, rarely diffuse osteosclerotic bone lesions may be detected in MM [3]. However, the general approach for these lesions is to interpret them as metastases. Sometimes the differential diagnosis to search for a primary tumor causes wasting time. Therefore, while the specialists comment on the findings, they also should consider the rare findings of MM.
Case Presentation
Our initial case involves a 70-year-old male patient who has been referred to oncology. After a traffic accident, he reported knee pain and underwent magnetic resonance imaging which revealed round and oval-shaped infiltration areas in the bony structures of the knee joint. These areas merged in places and appeared hypointense on T1-weighted images and hyperintense on fat-suppressed T2-weighted images. Furthermore, after intravenous contrast material, the areas showed marked contrast brightening. The results have been interpreted as indicating metastasis (Figure 1). Widespread hypermetabolic lesions at the bone/marrow level were detected by positron emission tomography/computerized tomography (PET/CT). These lesions were also interpreted as metastatic bone lesions (Figure 2). Over the past 2 years, the patient has experienced weakness and a weight loss of 10 kg. During the upper gastrointestinal system endoscopy, no malignancies were found. The patient’s prostate-specific antigen (PSA) levels were normal, and the PET/CT scan did not reveal any clues regarding a possible primary tumor. Finally, the patient underwent a biopsy of bone lesions in interventional radiology and was referred to hematology during this period. We detected anemia, conducted nutritional tests for anemia, and found average levels of iron, vitamin B12, and folate. In addition, we found a reversed albumin-globulin ratio and a history of thyroid nodules with suppressed thyroid stimulating hormone, but the free T4 level was within the normal range. The results of the laboratory tests are presented in Table 1. While the serum protein electrophoresis showed that the monoclonal M spike was 1. 7 g/dl in the gamma fraction, immunoglobulin G-kappa monoclonal gammopathy was detected with serum immunofixation immune fixation electrophoresis. We diagnosed grade 1 reticulin fibrosis with 13% kappa-positive monoclonal plasma cell infiltration as small groups in the bone marrow through aspiration and biopsy. In addition, the results of his bone biopsy showed the existence of a plasmacytoma. Started weekly bortezomib-cyclophosphamide-dexamethasone (VCD) treatment. The patient developed peripheral neuropathy in the third cycle and switched to lenalidomide and dexamethasone (Rd). After the third cycle of Rd, partial remission occurred with stable sclerotic lesions and negative flourodeoxyglycose (FDG)-uptake. He has been undergoing treatment and follow-up. The second patient, a 72-year-old man, was referred to hematology from oncology just 2 months after the first patient. An internal medicine evaluation was performed due to the complaints of intermittent body swelling over the past 2 years. Sclerotic bone lesions were found in thorax CT, interpreted as metastasis. As a result, the patient was assessed for primary unknown metastatic cancer in the oncology department. Widespread sclerotic bone lesions were detected in the skeletal system by PET/CT, most of which had not shown FDG involvement but had been shown FDG involvement in silhouette form at the bone marrow level (Figure 3). Thus, while PET/CT, endoscopy, and tumor markers (such as PSA) fail to locate a primary tumor, the first examinations for MM were conducted due to widespread sclerotic bone lesions, as in the previously referred patient. The ratio of kappa/lambda was 1.1/170, and immunoglobulin levels were normal but on the lower limit. The bone marrow trephine biopsy revealed the presence of clustered monoclonal plasma cells at a rate of 40%. Plasma cells were positive for lambda and CD138 stains but negative for kappa (Figure 4). The other results of the patient’s laboratory tests are shown in Table 2. After eight treatment cycles with the weekly VCD protocol, we achieved a very good partial remission.
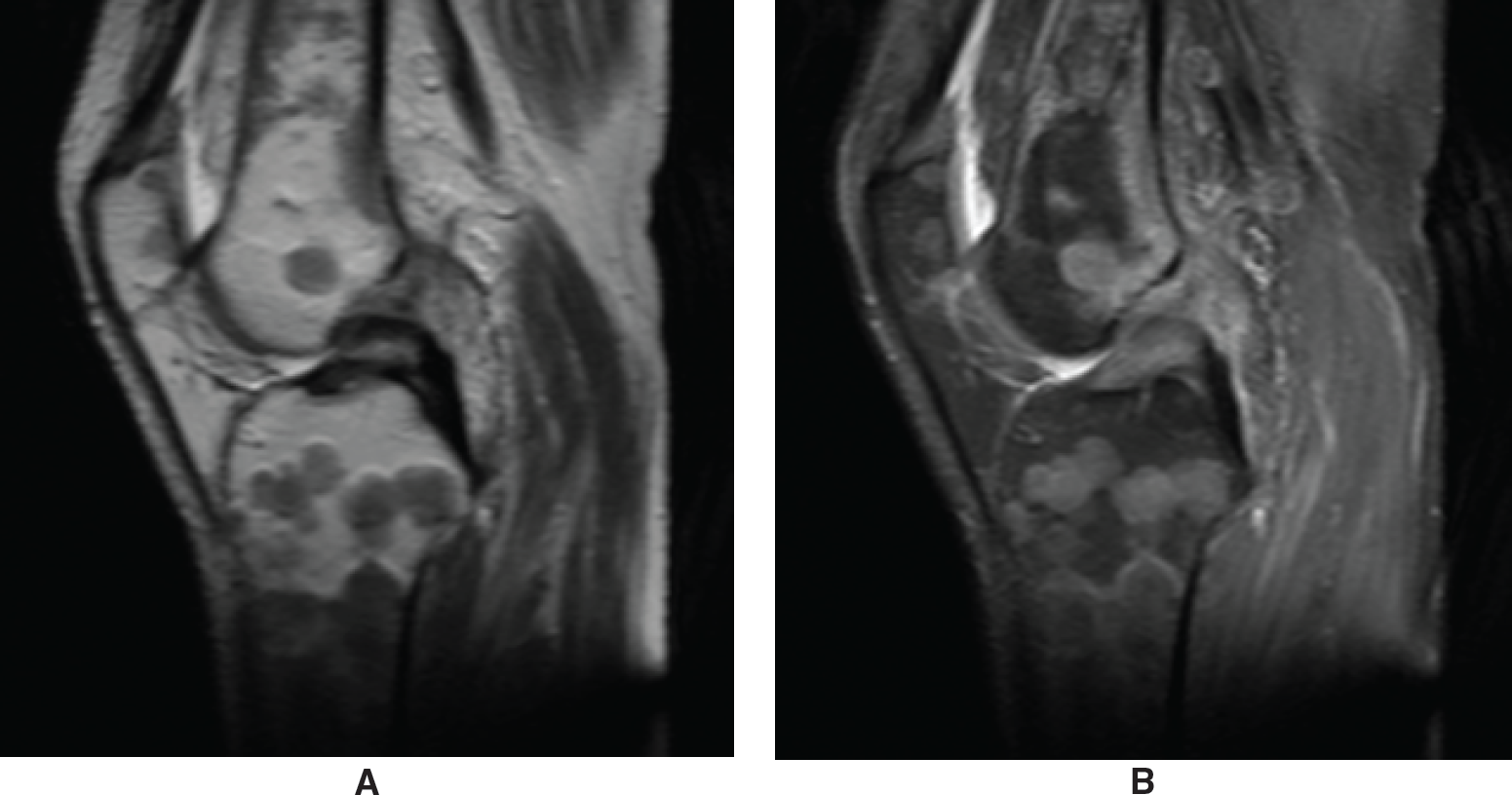
There are areas of bone infiltration in the knee joint that are round or oval in shape. (A) These lesions merge in some places and appear hypointense on T1-weighted images and (B) hyperintense on fat-suppressed T2-weighted images.
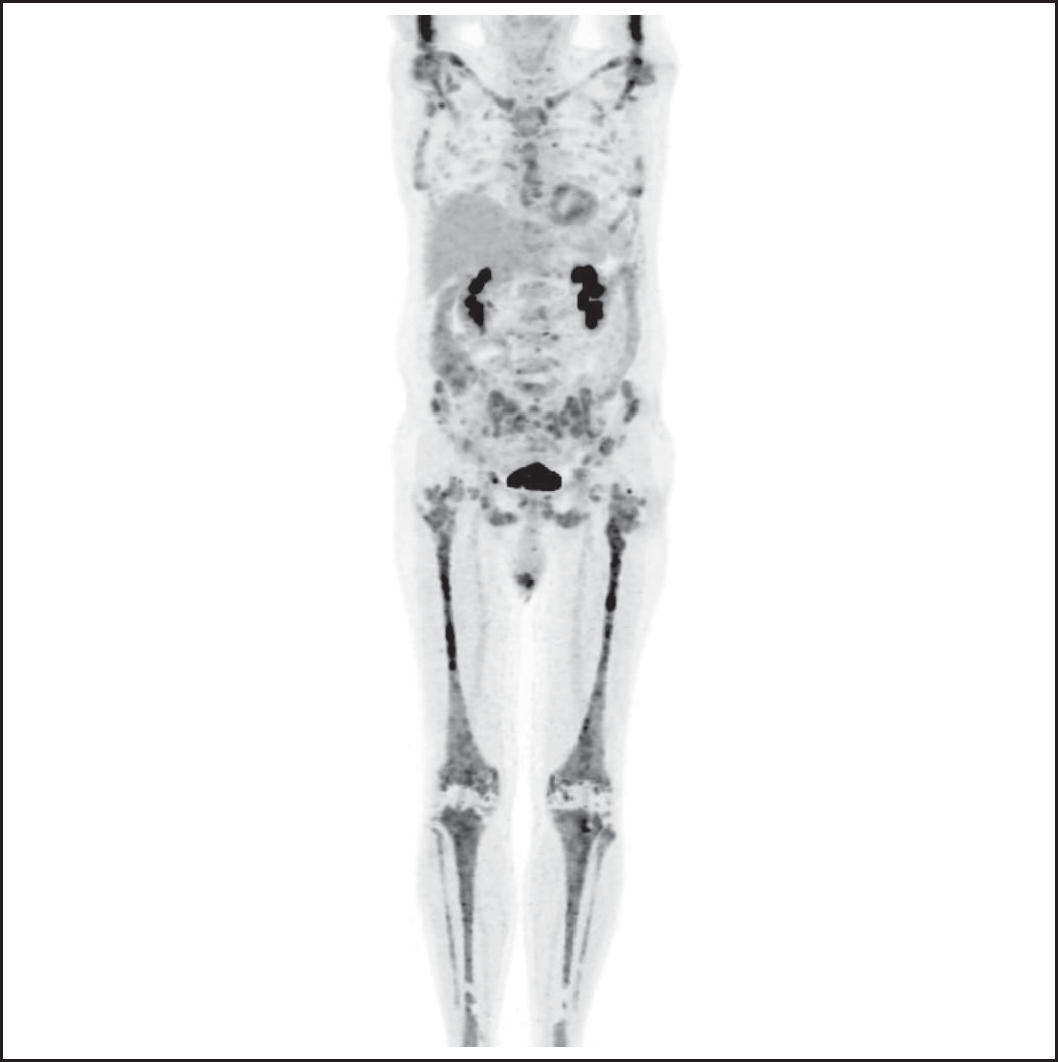
Widespread hypermetabolic lesions in the skeletal system at the bone/marrow level in PET/CT. MIPI images of the PET/CT
| LABORATORY PARAMETERS | RESULTS (NORMAL RANGE) |
|---|---|
| Hemoglobin | 11.21 × 106 g/dl (13.5-18) |
| Erythrocyte sedimentation rate | 61 mm/hour (0-20) |
| PSA | 1.72 µg/l (0-4) |
| Calcium | 8.6 mg/dl (8.8-10) |
| ALP | 86 U/l (40-150) |
| PTH | 31 pg/ml (15-68.3) |
| Albumin/Globulin | 33.40 g/l (32-46)/38.1 g/l |
| Beta-2 microglobulin | 3.22 mg/l (0.97-2.64) |
| Ig A, Ig G, Ig M, | 0.51 g/l (1.01-6.45) 21.17 g/l (5.40-18.22) 0.34 g/l (0.22-2.40) |
| Serum kappa free light chain | 86.6 mg/l (3.30-19.4) |
| Serum lambda free light chain | 31.8 mg/l (5.70-26,3) |
| Serum protein electrophoresis | M spike = 1.7 g/dl |
| Serum immunofixation | Ig G, Kappa monoclonal gammopathy |
PSA, prostate-specific antigen; ALP, alkaline phosphatase; PTH, parathormone; Ig, immunoglobulin.
Discussion
The diffuse osteosclerotic bone lesion in the MM is detected rarely, and this entity is different from the osteosclerotic myeloma (syndrome of polyneuropathy, organomegaly, endocrinopathy, monoclonal gamapathy, and scin lesions) syndrome. The incidence revealed in the literature is 3% [4]. Osteosclerosis in myelomas is classified into four different subgroups [5]. These are diffuse, focal osteocondensation, bone spiculation on the surface of the bone, and sclerotic reaction at the rim of a lytic lesion [5]. Typically, most of the cases are presented with discrete and purely lytic lesions. When we searched the literature, only less than 20 patients (including ours) reported myeloma-related diffuse osteosclerosis. The survival of myeloma patients with diffuse osteosclerosis reported varies ranging from 11 months to 10 years [6]. The patients were aged 40-74, and the clinical presentations were anemia, subcutaneous plasmacytoma, anorexia, pain, and weight loss. Our two patients are also 71 and 72 years old, and the symptoms were similar. They were interestingly referred to the hematology clinic from oncology related to the report of PET/CT. They had been investigated for solid organ tumors with widespread sclerotic bone metastasis. In this process, the interpretation of all findings as metastatic diseases from the first radiological evaluation of their symptoms had a large share. Unfortunately, this caused a waste of time in diagnosing these patients.
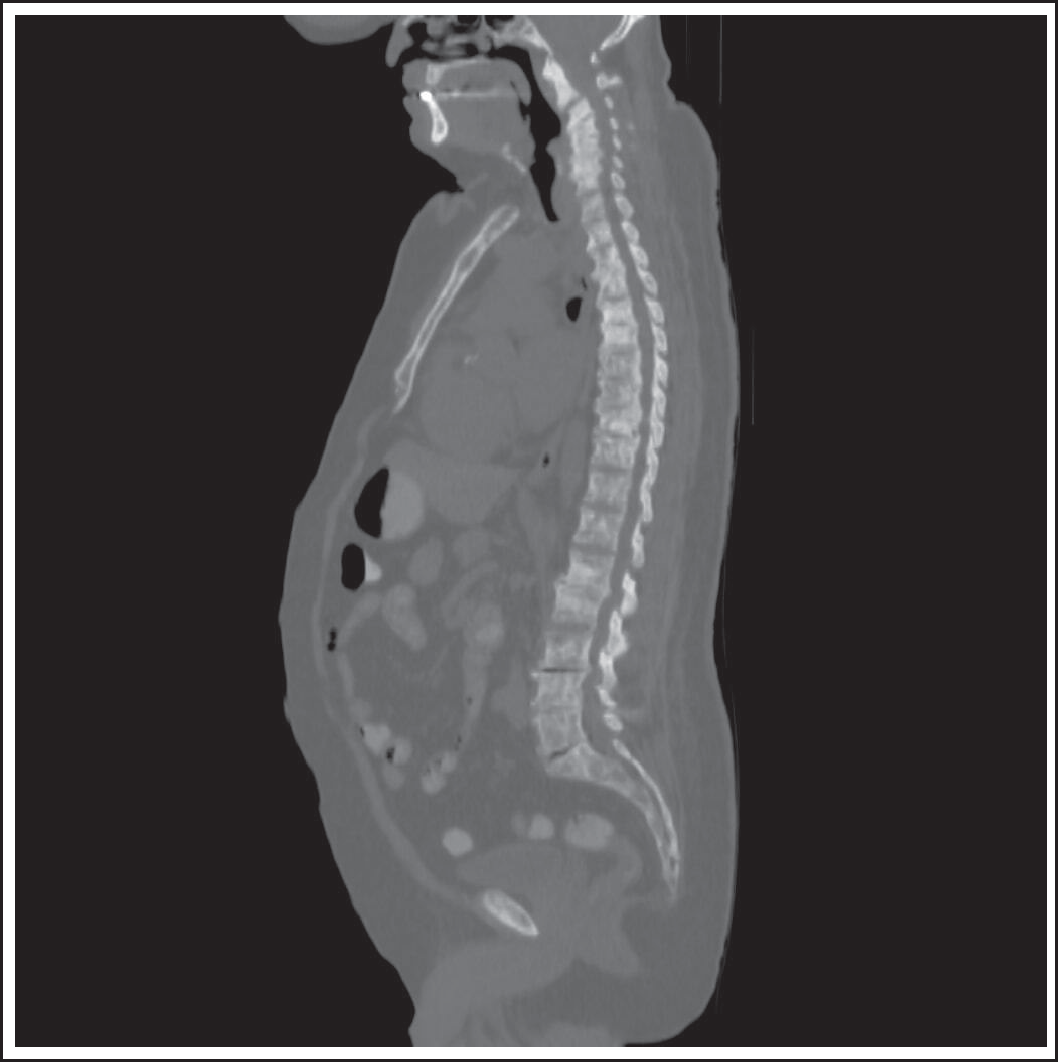
The widespread sclerotic bone lesions are shown in the low-density computerized tomography part of PET/CT.
The pathology of lytic bone lesions of myeloma is likely mediated through the stimulation of osteoclasts by several cytokines, including interleukin-1b and tumor necrosis factor [7]. The diffuse osteosclerosis pathogenesis in myeloma was not demonstrated clearly. Osteosclerotic bone metastasis in solid organ malignancies, commonly prostate cancer, is related to cytokines synthesized by cancer cells that are known to stimulate only osteoblasts [8]. These are platelet-derivated growth factor (PDGF) and transforming growth factor beta. Diffuse fibrosis and osteosclerosis in primary myelofibrosis are known to be associated with PDGF, too. Therefore, it can be assumed that similar mechanisms are likely responsible for osteosclerotic bone lesions in patients with MM.
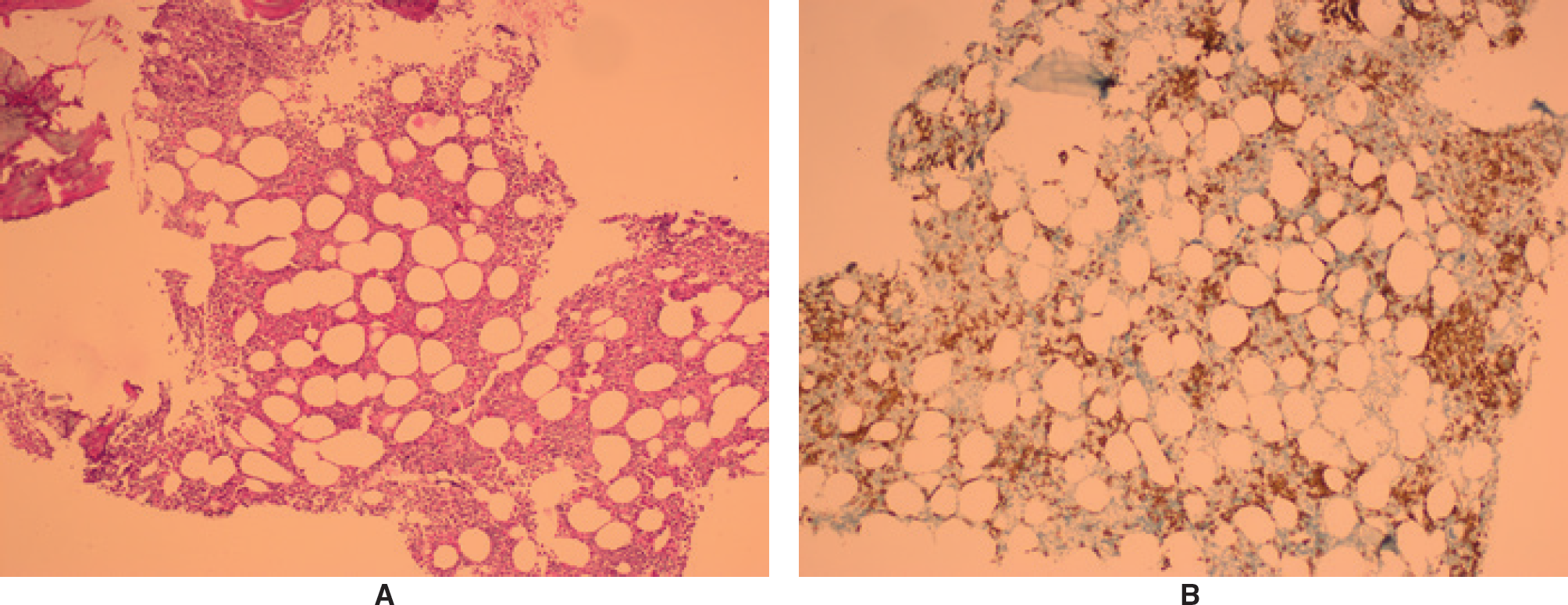
(A) Hematoxylin and eosin staining and (B) immunohistochemical staining for CD 138 showing a plasma cell infiltrate (original magnification, ×100).
| LABORATORY PARAMETERS | RESULTS (NORMAL RANGE) |
|---|---|
| Hemoglobin | 11.83 × 106 g/dl (13.5-18) |
| Erythrocyte sedimentation rate | 20 mm/hour (0-20) |
| PSA | µg/l (0-4) |
| Calcium | 8.4 mg/dl (8.8-10) |
| ALP | 51 U/l (40-150) |
| PTH | 178 pg/ml (15-68.3) |
| 25-HO vitamin D | 13.70 ng/ml (6.60-49.90) |
| Albumin/Globulin | 40.1 g/l (32-46)/20.70 g/l |
| Beta-2 microglobulin | 2.42 mg/l (0.97-2.64) |
| Ig A, Ig G, Ig M, | 1.39 g/l (1.01-6.45) 5.73 g/l (5.40-18.22) <0.25 g/l (0.22-2.40) |
| Serum kappa free light chain | 11.1 mg/l (3.30-19.4) |
| Serum lambda free light chain | 171.0 mg/l (5.70-26.3) |
| Serum protein electrophoresis | Hypogammaglobulinemia |
| Serum immunofixation | Lambda monoclonal gammopathy |
PSA, prostate-specific antigen; ALP, alkaline phosphatase; PTH, parathormone; Ig, immunoglobulin.

