Background
Hepatic portal venous gas (HPVG), defined as the presence of gas in the portal venous system, had long been considered an alarming finding which should be urgently addressed surgically. However, very few cases have described the benign etiologies of HPVG such as gastric emphysema, which can be managed conservatively and thus can have an important impact on the mortality rate and the prognosis of the patient’s condition. HPVG has been also rarely presented in the literature in the presence of gastric dilatation, where, in addition to the conservative management, close monitoring is required to assess for any signs of gastric necrosis.
The literature regarding this topic has been scarce and few such cases have been described.
Here, we report a case of acute gastric dilatation with HPVG in the presence of gastric emphysema and closed loop small bowel obstruction, complicated by gastric necrosis, in a patient with adjustable gastric band (AGB).
Case Report
A 59-year-old lady, previously healthy, presented to the emergency department complaining of severe, diffuse, colicky, non-radiating abdominal pain of 1 day duration. It was associated with nausea and multiple episodes of watery, non-bloody diarrhea. The patient denied having fever, chills, vomiting, or other symptoms.
Past surgical history included C-section 33 years ago, open cholecystectomy 25 years ago and laparoscopic AGB (LAGB) placement 16 years ago. The latter was complicated by acute band slippage for which midline revision laparotomy was done.
Upon presentation, the patient was in pain, but her vital signs were within normal range. Physical examination revealed a distended and rigid abdomen, diffusely tender to palpation with hyperactive bowel sounds.
Laboratory studies showed leukocytosis (16.62 × 109 cells/l) with 83% neutrophils, elevated lipase (499 U/l), and elevated lactate dehydrogenase (800 U/l) levels. Arterial blood gas test revealed metabolic acidosis (pH: 7.27, HCO3 -: 19 mmol/l) and lactic acid levels of 2.59 mmol/l.
Abdominopelvic computed tomography (CT) scan demonstrated over-distension of the stomach and HPVG (Figure 1a). Adequate positioning of the gastric band with a phi angle of 39° was shown (Figure 1b). Diffuse dilatation of the small bowel loops was also seen, with an abrupt change of caliber at the level of a collapsed distal ileal loop, showing the transitional zone (Figure 1c). There was no evidence of bowel wall thickening, pneumatosis intestinalis or free air. The patient was diagnosed with closed loop small bowel obstruction with secondary acute gastric dilatation and HPVG in the presence of gastric emphysema.
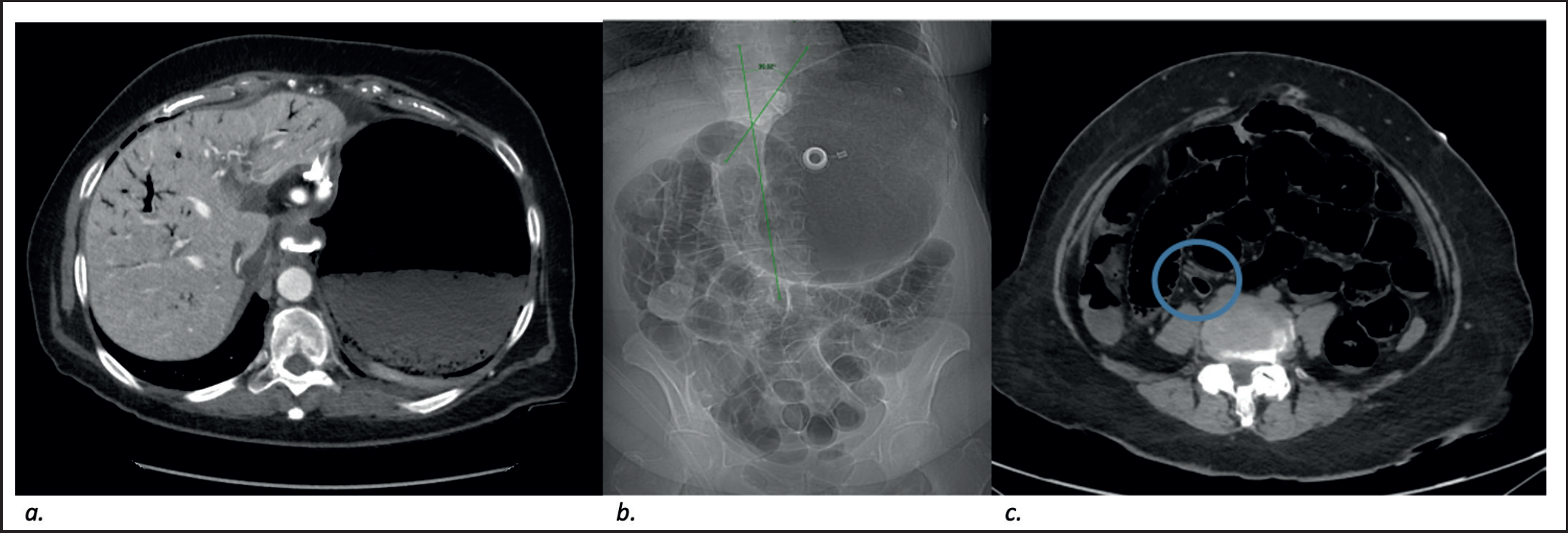
Contrast enhanced CT scan performed upon admission (a) axial image demonstrating over-distension of the stomach, interval appearance of gastric wall pneumatosis and portal venous gas. (b) Scout images demonstrating adequate positioning of the gastric band with a phi angle of 39° (normal range 4°-52°). (c) Axial cuts at lower level showing diffuse dilatation of the small bowel loops, with an abrupt change of caliber at the level of a collapsed distal ileal loop, indicating a possible transitional zone (blue circle).
Subsequently, the AGB was deflated, a nasogastric tube was inserted, and the patient was started on antibiotic therapy. She was admitted to the intensive care unit (ICU) for close surveillance with frequent checkups and close monitoring for any signs of necrosis.
Four hours after band deflation, CT scan of the abdomen and labs were repeated for follow up. Imaging demonstrated interval regression of the gastric distention with gastric wall pneumatosis. There was interval marked decrease in the portal venous gas, with residual air seen at the anterior liver segments (Figure 2a). The gastric band was then seen at a phi angle of 33° and interval minimal decrease in the small bowel dilatation was noted (Figure 2b). Lab tests also markedly improved.
Over the next 3 days, the patient’s physical exam became less remarkable; the abdomen became soft, flatus, and stools were positive. However, the nasogastric tube was still draining a large output of fecaloid material which then became bilious starting day 2 post ICU admission. Nevertheless, C-reactive protein, lactate, and white blood cell count were decreasing gradually. The patient was transferred to the floor on her fourth day of admission and scheduled for exploratory laparoscopy with AGB removal for the next day.
The nasogastric tube gave black tarry material on the scheduled surgery day, so gastroscopy (Figure 3a) was performed preoperatively to evaluate the gastric mucosa. It revealed a gangrenous stomach wall at the greater curvature from the fundus to the body of the stomach with a healthy antrum and duodenum. The lesser curvature showed a viable mucosa. An urgent exploratory laparotomy was mandated and a total gastrectomy with Roux-en-Y-esophagojejunostomy was performed. Perioperative findings confirmed the gastroscopy discoveries (Figure 3b).
The patient had a smooth and uncomplicated postoperative course. At day 5, a postoperative upper gastrointestinal series showed a patent anastomosis with no signs of leak or stasis (Figure 3c). Enteral feeding was initiated, and she was subsequently discharged home on post-op day 13.
Discussion
HPVG is defined as the presence of gas in the portal venous system. It was thought to be a highly feared sign indicating a life-threatening condition and associated with 75% mortality rate. However, recent studies have shown that, with the increased use of CT scans, which helped in diagnosing benign causes of HPVG, mortality rates decreased to 35% [1]. Thus, mortality related to HPVG highly depends on the etiology. In a case series conducted in Japan, Ikegame et al. [2] reported that 61% of patients with HPVG were treated successfully non-operatively. This suggests that HPVG is not always an ominous sign and its management is also cause dependent. The most common cause being pneumatosis intestinalis. In most cases, this can result in bowel necrosis and bacterial translocation from the bowels into the portal vein.
However, as previously stated, HPVG can also have multiple benign etiologies which do not require surgical intervention, such as emphysematous pyelonephritis, chronic obstructive pulmonary disease, or gastric emphysema [3]. The latter is defined as the dissection of air through the gastric mucosa into the gastric wall. It may be associated with HPVG because the left gastroepiploic veins are part of the portal venous system [4]. It is of high importance to differentiate gastric emphysema from emphysematous gastritis which is a rare form of gastritis with invasion of gas producing organisms into the gastric wall. Moreover, both entities can have similar radiological findings, but different clinical presentations and prognosis [5].
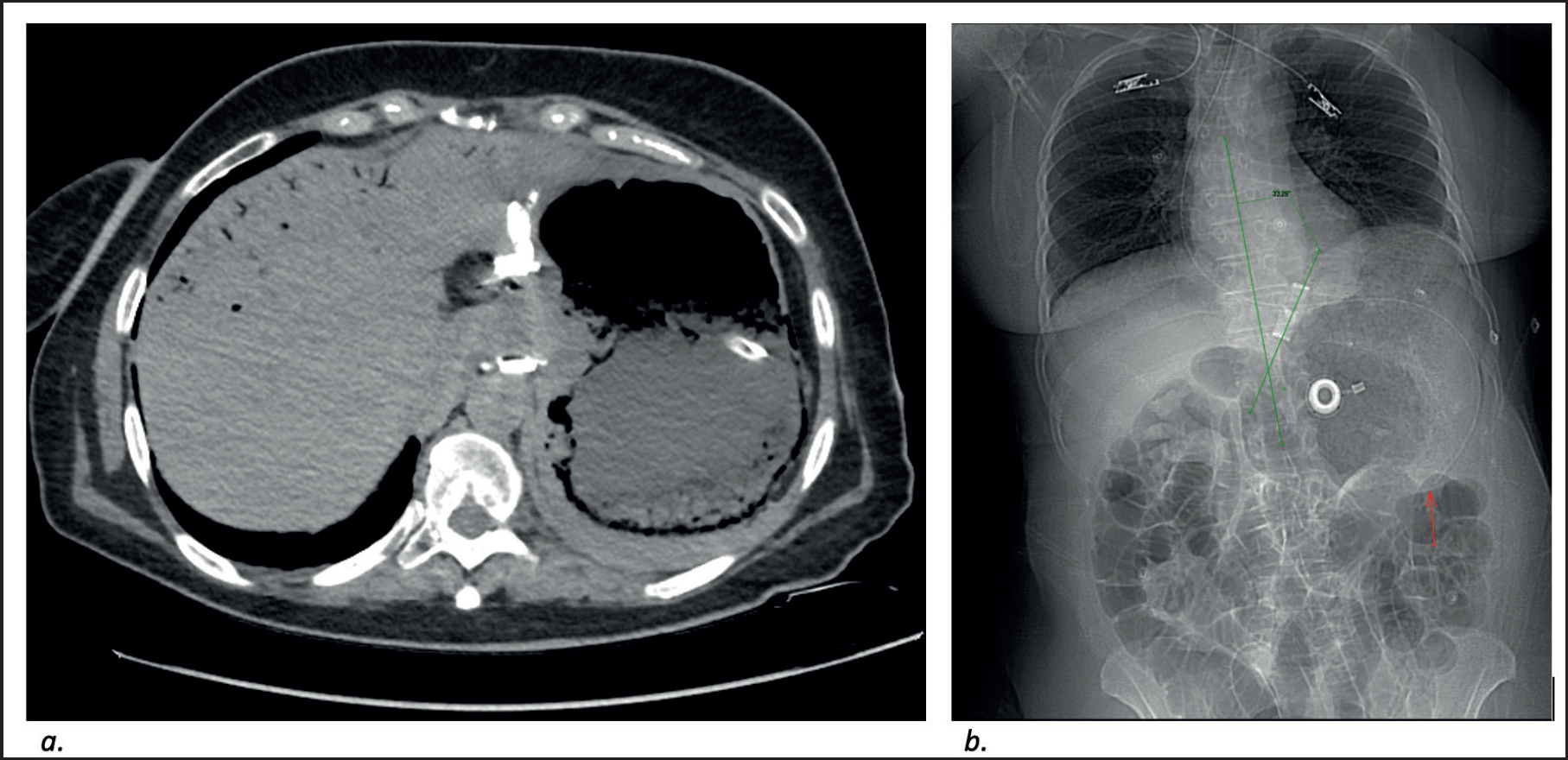
Non-enhanced CT scan of the abdomen and pelvis performed 4 hours after balloon deflation. (a) Regression of the gastric distention with marked decrease in the portal venous gas with persistent gastric wall pneumatosis. (b) Scout imaged showing phi angle of 33°. Tip of the nasogastric tube seen within the lower body of the stomach (red arrow)
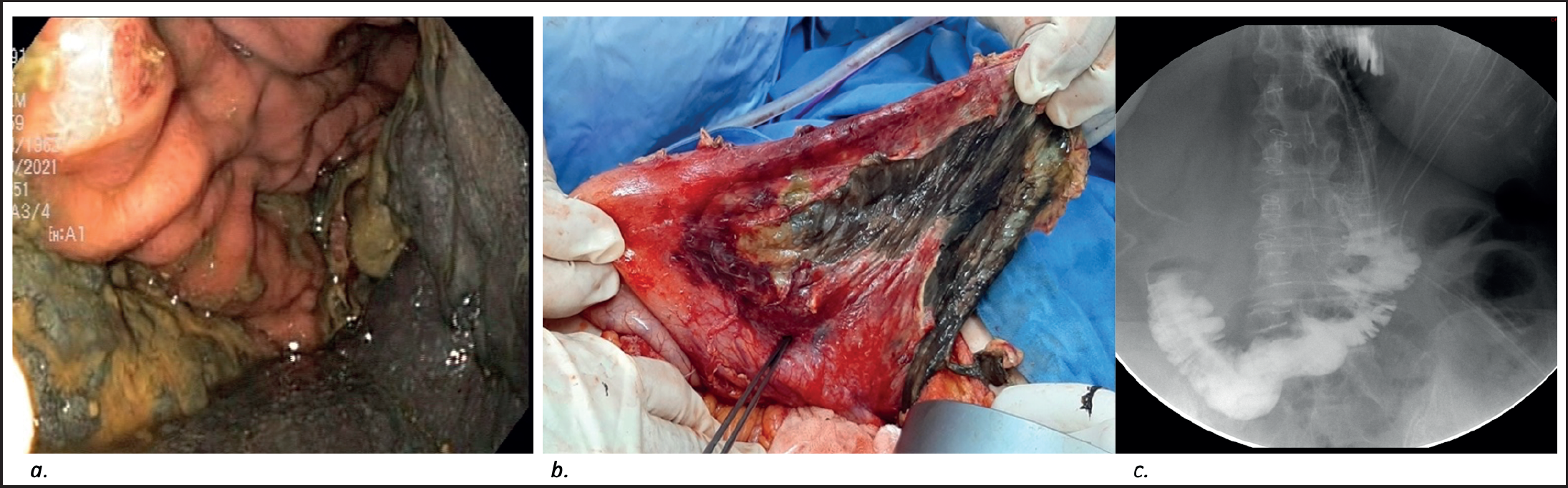
(a) Gastroscopy done preoperatively showing gangrenous stomach wall over the greater curvature with normal lesser curvature mucosa. (b) Stomach view intraoperatively revealing area of necrosis. (c) Dynamic analysis performed using a thin hydrosoluble bolus, showing adequate progression of contrast material through the esophagojejunostomy and jejunojejunostomy distally, with no evidence of leak or stasis.
On the one hand, a patient with emphysematous gastritis usually presents with severe abdominal pain, fever, hematemesis, and leukocytosis. On physical examination, severe abdominal tenderness or peritoneal signs can be noticed. It is a life-threatening condition, usually a surgical emergency with a mortality rate of 60%-80%. There are new reports of conservative management of emphysematous gastritis however the threshold for intervention is still relatively low [6]. On the other hand, a patient with gastric emphysema can be asymptomatic and afebrile. It is usually not an indication for surgery on its own, especially in patients who remain hemodynamically stable, it can be managed conservatively as a benign condition [4].
Causes of this condition were described since 1984 by Agha [7] and can be divided into: obstructive type, observed in patients with gastric distension or dilatation, post cardiopulmonary resuscitation, and patients on mechanical ventilation; traumatic type (post nasogastric tube placement or post blunt trauma) and pulmonary type (air from a ruptured bullae that spreads through vascular sheaths into the paraesophagus and downward to the gastric wall). Furthermore, gastric emphysema can be self-resolving, and its management depends on the etiology. Several case reports in the literature present patients with gastric emphysema who were successfully treated non-surgically [3,8,9]. In a literature review, Matsushima et al. [5] included all reported cases of emphysematous gastritis and gastric emphysema: about half of the cases reported between 1981 and 1997 and almost all cases reported between 2001 and 2012 were treated non-operatively; only one patient in 1995 underwent total gastrectomy secondary to acute gastric distention whereas the other cases that were treated surgically were found to have other etiologies (small bowel obstruction, parastomal hernia, etc.). Furthermore, surgical removal of the stomach was done in 23.1% of emphysematous gastritis cases and in only one patient with gastric emphysema [5].
HPVG has been reported along with gastric dilatation in a rare case by Radin et al. [10]. The affected patient was treated non-operatively and there was resolution of the HPVG 6 hours after nasogastric tube decompression [9]. In fact, the pathophysiology of acute gastric dilatation has not yet been thoroughly elucidated in the literature, but it is often seen with small bowel obstruction, eating disorders, superior mesenteric artery syndrome, postoperative gastric distension, anesthesia, and chronic debilitation [11]. The pathophysiology has not yet been thoroughly elucidated in the literature, but it is often seen with small bowel obstruction, eating disorders, superior mesenteric artery syndrome, postoperative gastric distension, anesthesia, and chronic debilitation [11]. Its management is usually non-operative with the insertion of a nasogastric tube for decompression and close monitoring for signs of gastric necrosis [12].
Lastly, LAGB has been shown to result in several complications since its emergence, some of them being more severe than others and requiring prompt assessment and management either conservatively or surgically, depending on the etiology. It has been depicted that 50% of patients who have undergone LAGB require reoperation, of which 25% present with major late complications [13]. These include but are not limited to port infection, leak, severe food intolerance/severe reflux symptoms, port or tubing malfunction, insufficient weight loss, band erosion, and pouch dilatation/slippage. The latter two being the most common complications seen [14]. In a retrospective study by Kerpel et al. [15], 43% of patients were found to have complications post LAGB observed on CT scan, of which small bowel obstruction, the complication seen in our patient on presentation, comprised a minority of 3.1% of cases.
Finally, our patient presented with an acute gastric dilatation with small bowel obstruction which was unlikely to resolve due to the presence of the gastric band. Hence the closed loop small bowel obstruction presentation on admission with no vomiting episodes. This led to the obstructive-type gastric emphysema that was seen on the abdominopelvic CT scans, which in turn led to the HPVG. Upon band deflation and nasogastric tube placement, the patient improved clinically but the high output in the nasogastric tube despite clinical improvement, the persistent gastric dilatation and the gastroscopy findings were noteworthy, especially that the patient’s clinical picture was not coherent with the findings of the stomach necrosis. This led to the decision of performing the open total gastrectomy with Roux-en-Y esophagojejunostomy.
Conclusion
To conclude, HPVG can have benign etiologies which influence its mortality rate, management, and prognosis. One of these etiologies is gastric emphysema, which can be treated conservatively. So, HPVG is not always an ominous sign as thought previously. Another entity that has been rarely described with HPVG is acute gastric dilatation, in which the main treatment goal is not intervene surgically but to perform decompression and close monitoring for any signs of necrosis. It was noticeable for us that there has been scarce data depicting this topic. Thus, it was worthy shedding the light on this case especially that our patient had no obvious clinical signs of gastric necrosis.

