Background
Invasive lobular carcinoma (ILC) of the breast accounts for 5%-15% of all breast cancers and is the second most common histological subtype after invasive ductal carcinoma (IDC) [1]. We know that it differs in clinical and biological properties when compared to IDC. For the ILC group, older age, larger tumor size, more advanced stage, lower grade, more frequent metastasis disease, more positive lymph nodes, and estrogen-receptor-positive (ER)/progesterone receptor-positive (PR) were reported [2-4]. Therefore, the prognosis of ILC compared to IDC remains controversial. Due to the follow-up period of the study series being 5 years or less, the overall survival is found to be similar or higher to the IDC, while we see a lower survival rate in the studies with a follow-up period of more than 5 years [5-12].
Metastatic development of ILC, unlike IDC, can occur in atypical regions such as the gastrointestinal tract, myometrium, endometrium, cervix or ovaries, and peritoneal surface, gallbladder, leptomeninges, orbital soft tissue apart from common sites (liver, lung, bone, central nervous system, and distant nodes involvement) metastasis [2,13-17]. In our case, metastasis developed from an atypical region, which we did not find in the literature. In addition, we know from case-based literature sources that while only 2%-3% of patients with systemic cancer develop orbital metastases, these cancers are ILC histological subtypes of the breast, especially in female patients [13,14]. Although right orbital swelling occurred in our patient and facilitated the diagnosis, there was no direct orbital involvement. There was swelling in the right eye due to the pressure of the mass starting from the buccal mucosa and growing upward.
It was our first experience that breast cancer had metastasized to the buccal mucosa in the ILC subtype. We wanted to share this atypical metastasis site and the treatment we applied and the response we received to the treatment with the literature.
Case Presentation
A 50-year-old female, the pre-menopausal patient had left breast-conserving surgery and sentinel lymph node sampling after biopsy due to a mass in the left breast in 2011. Histopathology was ILC, tumor diameter was 3 × 2, 5 × 2 cm, reactive hyperplasia of two lymph nodes removed, the lympho-vascular invasion was negative, the perineural invasion was positive, and skin involvement was negative. ER 70% positive, PR 90% positive, Cerb-B2 has been found negative and Ki67 8%. The subgroup was luminal A. The patient who was staged as T2N0M0 was treated with six cycles of Cyclophosphamide, Epirubicin, and Fluorouracil chemotherapy regimen and radiotherapy (RT) to the left breast in an external center. The patient, who had regular follow-ups, had used tamoxifen citrate 20 mg/day for 8 years. In the last follow-up period, a slight elevation of the CA15-3 tumor marker is detected in the blood, but since there was a slight elevation, it was not taken into account (considered) by the physician in the center where it is followed. In those days, the patient applied to the Ear Nose and Throat polyclinic of the same hospital due to hearing loss in her right ear. No pathology is detected in the tests performed. 6 months after the onset of her complaints, a swelling on the right eye developed. Furthermore, the patient applied to the oncology outpatient clinic of our faculty (Figure 1a).
A head and neck magnetic resonance imaging (MRI) were performed immediately after our patient applied to us. In head and neck MRI, soft tissue formation with dimensions of 59 × 31 mm was detected in the axial plane, extending from the level of the right infratemporal fossa to the right buccinator muscle and right maxillary sinus in front of the alveolar process. In the right periorbital region, there was a thickness increase of up to 1 cm in the thickest part of the globe, which was evident in the anterolateral. In addition, there was no bone destruction in the right infratemporal region in maxillofacial computed tomography (CT), and no additional organ metastasis was detected in positron emission tomography (PET)-CT performed for systemic scanning (Figure 2).
After the mass causing the patient’s complaints were detected on MRI, a hard viscous tumoral mass behind the fat tissue of the gingivobuccal sulcus was reached and an incisional biopsy was performed by Trakya University Faculty of Medicine, Department of Otorhinolaryngology. In the histopathological examination of the tissue, carcinoma infiltration was detected in the plasmacytoid signet ring cell morphology. It was reported that ER, PR, and GATA binding protein 3 (GATA-3) positivity, and Ki67 20%-30% were detected immunohistochemically, Figure 3d and e depending on the morphology, it could be ILC and primary breast cancer. The buccal metastasis subgroup was Luminal B. Since the histopathology of breast cancer was also ILC, a retrospective comparison was made. Buccal metastasis due to breast cancer of our patient was accepted and treatment planning was made.
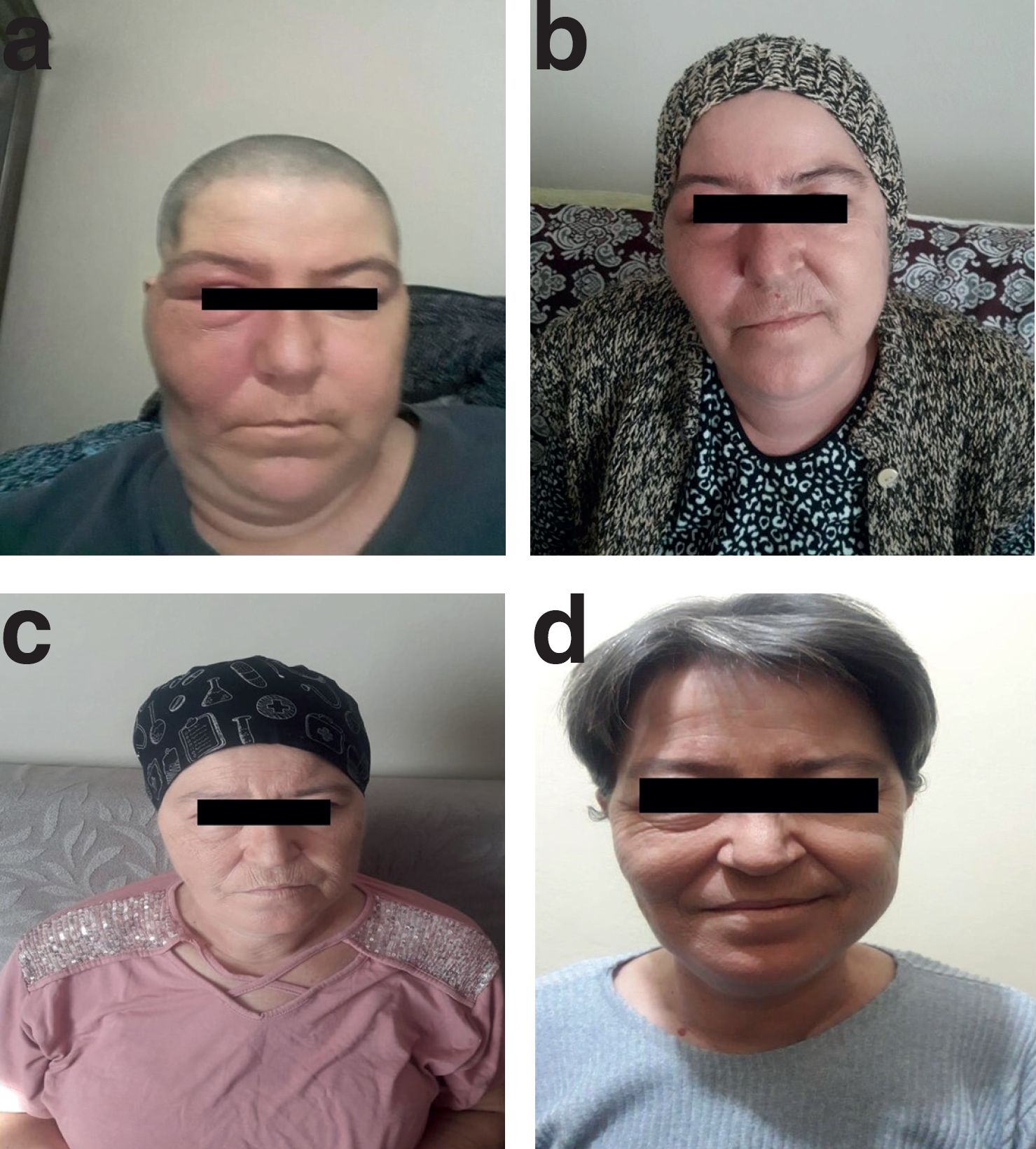
(a) The appearance of our patient’s complaint of swelling in the right cheek and right eye before RT (b) The appearance of our patient 2 months after the completion of RT (c) The appearance of our patient 6 months after the completion of RT (d) The appearance of our patient 15 months after the completion of RT.
Due to buccal metastasis detected 9 years after the diagnosis of breast cancer ILC, by medical oncology, Tamoxifen treatment was terminated and Anastrozole treatment was started. Systemic chemotherapy was not required. A RT plan was also made for the metastatic buccal mass. RT was applied to the right buccal and orbital region of the patient, who did not detect any other metastases in PET-CT imaging and was evaluated as inoperable. 30 Gy in 12 fractions (2.5 Gy per fraction) with volumetric modulated arc therapy (VMAT) technique and 15 Gy in 6 fractions (2.5 Gy per fraction) boost RT with the adaptive technique were applied (Figures 4 and 5). At the follow-up 2 months later, clinical response was observed in the patient, and significant regression of the lesion was observed in facial and orbital MRI. At the 15th month follow-up of the patient, clinical and radiological remission continues. All clinical complaints regressed (Figure 1c and d).
Discussion
The incidence of ILC has been increasing in recent years and the number of studies proving different clinical and biological features of the disease from IDC is increasing [2-4]. The most obvious difference is that ILC has the potential to include unusual sites concerning the pattern of metastatic spread from IDC. The ability of ILC to make atypical metastases becomes clearer when examining the case-based literature [2,13-17]. However, in our literature review, we did not find any case report of breast metastasis in the buccal region due to ILC.
Although our patient was diagnosed with primary breast cancer 11 years ago, ILC chose the buccal mucosa as the atypical metastasis site, and the diagnosis of metastasis was started with a 6-month delay. Could we understand before 6 months that we are facing the unusual metastasis development of ILC? When we examine the retrospective laboratory and clinical conditions of our patient, we see that the CA15-3 marker in the blood is slightly elevated. Hearing loss begins in the right ear at that time. If CA15-3 height was considered and systemic imaging could be performed, metastatic involvement could have been detected much earlier.
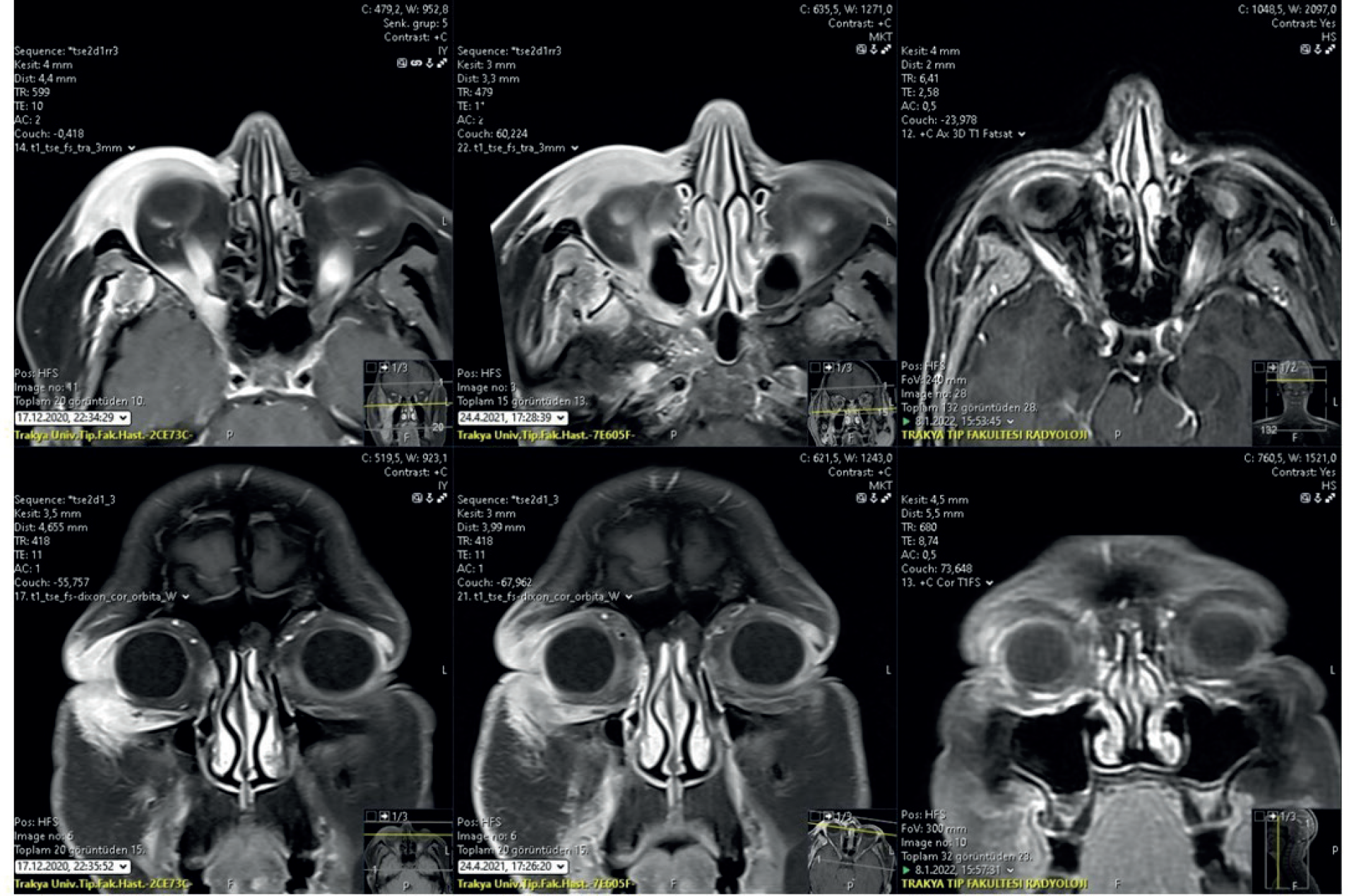
Contrast-enhanced axial T1-weighted sections showing significant T1-isointense soft tissue density and contrast enhancement around the right periorbital region, pre-septal area, superior orbital fissure, and chewing area, respectively, before RT 2 months after RT is completed, and 1 year later, MR images before treatment.
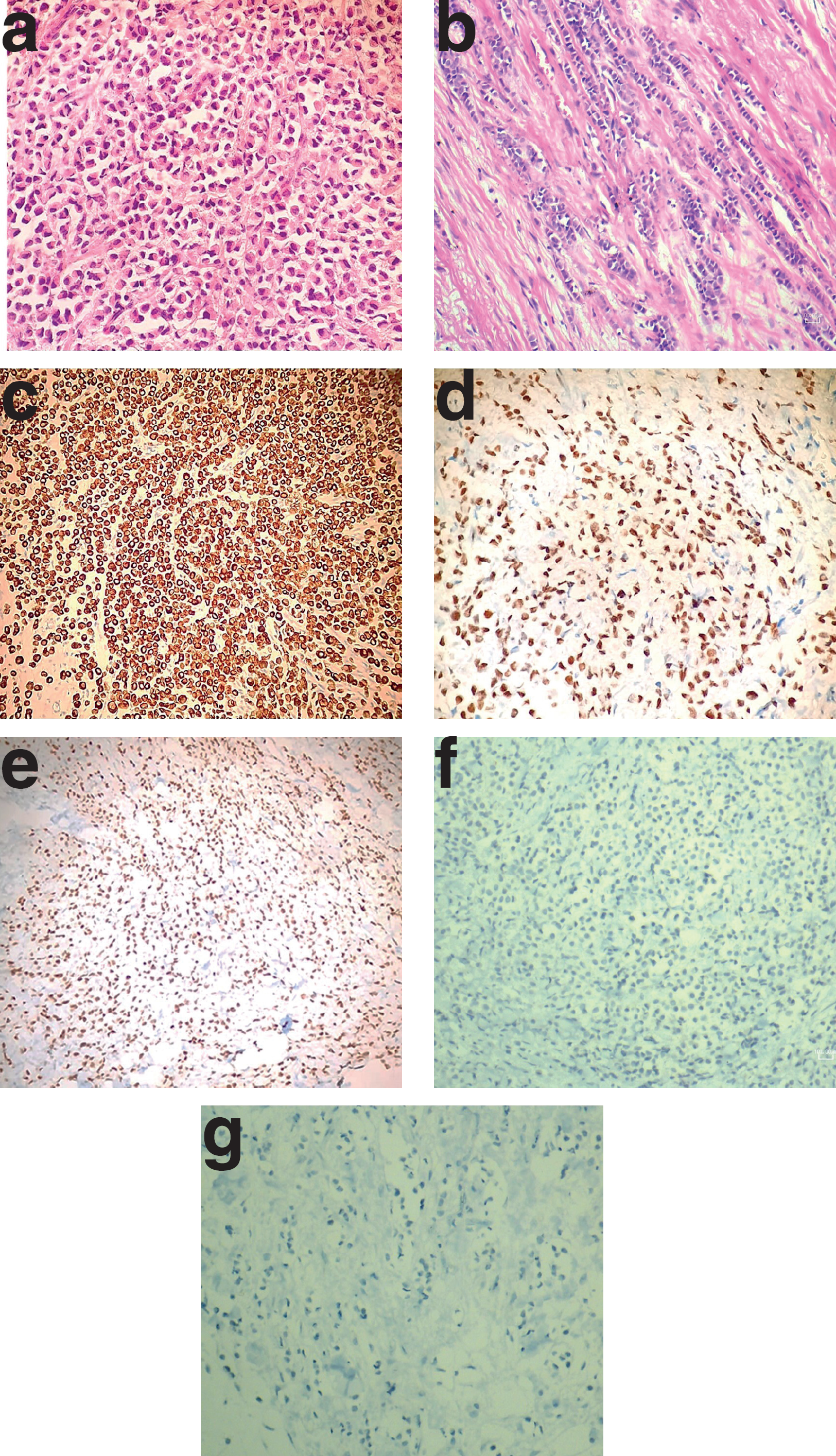
(a) Histopathological findings of the buccal mucosa. The pattern of small cells (hematoxylin and eosin, ×400 magnification) suggests breast tumor tissue from an ILC of the breast. (b) Histopathological findings of a breast tumor. The pattern of small cells (hematoxylin and eosin, ×400 magnification) suggests breast tumor tissue from an ILC of the breast. (c) CK7 positivity in the metastatic (buccal tissue) tumor tissue. (d) ER positivity in the buccal tumor tissue (e), GATA3 positivity in the metastatic buccal tumor tissue. (f) E-cadherin negativity in immunoreactivity staining in buccal metastasis tissue of ILC. (g) E-cadherin negativity in immunoreactivity staining of ILC in breast tumor tissue.
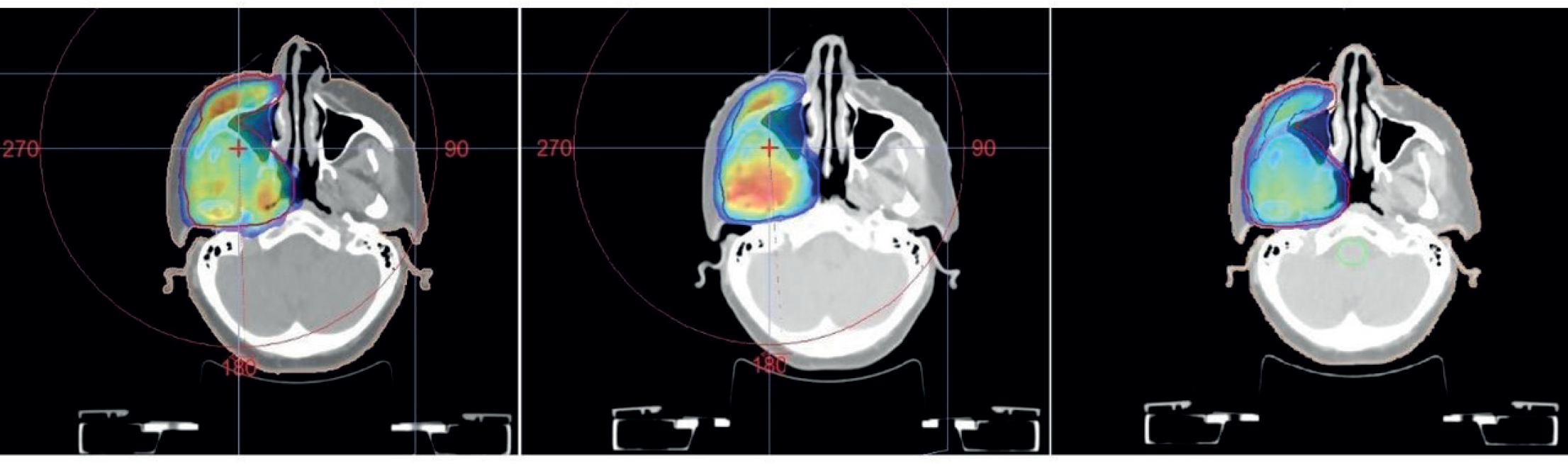
(a) VMAT RT plan for 30 Gy in 12 fractions. (b) Additional dose to shrink tumor volume with VMAT planning for 15 Gy in 6 fractions. (c) Total 45 Gy VMAT plan.
In another case, the diagnosis was made by biopsy taken from the mass due to peritonitis carcinomatosis in the laparotomy performed during the investigation due to constipation. The patient was diagnosed with ILC-related cancer of the breast and ILC metastasis in the mass that compresses adjacent to the rectum, as a result of a mild elevation of CA15-3 in the blood and a suspicious appearance on mammography, and chemotherapy was started quickly [17]. In our case, during the follow-up of a patient diagnosed with breast cancer ILC, the CA15-3 level increased in the blood before the abnormal situation developed.
E-cadherin loss is an easy diagnostic tool with the most common immunohistochemical method, as it precisely directs the pathognomonic and ILC phenotype due to e-cadherin mutation. Moreover, it is estimated that this loss occurs not only in the tumor cell but also in the stromal, endothelial, and extracellular matrix tissue. The unusual preference for sites of metastasis by ILC is thought to be related to the loss of the adhesion molecule E-cadherin [3,12-18]. In our case, we detected loss of E-cadherin both in the breast tumor that we looked at retrospectively and in the biopsy tissue of the buccal mucosa. Moreover, E-cadherin loss was more severe in metastatic tissue than in tumor tissue in the breast (Figure 3f and g).
GATA3 is an important determinant of disease outcome in breast cancer patients. GATA3 binding protein recognizes G-A-T-A nucleotide sequences, is a transcription factor, and activates or represses target gene promoters. GATA3 is expressed in primary and metastatic ductal and lobular carcinomas of the breast. Low expression of GATA3 is an important marker of disease-related death in all patients, as well as in subsets of ER-positive or low-grade patients. Therefore, GATA3 expression is used as a prognostic indicator in breast cancer, both at low and high rates [19,20]. Extended and severe GATA3 positivity was also detected in our case, and this may have contributed to the development of unusual metastases with loss of E-cadherin.
Staining with Cytokeratin-7 (CK-7) is not specific but suggested in the report, when a 5% tumor cell positivity is detected in metastatic biopsy studies of unknown primary in daily routine, the possibility of primary breast cancer [21,22]. In the biopsy tissue taken from the buccal mucosa of our patient, CK-7 positivity was detected in all tumor cells. The results of studies on CK-7 positivity, which is an important marker for the diagnosis of metastatic breast cancer, and its prognostic value are still controversial.
While the primary breast cancer of our patient was Luminal A, buccal metastasis changed to Luminal B. An article investigating the subgroup chance of recurrence or metastasis showed that the change in subgroup and receptor status effect survival negatively. Understanding the biology of subgroup change in metastasis will be important to developing better treatment strategies [23].
Conclusion
ILC is a subtype of breast cancer that is characterized by unusual metastasis. Therefore, during the follow-up of these patients, it is necessary to monitor both laboratory values and clinical complaints very carefully. We would like to express the necessity of not delaying the diagnosis of metastasis in patients with ILC who were previously diagnosed with breast cancer and to check whether there is a primary focus on the breast tissue of patients with unusual metastases.


