Background
Since COVID-19 first emerged in December 2019, patients across all healthcare settings have been affected, including pregnant women and their babies. Understanding of transmission of the virus, clinical symptoms, and management strategies in the neonatal period is still emerging.
A review of the literature generally describes experiences prior to the emergence of the delta variant in December 2020, which had become the most dominant strain globally at the time of this case [1]. During this time, there was uncertainty around vertical transmission, with around 3% of infants born to COVID-19 positive mothers also testing positive in the days following birth [2-5] even in the absence of exposure via parental contact [6].
High levels of virus have been identified in inflamed placental tissue, which has been suspected as a cause for vertical transmission in the third trimester [4,7] and there are reports of infants being affected in-utero at early gestations [8].
With non-delta variant infections, affected babies were generally well, with minor respiratory or no symptoms, [2,3,7,9] but maternal infection was associated with an increased risk of the newborn being admitted to the neonatal unit [3]. Maternal COVID-19 infection has been associated with increased requirement for Caesarean section, primarily for respiratory deterioration in mother [5,10], many at preterm gestations [11]. COVID-19 infection has been shown to cause placental failure [8] and may precipitate spontaneous premature labor [12]. There have been no reports on the efficacy or side effects of treatments - such as remdesivir or corticosteroids - used in adult populations in the newborn population.
This report describes the case of a very premature infant with vertical transmission of the delta variant of the virus which resulted in significant clinical illness with subsequent trial of compassionate off-label treatment with remdesivir. This case report characterizes presentation of COVID-19 in a premature neonate and their response to remdesivir compared to the widely reported data with other variants of the virus.
Case Presentation
This infant was born to a 26-year old primiparous woman following a low risk pregnancy. Antenatal screening was unremarkable, and the only regular medication used was sertraline. Of note, the mother had not been vaccinated against COVID-19. At 27+5 weeks post menstrual age, she developed symptoms of pyrexia and lethargy, with a positive COVID-19 rapid antigen test confirmed within 48 hours. Five days after the onset of symptoms, she presented to maternity services with a one day history of absent fetal movements, with a pathological trace on the cardiotocogram. The baby was delivered by emergency Caesarean section with one dose of betamethasone given 1 hour prior to delivery alongside a loading dose of magnesium sulphate.
A liveborn male infant weighing 1,193 g at 28+3 weeks post menstrual age was born. The umbilical cord was clamped at birth, in keeping with local policies at the time. The baby was immediately moved out of theatres and to the neonatal stabilization room, as per hospital guidelines at the time. Standard Neonatal Life Support algorithms and guidelines were followed. Apgar scores were 4, 7, and 9 at 1, 5, and 10 minutes respectively. Arterial cord gases were acidotic with a pH of 7.06, base excess of -12.4 and lactate 11.1 mmol/l. At 11 minutes, the baby was intubated, and surfactant was given as a standard of care. He was admitted to the neonatal intensive care unit’s isolation room where he was started on conventional ventilation and had central access sited. The initial blood gases showed a degree of antenatal compromise with a pH of 7.14, base excess -16.7 and lactate of 14.9 mmol/l. The day 3 nasopharyngeal swab was positive for COVID-19. This swabbing plan and infection control strategies was in line with recommendations from the Royal College of Pediatrics and Child Health at the time [13]. The baby was taken from theatres immediately and subsequently had no direct contact with either parent due to the need to isolate following the COVID-19 infection.
During the first 5 days, he remained ventilated whilst clinically stable, in 25%-45% oxygen. Antibiotics were given empirically; a patent ductus arteriosus was managed conservatively and one packed red cell transfusion was given for anemia. By day 5, the oxygen requirement rose to 80% and higher inspiratory pressures needed on the ventilator. Following a suspected pulmonary hemorrhage with respiratory acidosis on day 8, high frequency oscillatory ventilation was started with modest clinical improvement.
On day 10, there was an acute and severe deterioration whilst on the ventilator with problems in both carbon dioxide clearance and oxygenation. Further surfactant was given and inhaled nitric oxide started. COVID-19 was presumed to be the most likely cause for the deterioration. Comprehensive fungal, bacterial and viral workup yielded no results. Other causes for the acute deterioration were investigated, with normal echocardiogram and abdominal radiographs.
Hydrocortisone was started as per the RECOVERY trial protocols [14] in view of potential COVID-19 pneumonitis, which was in keeping with chest radiograph appearances. Antibiotic and antifungal cover were added (Figure 1), although throughout this time there was no rise in inflammatory markers. At the time of this deterioration, the C-reactive protein peaked at 7 mg/l, with a white cell count of 6.2 × 109/l and platelets of 101 × 109/l.
At this stage repeat nasopharyngeal COVID-19 swabs confirmed the same P681R mutation as seen in his mother’s samples, and the replication counts were dropping, suggesting a greater burden of disease (Figure 2). The disease burden was greater by a factor of approximately 10,000 times between the first and second test results.
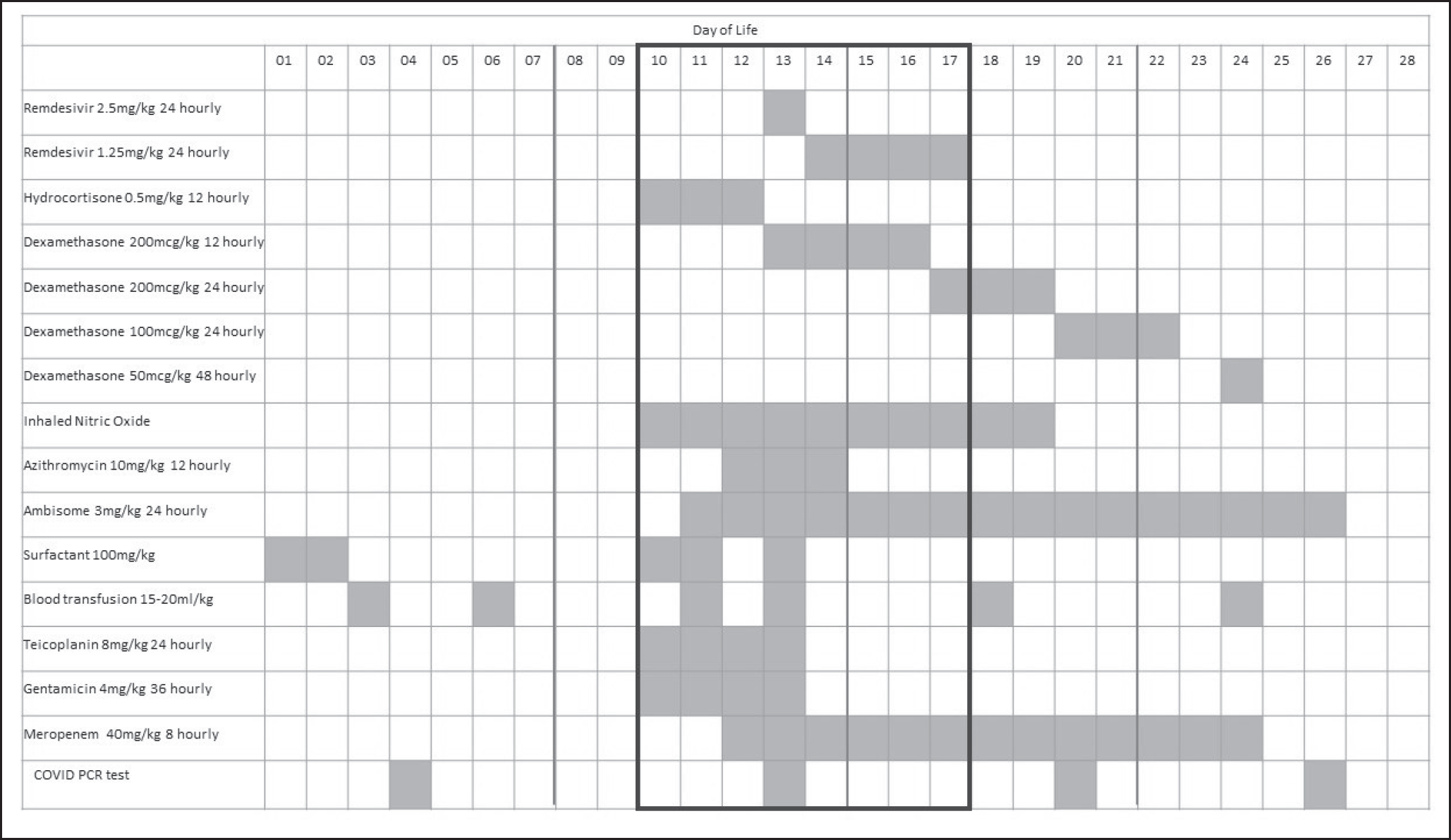
A summary of the medical treatments given on each day. The boxed area highlights the time of deterioration and most severe sickness.
There had been no improvement after 3 days of hydrocortisone treatment. The case was discussed with regional infectious disease specialists as well as pediatric intensivists. As a result, dexamethasone was added and remdesivir was started on compassionate grounds in view of prematurity and confirmed COVID-19 who remained critically unwell despite maximal intensive care. Remdesivir was given at 2.5 mg/kg once daily for 1 day and then 1.25 mg/kg once daily for 4 more days.
This brought about a clinically significant improvement over the next 72 hours and the baby was extubated to non-invasive ventilation on day 20, 10 days after a life-threatening deterioration. He continued to make good progress with a gradual weaning off respiratory support. He was discharged home at 39+2 weeks with no additional oxygen requirement. Throughout his admission he required no inotropic support. Cranial ultrasound screening showed a unilateral, small grade 1 intraventricular hemorrhage, which resolved prior to discharge. He remains under the neonatal follow up programme. There were no side effects noted to date from remdesivir, which are known to include hypersensitivity reactions or rashes in the pediatric population.
Successive COVID-19 swabs remained positive in the first month, at which stage the clinical recovery was sufficient not to warrant repeated testing. The placental histology showed extensive and excessive early necrosis throughout all sections of the placenta with fibrin filling the intervillous space. All of these changes were in the same phase, suggesting acute COVID-19 placentitis as the cause for the fetal distress and need for preterm delivery.
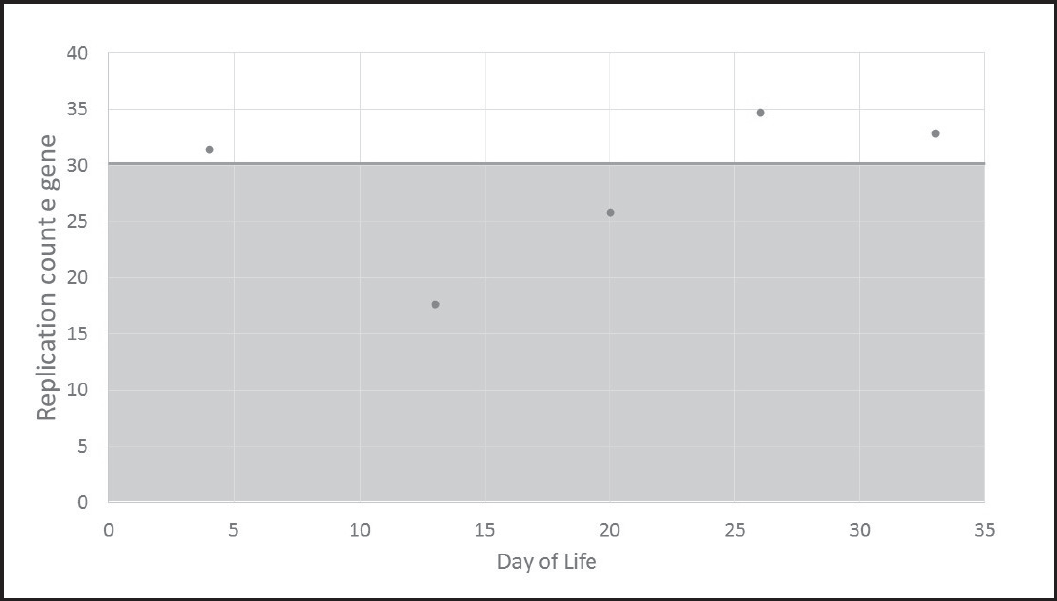
A summary of the replication counts of the COVID-19 e gene throughout this patient’s admission. The peak of viremia is associated with the deterioration in clinical status as a low replication count relates to a high burden of disease. Each reduction in the replication count by one corresponds to approximately double the viral load. The locally agreed threshold of below 30 counts is marked as indicative of a high risk of infectivity to others.
Following delivery, the infant’s mother required escalating respiratory support, and subsequent recovery was complicated by pulmonary embolism necessitating anticoagulation.
Discussion
This case-report describes severe clinical illness due to COVID-19 in a very premature infant and is the first to outline the use of remdesivir in a compassionate off-label setting in this population.
The first sign of the infant being affected was reduced fetal movements, which became apparent four days after the mother first showed symptoms suggestive of COVID-19. This was subsequently shown to be due to placentitis with early placental failure resulting in fetal compromise and significant initial metabolic acidosis at birth. It is plausible that the infant became infected with COVID-19, with transplacental migration of the virus. The same mutation in the virus was detected in both mother and in child, with no realistic postnatal exposure due to the resuscitation practices and patient isolation from admission. This could be considered as convincing evidence for the vertical infection of the delta variant of COVID-19 from mother to child, as well as the rapid progression of antenatal effects on the placenta. It is possible that the degree of placentitis may be related to vertical transmission, but this is outside of the scope of this case report and has not been explored further.
At the time of this case, the delta variant of COVID-19 predominated. Subsequently, the omicron variant has become more prevalent and further variants may yet develop. As the nuanced differences between strains are not fully understood, it is conceivable that the good response seen to remdesivir, and dexamethasone could be seen in any severe COVID-19 infection.
In the first few days after delivery, the baby had confirmed infection with COVID-19, but was not unstable or critically unwell. However, from admission he did have higher ventilation requirements than might have been expected from surfactant deficiency respiratory distress syndrome alone. Although he did not get the benefits of a full course of antenatal steroids, his lung disease was still worse than anticipated for a baby of that gestation.
Severe deterioration in his ventilatory parameters happened on day 10 after birth, which was 15 days after the onset of maternal symptoms, and 11 days after fetal movements reduced; potentially mirroring the clinical picture in adult populations: an initial period of stability followed by deterioration. This patient had serial swabs and was shown to have reducing replication counts for COVID-19 around the time of clinical deterioration. A lower replication count correlates to a higher viral load and can therefore be used as a proxy for the overall disease burden increasing at that stage. It was used as a marker for his viral burden and in determining isolation measures, but not clinical management. A replication count of 30 polymerase chain reaction cycles was used as the threshold for stepwise reduction in isolation and barrier nursing, as set by local infection control policy.
COVID-19 is known to be associated with hyper-inflammatory processes. As such, it was interesting that this patient did not mount any significant inflammatory response. C-reactive protein was very marginally above the normal upper limit, and platelets mildly reduced. There was no improvement with hydrocortisone as monotherapy, and the patient only improved when hydrocortisone was switched to dexamethasone with concurrent remdesivir. It is unclear how much of the recovery was due to the anti-inflammatory effects of the corticosteroids, or the antiviral effects of the remdesivir; or whether the combination was responsible.
Conclusion
Given the severity of the illness, in the context of proven COVID-19 infection, a multidisciplinary team involving specialists across different specialties and hospitals agreed that remdesivir could be trialed as he approached maximal intensive care treatment without improvement. This RNA polymerase inhibitor has been used safely in the context of Ebola virus in children under five days of age [15], but there is no comparable data for the use in COVID-19 in newborn infants and none in premature infants. Although this was started in the context of multiple concurrent drug therapies, this report has shown that it is feasible to use remdesivir in preterm neonates, acknowledging the extreme and compassionate circumstances of this case.

