Background
Chylopericardium is a rare entity characterized by the accumulation of chylous fluid containing high concentrations of triglycerides in the pericardial cavity. It is diagnosed by pericardiocentesis and can be idiopathic or associated with cardiothoracic surgery, trauma, radiation, malignancy, cystic hygromas, or lymphangiomas [1]. Chylopericardium in association with lymphangioleiomyomatosis (LAM) is exceedingly rare, with only a few cases reported in the literature [2,3]. Moreover, chylopericardium as a defining diagnostic characteristic of LAM has not been described before. We describe the first case of chylopericardium causing cardiac tamponade as a defining diagnostic characteristic of LAM that was successfully managed with sirolimus.
Case Presentation
A 61-year-old woman presented to the emergency department with a 15-year history of increasing dyspnea with associated weight loss. Her dyspnea had significantly worsened in the previous 1 week with no associated chest discomfort, cough, sputum production, wheeze, lower limb swelling, orthopnea, or paroxysmal nocturnal dyspnea.
She was a non-smoker, and her medical history was significant for possible LAM. She had previously sought evaluation at the respiratory specialist clinic of the same hospital 5 years before this admission for dyspnea, and a computed tomography (CT) scan of the thorax at the time showed numerous, diffusely distributed thin-walled lung cysts characteristic of LAM but no evidence of angiomyolipomas or lymphadenopathy (Figure 1A-C). Spirometry showed very severe obstruction (Figure 1D). There were no clinical features of tuberous sclerosis, no family history of lung disease, and no previous history of pneumothoraces or pleural effusions. The patient declined further workup with surgical lung biopsy or vascular endothelial growth factor (VEGF) testing. Hence, she did not fulfill the diagnostic criteria for definite LAM. A treatment trial with sirolimus was also discussed, given the suspicion of LAM, but the patient declined. She was subsequently lost to follow-up.
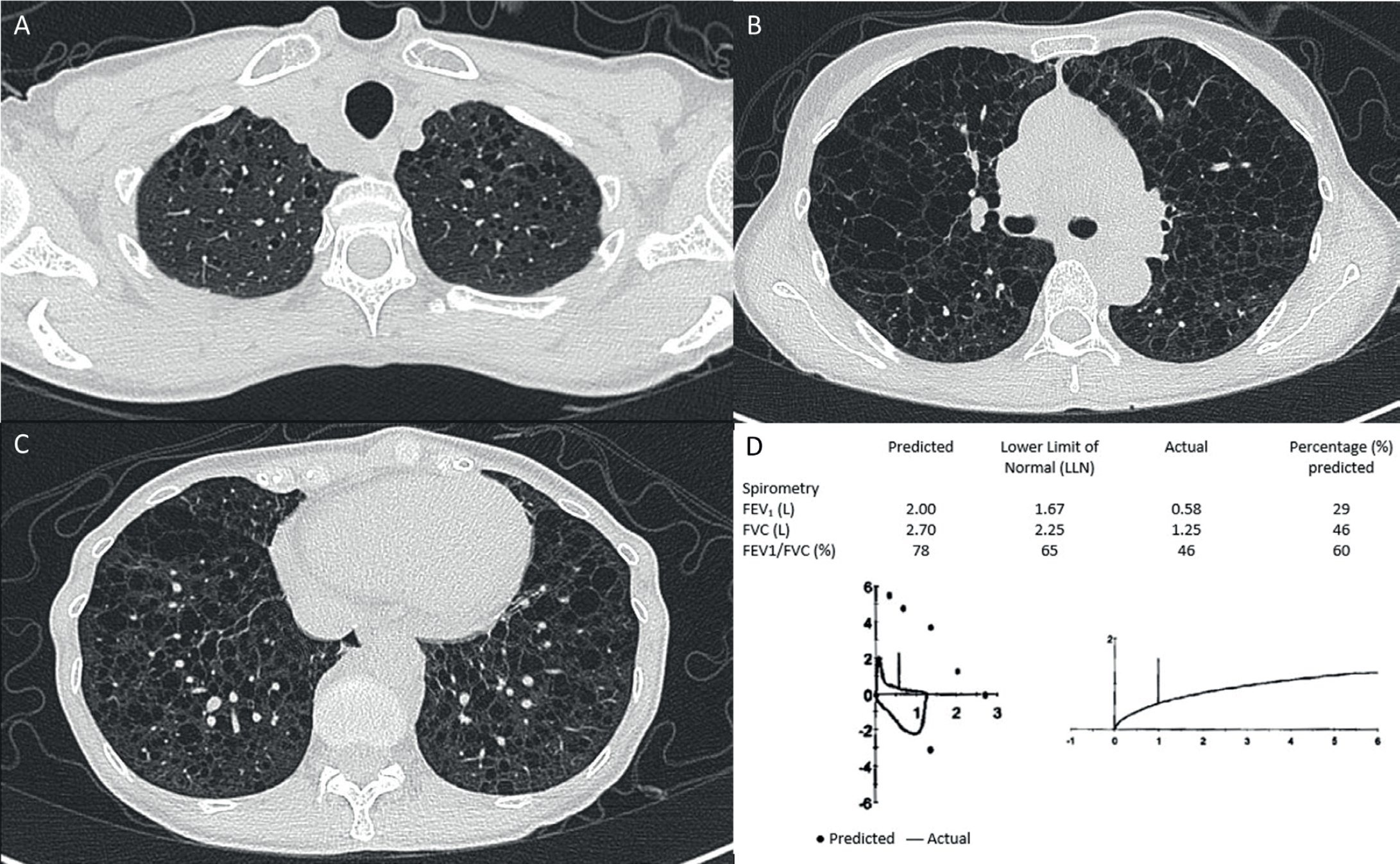
(A-C) CT-thorax 5 years before this admission showing bilateral diffusely distributed lung cysts. (D) Spirometry 5 years before this admission showed very severe obstruction (illustrated by Isaac Jun Jie Fong).
On physical examination, she appeared frail with a body mass index of 12.6, a heart rate of 106 beats/minute with pulsus paradoxus, a respiratory rate of 32 breaths/minute, and oxygen saturation of 77% on ambient air. Her blood pressure was 135/84 mmHg. Her jugular venous pressure was not elevated, heart sounds were normal and not muffled, and bilateral wheeze was present on auscultation of the lungs.
At admission, the patient’s blood levels of hemoglobin and troponin T were normal, but N-terminal pro-B type natriuretic peptide (NT-proBNP) was elevated at 3,206 pg/ml. Arterial blood gas analysis with the administration of 15 l/minute supplemental oxygen via a non-rebreather mask detected a pH of 7.47, pO2 of 383 mmHg, pCO2 of 42 mmHg, and SaO2 of 100%. Other laboratory test results are shown in Table 1.
A 12-lead electrocardiogram (ECG) showed sinus tachycardia at 106 beats/minute with no evidence of electrical alternans, low voltage QRS complexes, or P-R interval depression (Figure 2A). In addition, a portable anteroposterior chest radiograph performed was indeterminate for the presence of a small right apical pneumothorax or an apical bulla (Figure 2B).
Point of care ultrasound examination in the emergency department revealed a large circumferential pericardial effusion. This was confirmed with a trans-thoracic echocardiogram, which showed a large pericardial effusion seen mainly adjacent to the right ventricle and around the apex with intermittent right ventricular diastolic collapse and significant respiratory variation in the mitral and tricuspid inflows (Figure 3A-E), echocardiographic features of early cardiac tamponade.
Urgent pericardiocentesis was performed by cardiology in the cardiac catheterization lab, and 200 ml of thick milky fluid was aspirated (Figure 4). Analysis of the pericardial fluid showed total cholesterol of 1.89 mmol/l and triglycerides of 3.93 mmol/l, confirming the presence of a chylopericardium. No malignant cells were detected on cytologic examination, and microbiological studies were negative, including gram stain, bacterial culture, acid-fast bacilli stain, and acid-fast bacilli culture. A CT scan of the thorax, abdomen, and pelvis during this admission demonstrated that the cystic changes in the lungs had progressed, and no pneumothorax was present. Additionally, there were retroperitoneal soft tissue densities suggestive of lymphangioleiomyomata.
The patient was diagnosed with LAM based on the findings of multiple characteristic lung cysts on CT, a chylopericardium, and retroperitoneal lymphangioleiomyomata. Sirolimus administered orally was initiated with a 3 mg loading dose followed by a maintenance dose of 1 mg once daily. Her dyspnea and oxygen saturations improved, but she remained persistently hypoxic, requiring 1 l of supplemental oxygen via nasal cannula. Electrolyte disturbances were corrected intravenously with fluids and electrolytes, in addition to dietary supplementation.
| LABORATORY TEST (UNITS) | REFERENCE RANGE, ADULTS | AT ADMISSION |
|---|---|---|
| Hemoglobin (g/dl) | 12.0-16.0 | 12.1 |
| Hematocrit (%) | 36.0-46.0 | 33.2 |
| Total white blood cell count (109/l) | 4.0-10.0 | 13.3 |
| Differential count | ||
| Neutrophils (Absolute) (109/l) | 2.0-7.5 | 12.33 |
| Lymphocytes (Absolute) (109/l) | 1.0-3.0 | 0.19 |
| Monocytes (Absolute) (109/l) | 0.2-0.8 | 0.65 |
| Eosinophils (Absolute) (109/l) | 0.04-0.44 | 0.03 |
| Platelet count (109/l) | 144-440 | 287 |
| Sodium (mmol/l) | 136-146 | 132 |
| Potassium (mmol/l) | 3.6-5.0 | 3.2 |
| Chloride (mmol/l) | 100-107 | 87 |
| Bicarbonate (mmol/l) | 19.0-29.0 | 29.9 |
| Urea nitrogen (mmol/l) | 2.7-6.9 | 2.1 |
| Creatinine (umol/l) | 37-75 | 25 |
| NT-proBNP (pg/ml) | <150 | 3206 |
| Troponin T (ng/l) | <30 | 28 |
| Albumin (g/l) | 40-51 | 37 |
| Total bilirubin (umol/l) | 7-32 | 24 |
| Aspartate transaminase (U/l) | 12-42 | 40 |
| Alanine transaminase (U/l) | 6-66 | 40 |
| Alkaline phosphatase (U/l) | 39-99 | 64 |
| Gamma-glutamyl transferase (U/l) | 9-53 | 16 |
| Procalcitonin (ug/l) | <0.50 | 0.31 |
| Arterial blood gas | ||
| pH | 7.35-7.45 | 7.47 |
| pO2 (mmHg) | 75.0-100.0 | 383 |
| pCO2 (mmHg) | 35.0-45.0 | 42 |
| Bicarbonate (mmol/l) | 21.0-27.0 | 28.8 |
| SaO2 (%) | 95.0-100.0 | 100 |
Further management was discussed at a multi-disciplinary meeting involving cardiologists, cardiothoracic surgeons, and respiratory physicians to determine the best management approach given the risk of re-accumulation of the pericardial effusion. Given the patient’s frail clinical status, it was determined that she was at high surgical risk for pericardial window creation, and conservative measures such as initiation of a low-fat, high medium-chain triglyceride diet and for repeat pericardiocentesis in the event of recurrent cardiac tamponade were formulated as the primary components of a management strategy.
One week after the initial pericardiocentesis, an interval echocardiogram showed re-accumulation of the pericardial effusion with right ventricular diastolic collapse. However, the patient was asymptomatic and declined a second pericardiocentesis. The hospital palliative care service was consulted, and the goals of care were discussed with the patient, who opted for a comfort-oriented approach. The patient continued conservative management with sirolimus, and dyspnea was managed with physical therapy and opioids. She was discharged to an inpatient hospice because of increased care requirements and limited social avenues for return to independent living.
At the time of writing, 1 year has lapsed since her initial presentation with cardiac tamponade, and the patient has made a remarkable recovery. There was no clinical evidence of cardiac tamponade, and she no longer required supplemental oxygen. Her mobility improved, and she was discharged from the inpatient hospice to a nursing home.
Discussion
This case describes chylopericardium causing cardiac tamponade as a defining diagnostic characteristic of LAM. While the diagnosis of LAM was initially suspected based on characteristic CT findings, an obstructive defect on spirometry, and compatible symptoms of progressive exertional dyspnea, there were no additional defining diagnostic characteristics to confirm the diagnosis of LAM, such as clinical features of tuberous sclerosis or the presence of angiomyolipomas or lymphadenopathy. Furthermore, the patient had declined upfront surgical lung biopsy, VEGF testing, and treatment with sirolimus when she first presented. In her sixth decade of life, her presentation with chylopericardium remains the defining diagnostic characteristic of LAM.
Chylopericardium is most commonly reported following open-heart surgery and may be due to transection of the tiny lymphatic channels in the pericardial reflections or by operative injury to tributaries of the thoracic duct. The standard approach to treating chylopericardium is with a chyle diet, pericardiocentesis, and surgical intervention if conservative therapy fails [4]. Surgical intervention typically entails ligation of the thoracic duct with the creation of a pericardial window. When the diagnosis of chylopericardium has been established and is unrelated to cardiac surgery, a search for an underlying cause should be done. Investigations should be conducted specifically for malignant disease, tuberculosis, previous trauma, and radiation [5].
LAM is a rare, progressive systemic disease with prominent features of characteristic lung cysts, abdominal tumors, and the development of chylous effusions. It is considered a low-grade neoplastic condition caused by the proliferation and infiltration of LAM cells. LAM may be associated with tuberous sclerosis complex (TSC), a multisystem genetic disease caused by germline mutations in either TSC1 or TSC2 or may also arise sporadically [6]. Sporadic LAM affects mainly women, and the prevalence has been estimated to be approximately 3.4-7.8 per million women [7]. Current guidelines have established the diagnostic criteria for definite LAM as the presence of multiple characteristic cysts on lung CT and any of the following: kidney angiomyolipomas, thoracic or abdominal chylous effusion, lymphangioleiomyomas or lymph node involvement, and TSC or the presence of elevated VEGF-D (>800 pg/ml) [8]. Chylothoraces are the presenting manifestation in about 20% of patients, and chylous ascites in around 5% [9].
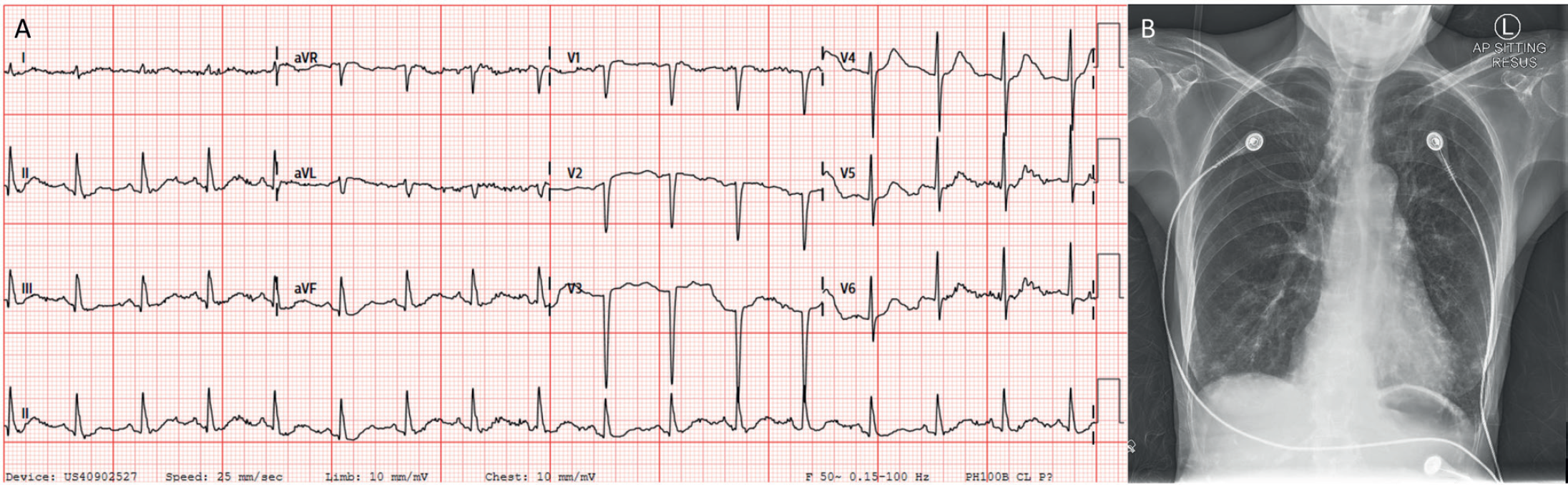
(A) ECG at admission showing sinus tachycardia. Other ECG features of cardiac tamponade (not seen in this ECG) include low voltage, P-R interval depression, and electrical alternans. (B) Portable chest radiograph demonstrating paucity of lung markings in the right upper zone, indeterminate for pneumothorax or a lung bulla.
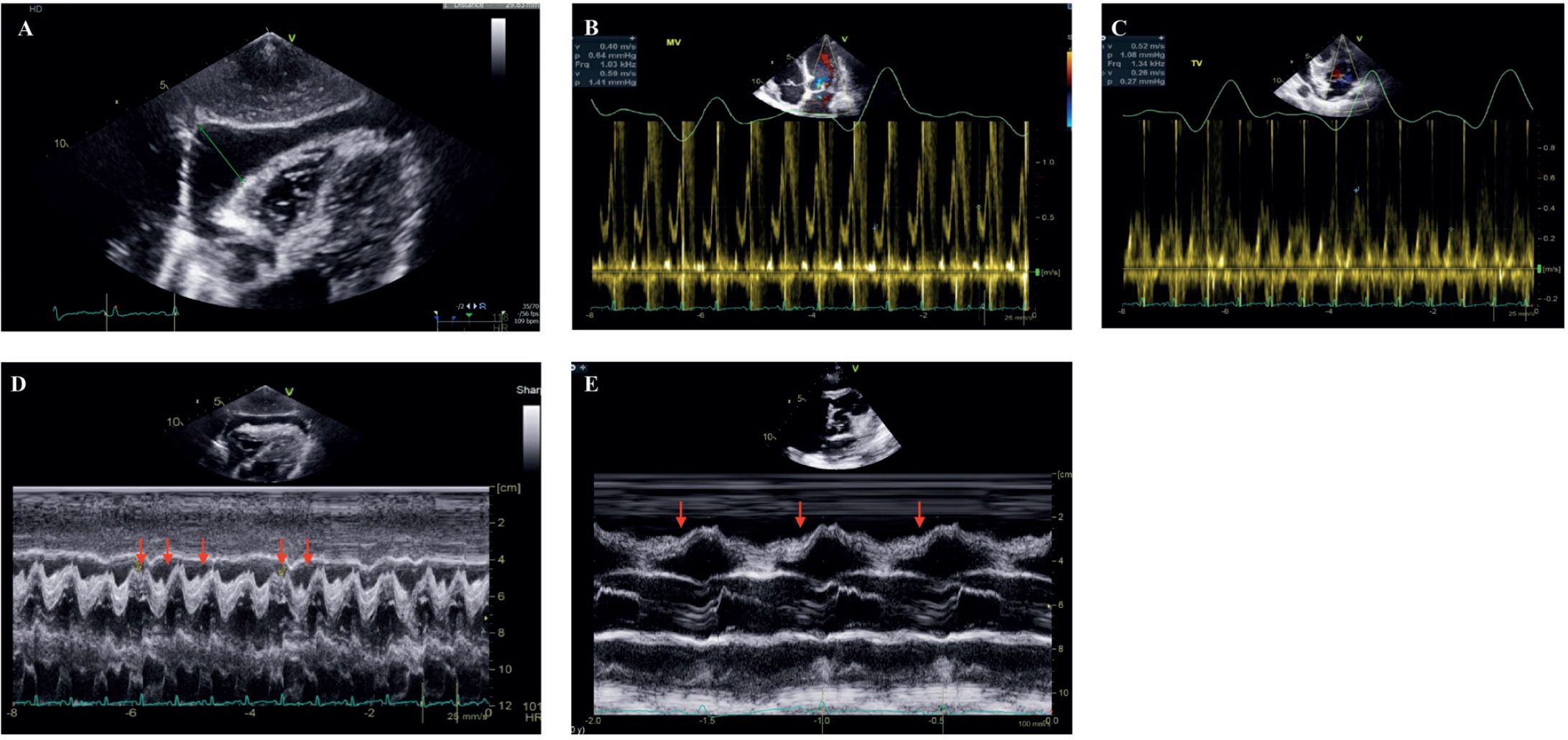
(A) Subcostal view with a large pericardial effusion measuring 3.0 cm adjacent to the right ventricle. (B) Pulsed wave Doppler tracing of the mitral inflow showing a change of more than 25% inflow velocity with a respiratory variation. (C) Pulsed wave Doppler tracing of the tricuspid inflow showing a change of more than 40% inflow velocity with a respiratory variation. A change of inflow velocity by more than 40% for the tricuspid valve or 25% for the mitral valve suggests ventricular interdependence, a feature detected in cardiac tamponade. (D) M-mode of the subcostal view demonstrating right ventricular diastolic collapse (illustrated by red arrows). (E) M-mode of the parasternal long-axis view of another patient with more pronounced right ventricular diastolic collapse (represented by red arrows).
Chylopericardium as a presenting manifestation in LAM has scantily been reported in the literature. In 2004, Hovland and Bjornstad [2] reported a female in her early 60’s with definite LAM as evidenced by renal angiomyolipoma and bilateral pneumothoraces requiring pleurodesis who subsequently presented with chylopericardium. Similarly, Paul et al. [3] described a patient with retroperitoneal lymphangioma and prior history of recurrent pneumothoraces requiring pleurodesis who later presented with chylopericardium with tamponade. To our knowledge, we report the first case of LAM in which chylopericardium was the first defining feature.
Sirolimus is a mechanistic target of rapamycin inhibitor, and it is the first-line treatment for LAM [10]. It has been shown to reduce angiomyolipoma size [11], lower the rate of FEV1 decline, and improve the quality of life in LAM patients compared with placebo [12]. Sirolimus has also been shown to have efficacy in reducing the accumulation of chylous pleural or peritoneal collections [13]. However, data on the efficacy of sirolimus in reducing the re-accumulation of chylopericardium is lacking. In this report, we have described the successful use of low-dose sirolimus [14] in managing a patient with LAM-associated chylopericardium that was able to obviate the need for repeated pericardiocentesis despite the initial presentation with tamponade physiology. The long-term follow-up of our patient is significantly impacted by her lack of financial and social support, including limited avenues for discharge back to independent living, her significant symptom burden, and her values and self-determined goals of care. Our case is hence significantly limited by the lack of follow-up echocardiographic studies to determine the time course of the chylopericardium. However, it may be inferred from clinical stability and survival of 1 year from initial pericardiocentesis that there were no further episodes of cardiac tamponade.
Other options for treatment include a therapeutic trial of bronchodilators for symptomatic patients, especially those with an obstructive defect on spirometry, and lung transplant in patients with end-stage respiratory failure, as defined by one of the following: FEV1 percentage predicted of 30% or less, resting hypoxemia, progressive lung function decline, and New York Heart Association functional class 4. Early-stage investigations of statins, hydroxychloroquine, and celecoxib have been published, but larger studies are needed to determine their clinical effects [10].
Conclusion
Patients with LAM can develop chylopericardium apart from chylous pleural effusion and ascites. Treatment with sirolimus appears to be effective in reducing the accumulation of chylopericardium in LAM.
What is new?
Chylopericardium in association with lymphangioleiomyomatosis (LAM) is exceedingly rare, with only two cases reported in the literature to date. Moreover, chylopericardium as a defining diagnostic characteristic of LAM has not been described before. The authors’ manuscript describes, to their knowledge, the first case of chylopericardium causing cardiac tamponade as a defining diagnostic characteristic of LAM that was successfully managed with sirolimus.


