Background
Ovarian germ cell tumors (OGCTs) are the second most common ovarian neoplasms, after surface epithelial tumors, and represent 70% of ovarian neoplasms in young, adolescent females [1]. Ovarian tumors may cause increase in thrombogenesis because of tumor-derived interleukin-6, tissue-factor expression, thrombocytosis, direct compression of pelvic veins, and several other postulated mechanisms [2]. Systemic thrombosis, often reported with surface epithelial tumors of ovary is uncommonly reported in OGCTs [3]. Similarly, chylothorax, commonly associated with lymphoproliferative malignancies, is rare in ovarian malignancies [3,4].
We report a case of OGCT that developed systemic thrombosis, pulmonary infarct, and chylothorax simultaneously and was successfully managed with chemotherapy and anticoagulation.
Case Presentation
A 13-year-old girl presented with complaints of fever, dry cough, and progressive shortness of breath of 7 days duration. There was no history of chest pain, palpitations, or syncope. She had also been symptomatic with menorrhagia and dysmenorrhea for the preceding 6 months.
On examination, she had fever with a maximum recorded temperature of 38.3°C, tachycardia (heart rate 134 per minute), tachypnoea (respiratory rate 28 per minute), and oxygen saturation of 88% (in ambient air by pulse oximetry). Blood pressure was normal. She had facial puffiness and neck veins were engorged (Figure 1). On systemic examination, decreased left sided chest movements with dullness on percussion and decreased air entry in left infra-axillary and infra-scapular regions were noted. There was fullness in the abdomen with a palpable abdomino-pelvic mass.
On further evaluation, she had anemia (hemoglobin 8.8 g/dl) and leucocytosis (total leucocyte count 16,800 cells per mm3 with neutrophils of 80%). Her liver function and renal function tests were normal. Work-up for common endemic tropical infections (dengue, malaria, enteric fever, and scrub typhus) and coronavirus disease 2019 was negative. D-dimer level was raised at 1,741 ng/ml.
Chest radiograph showed left sided pleural effusion (Figure 2). Thoracentesis was performed and 300 ml pleural fluid was drained which was milky white in appearance. Analysis of the pleural fluid revealed an exudative character with 95% lymphocytes, and elevated levels of lactate dehydrogenase (480 U/l) and triglyceride (930 mg/dl) while serum triglyceride level was 133 mg/dl. Adenosine dehydrogenase level was normal. Staining and culture of pleural fluid were negative for infection.
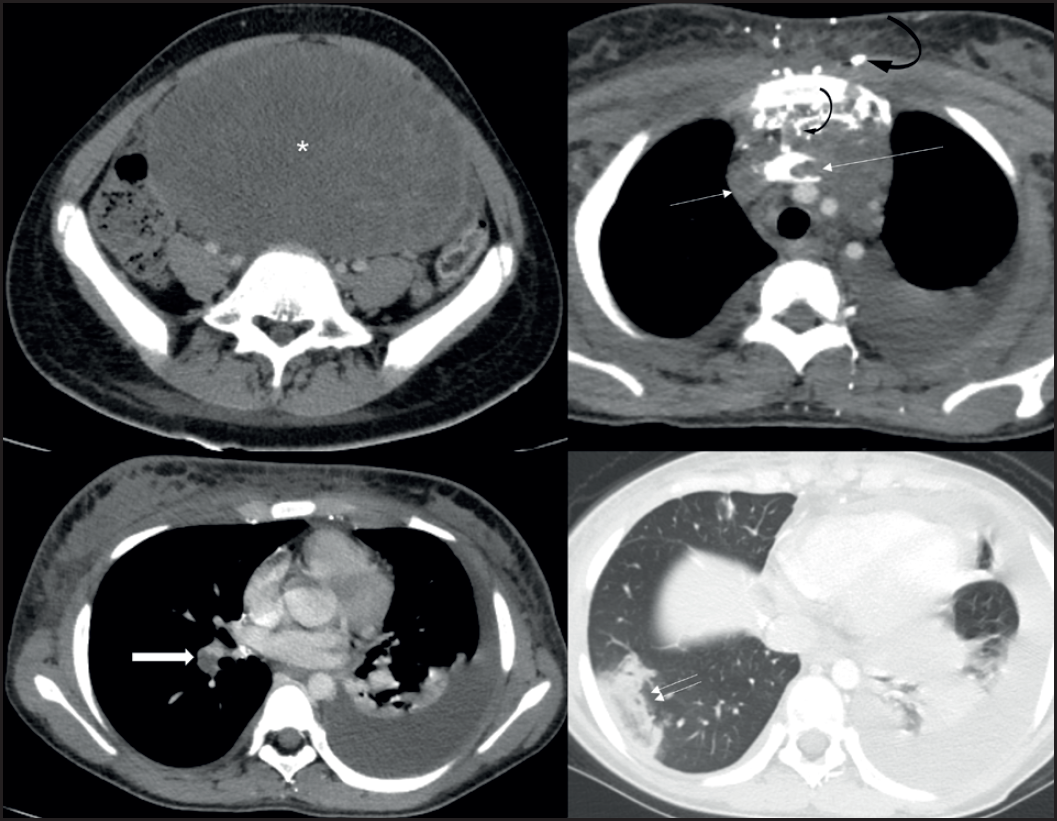
CT scan images showing the large solid-cystic ovarian tumor (asterix), left-sided pleural effusion and minimal right-sided effusion, thrombosis of SVC and left brachiocephalic vein (thin arrows) with multiple collaterals in thorax and mediastinum (curved arrows), pulmonary artery thrombus (thick arrow) and pulmonary infarct in right lower lobe showing reverse-halo sign (double-arrow).
Ultrasonography and subsequently contrast-enhanced computed tomography (CECT) scan of thorax, abdomen, and pelvis (Figure 3) showed an abdomino-pelvic solid cystic lesion, likely ovarian in origin with right hydroureteronephrosis. There was thrombosis of superior vena cava (SVC) and left brachiocephalic vein, along with thrombus in segmental branch of right pulmonary artery. Lower lobe of the right lung showed a peripheral opacity with central ground-glass haze and a surrounding dense rim of consolidation [reverse-halo sign (RHS) or atoll sign] that was interpreted as pulmonary infarct. Left-sided pleural effusion along with a small right-sided pleural effusion was seen. There was no evidence of pelvic vein or inferior vena cava thrombosis.
Carcinoma antigen 125 was raised at 350 IU/ml and alpha-fetoprotein at 2,873 ng/ml while beta human chorionic gonadotropin level was normal.
A diagnosis of ovarian yolk-sac tumor with chylothorax, SVC thrombosis and pulmonary embolism was made. Contrast-enhanced magnetic resonance imaging of abdomen was suggestive of a large well-defined, smooth-marginated, oval solid-cystic lesion in lower abdomen and pelvis, predominantly left sided and extending across midline, measuring 10 × 16 × 20 cm with cystic areas within lesion. Few foci of blooming were noted within the lesion, causing displacement of bowel loops to the right.
Since the patient had no central line insertion or any other obvious etiology for SVC thrombosis, the possibility of ovarian neoplasia related procoagulant state leading to SVC thrombosis was kept. Either an embolus from here or an in-situ thrombosis in the segmental branch of right pulmonary artery might have led to the pulmonary infarct. Thus, the thrombosis of SVC and left brachiocephalic vein leading to central venous hypertension, could have resulted in chyle leak from thoracic duct.
She was managed with anticoagulation for SVC and pulmonary artery thrombosis. In view of hypoxia, pulmonary infarction, and SVC thrombosis, she was considered medically inoperable after a multi-disciplinary discussion. She was started on chemotherapy with bleomycin, etoposide, and cisplatin.
Her general condition improved with chemotherapy and pleural effusion had regressed completely. Repeat CECT showed resolution of thrombosis and significant regression in the size of the ovarian mass. After three cycles of chemotherapy, she underwent interval debulking surgery involving left salpingo-oophorectomy with infra-colic omentectomy and multiple peritoneal biopsies.
Pathological specimen showed left ovarian mass with no residual tumor in the resected specimen or omental biopsy (chemotherapy response score 3- complete pathological response). She received one more cycle of adjuvant chemotherapy after surgery and is currently in remission.
Discussion
This patient was diagnosed as OGCT with venous thromboembolism and chylothorax. Prothrombotic state has been commonly associated with surface epithelial tumors of ovary, but rarely with OGCTs [2,3,5]. We inferred that the patient developed a thrombosis of SVC and left brachiocephalic vein, due to underlying prothrombotic state. RHS on CT chest, in presence of thrombus in pulmonary artery was suggestive of pulmonary infarct [6].
Thoracic duct usually joins the venous system at the junction between left internal jugular vein and left subclavian vein (formation of left brachiocephalic vein). Thrombosis of left brachiocephalic vein or SVC, as in our case, would increase central venous pressure leading to chyle leak and chylothorax. The phenomenon, although rare, has been described in pediatric [7-9] as well as adult age group [3,10-13].
Chylothorax has previously been described with ovarian tumors and rarely with OGCT. Etonyeaku et al. [14] described a case of chylothorax and chylous ascites in a case of bilateral primary Burkitt’s lymphoma of the ovaries. Gupta et al. [15] also reported a case of sex cord stromal tumor of ovary that developed left sided chylothorax after oophorectomy. Katwal et al. [3] presented a case of OGCT with bilateral chylothorax and central vein thrombosis, like our case. The authors suggested that mediastinal lymphadenopathy along with the hypercoagulable state due to malignancy led to central vein thrombosis. Thus, the blockade led obstruction to the flow of thoracic duct, resulting in chylothorax.
Our patient had a large left-sided chylothorax, with a small right-sided effusion and no ascites. There were no significant nodes. In the absence of ascites, subdiaphragmatic lymphatic duct obstruction by the tumor was not a likely cause of chylothorax. Thus, the most probable explanation for chylothorax in our patient is central venous thrombosis. Conversely, chylothorax leading to a hypercoagulable state and venous thrombosis, due to loss of antithrombin in chyle, has also been suggested.
Conclusion
The case highlights that OGCT in young females can present as a prothrombotic state with central venous thrombosis. Thrombosis of SVC or left brachiocephalic vein can lead to central venous hypertension resulting in chyle leak from the thoracic duct. The case also underscores pulmonary infarct as an important differential of a solitary peripheral pulmonary lesion presenting with RHS. This report exemplifies application of “Occam’s razor” in Medicine - different varying aspects of clinical presentation are usually linked together, and a unified simplistic explanation should always be attempted.
What is new?
Chylothorax has previously been described with ovarian tumors and rarely with OGCT. Katwal et al. [3] presented a case of OGCT with bilateral chylothorax and central vein thrombosis, like this case. The case highlights that OGCT in young females can present as a prothrombotic state with central venous thrombosis. Thrombosis of SVC or left brachiocephalic vein can lead to central venous hypertension resulting in chyle leak from the thoracic duct. The case also underscores pulmonary infarct as an important differential of a solitary peripheral pulmonary lesion presenting with RHS.



