Background
Cerebral gas emboli (CGE) are rare cause of cerebral infarction, complicating 0.002% of hospitalizations, with a mortality rate of 21% [1,2]. Arterial emboli usually produce stroke-like symptoms and focal neurological deficits. Diagnosis is based on these symptoms after a precipitating event and brain imaging [3]. A 2018 systematic review identified the most common medical procedures to cause CGE: central venous catheter insertion (22%), cardiopulmonary bypass (18%), lung biopsy (10%), endoscopic retrograde cholangiopancreatography (6%), and hemodialysis (6%) [4]. Other sources include gas insufflation from thoracoscopic and laparoscopic surgeries. The mainstay of treatment for CGE is hyperbaric oxygen therapy (HBOT), if available [1].
Case Presentation
A 70-year-old female underwent video-assisted thoracoscopic lower lobectomy for right lung adenocarcinoma. Her past medical history included left breast cancer, hypertension, and asthma. Her breast cancer had been treated by wide local excision with axillary node clearance and she was in remission at the time of this presentation.
Carbon dioxide (CO2) insufflation was used during the thoracoscopy to allow better visualization of lung tissue while being dissected. A pulmonary vein injury occurred during dissection of the hilar vessels, which required conversion to mini-thoracotomy for its repair. Postoperatively, while in the recovery room, the patient’s conscious state deteriorated and she became quadriparetic. She was transferred for urgent imaging.
Initial brain computed tomography (CT) demonstrated subtle cerebral edema in both hemispheres, worse on the right (Figure 1a). A CT angiogram showed that all vessels were patent from aortic arch to cranium.
A left hemiplegia and right hemiparesis were clinically apparent. Repeat head CT was obtained 4 hours after the initial scans. It revealed extensive edema through the right frontoparietal lobes with loss of the normal cortical gyriform pattern and local obliteration of the subarachnoid space. Findings suggested an evolving ischemic cortical infarct (Figure 1b).
Brain magnetic resonance imaging (MRI) was performed 48 hours later, which revealed multiple acute cortical infarcts involving the anterior and middle cerebral artery territories of the right hemisphere, but some infarcts also present in the anterior cerebral artery territory of the left hemisphere (Figure 2). This was highly suggestive of embolic shower of central origin. There was associated edema, mild mass effect, and early subfalcine herniation. A thrombo-embolic source was excluded through imaging of the carotid arteries, aortic arch, the heart, lower limbs, and arrhythmia screening. A small patent foramen ovale (PFO) was noted on transthoracic echocardiogram and thought to be incidental.
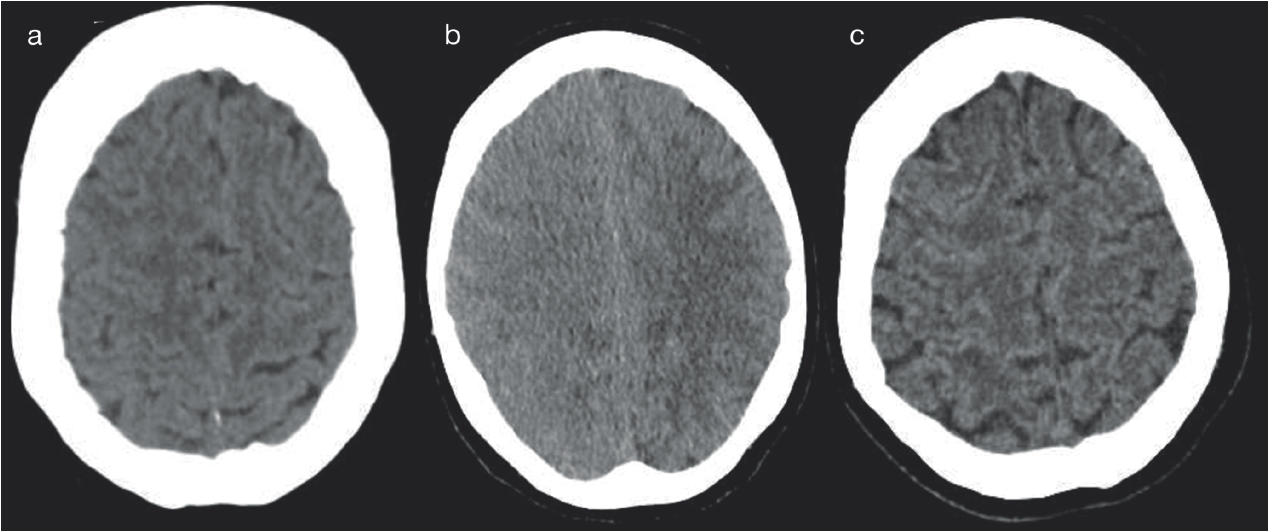
Acute CT imaging demonstrates subtle edema on initial CT(a), with evolution imaging on CT 4 hours later (b) where extensive edema, effacement of the sulcal and gyral pattern, loss of gray-white matter differentiation and mild mass effect is seen. At 1-month post event, repeat CT brain is normal (c).
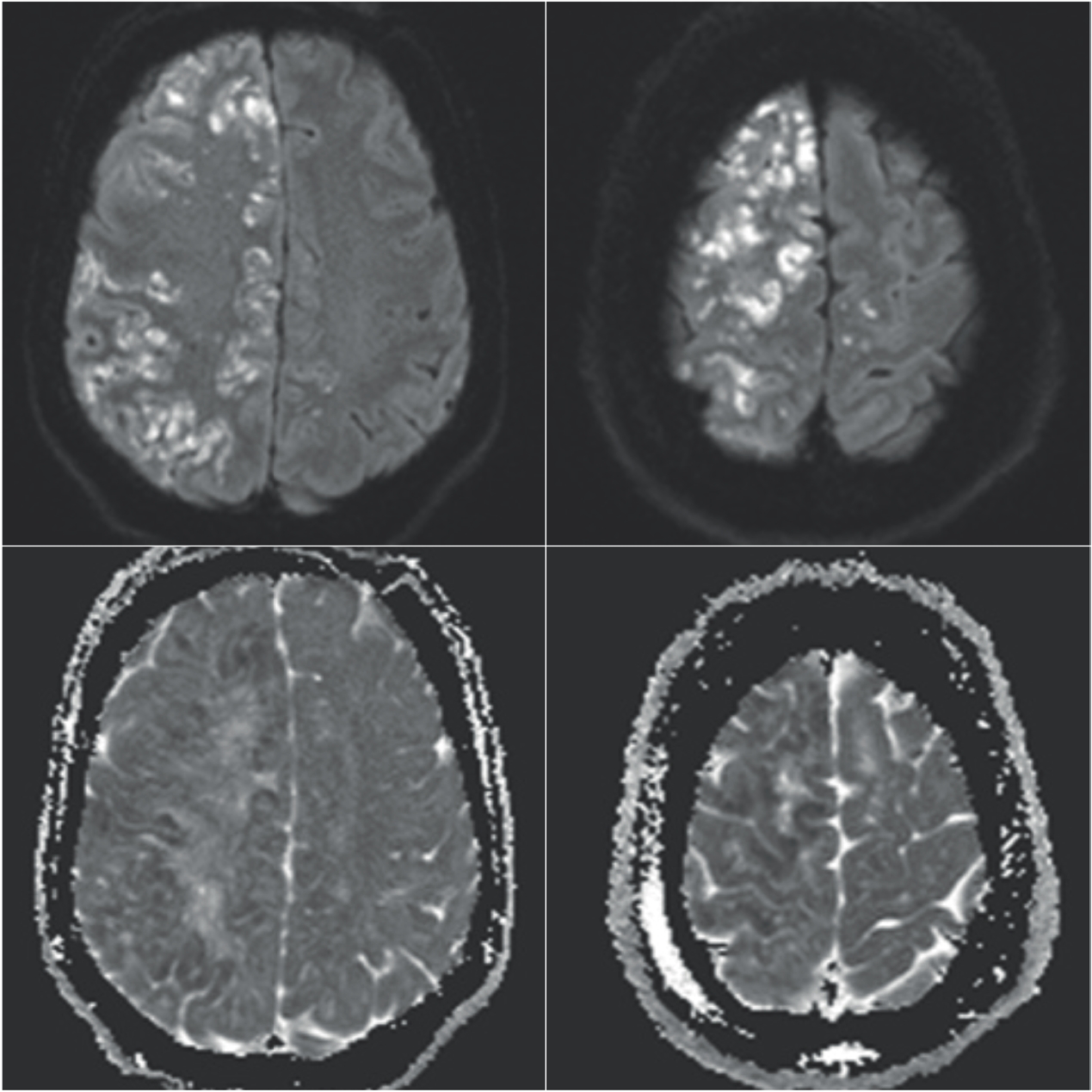
MR b1000 (a,c) and corresponding ADC map (b,d) demonstrate innumerable scattered small infarcts along the cortical gyri of the frontal and parietal lobes on the right, involving the anterior and middle cerebral artery territories. Occasional scattered punctate infarcts within the left anterior cerebral artery territory.
The patient received several days of conservative management, which incorporated high fraction of inspired oxygen (FiO2) therapy. She improved enough to be discharged from the intensive care unit (ICU). She proceeded to neurological rehabilitation with improvement of right hemiparesis and gradual but progressive recovery of left hemiparesis. Repeat CT brain at 4 weeks did not show any infarct progression (Figure 1c).
Discussion
CGE is an uncommon, but potentially fatal, event that occurs as a consequence of air entry into the vasculature. Surgery, trauma, vascular interventions, and barotrauma from mechanical ventilation are common causes.
The diagnosis of CO2 CGE in this case study was established based on a number of factors. First, there are published cases of CO2 emboli resulting from thoracoscopic and laparoscopic surgery where gas insufflation is utilized and there is a vessel injury. This allows a portal of entry into the circulation [5]. In this case, it is most likely that the pulmonary vein injury provided an entry point for CO2 into the arterial circulation with subsequent embolization to the intracranial vasculature.
Second, the patient was in the left lateral position with head up 45 degrees during the operation, which was conducive to gas emboli favoring mainly the superior regions of the right cerebral hemisphere. Both hemispheres were involved, which is suggestive of a central source.
Third, gas in the brain can be reabsorbed rapidly, particularly CO2 which dissolves readily in the blood [6]. This is consistent with literature that recognizes the poor sensitivity and specificity of acute CT findings in identifying CGE [7]. The acute phase edema may be associated with CO2 induced vasodilation. It is likely that the patient’s relatively rapid recovery was due to rapid reperfusion of the at-risk brain tissue, and relatively small volume of infarcted tissue, due to rapid CO2 dissolution. We recognize that another potential source of CGE exists in this patient in the form of paradoxical embolism from central venous access and the presence of a PFO; this was clinically considerably less likely given the uncomplicated central venous catheter insertion, small size PFO, and stable intra-operative course.
Initial supportive measures for CGE are focused on ensuring airway protection, adequate ventilation, oxygenation, and hemodynamic stability. High FiO2 therapy should be administered. The supplemental oxygen increases the partial pressure of oxygen and decreases the partial pressure of nitrogen in the blood. This causes diffusion of nitrogen from inside the air bubble (which has a high nitrogen content) into the blood (which has a low nitrogen concentration), which reduces bubble size and accelerates bubble resorption [8]. In severe cases of CGE with associated hemodynamic instability, HBOT is the mainstay of treatment. The mechanism is similar to high FiO2 therapy but on a much greater magnitude. In this case, the absence of residual intracranial gas in a hemodynamically stable patient was felt to outweigh any potential benefit of a limited resource unavailable at our institution. There is also a recognized risk of death during transfer to a suitable institution, which needs to be considered in all cases.
Risk factors for death or poor prognosis include cardiac arrest, increasing age, focal neurological deficits at ICU admissions, acute kidney failure, prolonged mechanical ventilation for more than 5 days, and gyriform air on brain imaging [9,10].
Conclusion
In patients who have acute neurological manifestations following a medical procedure, an acute cerebrovascular event must be considered. Rapid access to neuro-diagnostic imaging is paramount in order to initiate correct supportive and/or definitive therapy. As acute CT findings can be non-specific, MRI with diffusion weighted sequences is essential to confirm the diagnosis of acute embolic infarction and help elucidate the pathophysiological mechanism involved [5]. Initial supportive therapy for CGE should be commenced while investigations for more common thromboembolic sources are performed. Transfer to a center that provides HBOT should be considered in severe cases.

