Background
During the recent COVID-19 pandemic, there has been growing evidence that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) affects not only the lung but may also be systemic [1].
In a recent retrospective study [2], pericardial effusion was present in 19.6% of COVID-19 positive patients, but there is no evidence of SARS-CoV-2 RNA in pericardial effusion during the acute phase of COVID-19 infection.
Case Presentation
A 58-year-old man with a medical history of Paget's disease and thymectomy (2018) was admitted to the emergency department of a spoke-center for generalized weakness and continuous abdominal pain.
Four days earlier, he had been discharged from the cardiology department with non-ST elevation myocardial infarction. At that time, two nasopharyngeal swabs had been negative for COVID-19 in two consecutive days and the peak high-sensitivity cardiac troponin I (hs-cTnI) had been 1,653 ng/l (normal value 0-47 ng/l).
A coronary angiography had shown mild atherosclerosis without significant obstructive coronary disease. An electrocardiogram (ECG) had revealed non-specific left ventricular repolarization abnormalities and a left ventricular inferolateral wall hypokinesia had been described on transthoracic echocardiogram. No heart valve dysfunction or pericardial effusion had been reported at that time.
His medications included metoprolol 25 mg twice daily, ramipril 2.5 mg/day, atorvastatin 80 mg/day, and acetylsalicylic acid 100 mg/day.
On physical examination, he appeared pale, afebrile (36.8°C), and normotensive (125/80 mm Hg), his external jugular veins were distended, the heart rate was 102 beats per minutes, the respiratory rate (15 per minutes) and the peripheral oxygen saturation (95%) were normal without any oxygen supplement.
An ECG showed sinus tachycardia, low voltage in the limb leads and alternation of QRS complex amplitude, clearer in the precordial leads (Figure 1).
Blood test revealed raised hs-cTnI (149 ng/l, normal value 0-47 ng/l), N-terminal pro-brain natriuretic peptide (2.112 pg/ml, normal value 0-125 pg/ml), C- reactive protein (7.02 mg/dl, normal value 0-0.6), lactate dehydrogenase (842 U/l, normal value 0-240), aspartate amino transferase (415 U/l, normal value 0-40), alanine amino transferase (404 U/l, normal value 0-40), international normalized ratio (1.8), and total bilirubin (1.98 mg/dl, normal value 0.2-1.2). The blood panel showed increased white blood cell (18.70 × 10^3/mmc, normal value 4-10) and neutrophil (87.9%, normal value 40-75) with decreased lymphocyte counts (6.5%, normal value 20-45) and a slightly reduced value of hemoglobin (11.8 g/dl, normal value 12.5-17).
Nasopharyngeal swab for SARS-CoV-2 RNA was obtained and tested positive by real-time reverse-transcriptase polymerase chain reaction (RT-PCR) assay (Abbott RealTime SARS-CoV-2 kit on the Abbott m 2,000 integrated diagnostic system - declared LLOD:100 RNA copies/ml; Abbott, Lake Bluff, IL).
Owing to the initial presentation of abdominal pain and abnormal liver function tests, we performed a chest and abdominal computed tomography (CT) which revealed massive pericardial (Figure 2, panel A) and moderate pleural and peritoneal effusion.
An emergency echocardiogram showed a large and compartmentalized pericardial effusion (predominantly posterior and lateral) with signs of early diastolic collapse of atria and basal-mid segments of the ventricles free wall, consistent with cardiac tamponade (Figure 2, panels B and C) (Video 1).
Moreover, the echocardiogram demonstrated adherences between the cardiac apex and parietal pericardium layer (Figure 2, panel D) (Video 2).
We immediately transferred the patient to the hubcenter for urgent echo-assisted pericardiocentesis performed in a left parasternal approach while the patient was in left lateral decubitus position.
The procedure was performed in a dedicated negative-pressure room and all the professionals wore the recommended personal protective equipment.
An amount of 1,400 ml of sero-hemorrhagic fluid (hemoglobin concentration = 5.3 g/dl) consistent with exudate (protein 3.8 g/l, cholesterol 62 mg/dl, glucose 96 mg/dl, leukocytes 1,470 /mcl) was removed. The cytological analysis excluded the presence of malignant cells.
Thus, we tested and confirmed COVID-19 RNA in the pericardial liquid by RT-PCR assay (CT value 34.11; Abbott RealTime SARS-CoV-2 kit on the Abbott m 2,000 integrated diagnostic system; Abbott, Lake Bluff, IL). To exclude other causes of pericardial effusion, we documented the absence of Enterovirus RNA in a fecal sample.
Despite initial therapy with prednisone 25 mg daily, enoxaparin 4,000 IU daily, hydroxychloroquine 200 mg twice daily, and azithromycin 500 mg daily, the pericardial fluid remained abundant (250-300 ml per day with sero-hematic features) for several days. After 5 days, colchicine (0.5 mg twice daily) was added, followed by gradual pericardial fluid reduction. The drainage catheter was removed 16 days later without complications. An echocardiogram was repeated prior to discharge and showed no pericardial effusion.
Discussion
Several authors described cardiac complications of COVID-19 including cardiac tamponade [3-5], myopericarditis [6], and acute heart failure [7,8]; however, a viral cardiotropism has not been demonstrated [7].
Only one paper described the SARS-CoV-2 RNA detection in the pericardial fluid and the authors postulated the hypothesis that the pericardial fluid could be a virus persistence site [5].
We present the SARS-CoV-2 RNA detection in the pericardial fluid of a patient in an acute phase of COVID-19 infection.
Different mechanisms may help explain the viral genome detection in the pericardial effusion.
A small percentage of blood samples with positive RT-PCR test results [1] suggested that a transient viraemia could lead to the SARS-CoV-2 RNA distribution in the pericardial effusion. Another hypothesis could be the macrophage migration from nearby affected tissues (our patient had also bilateral pleural effusion), as suggested by Tavazzi et al. [7].
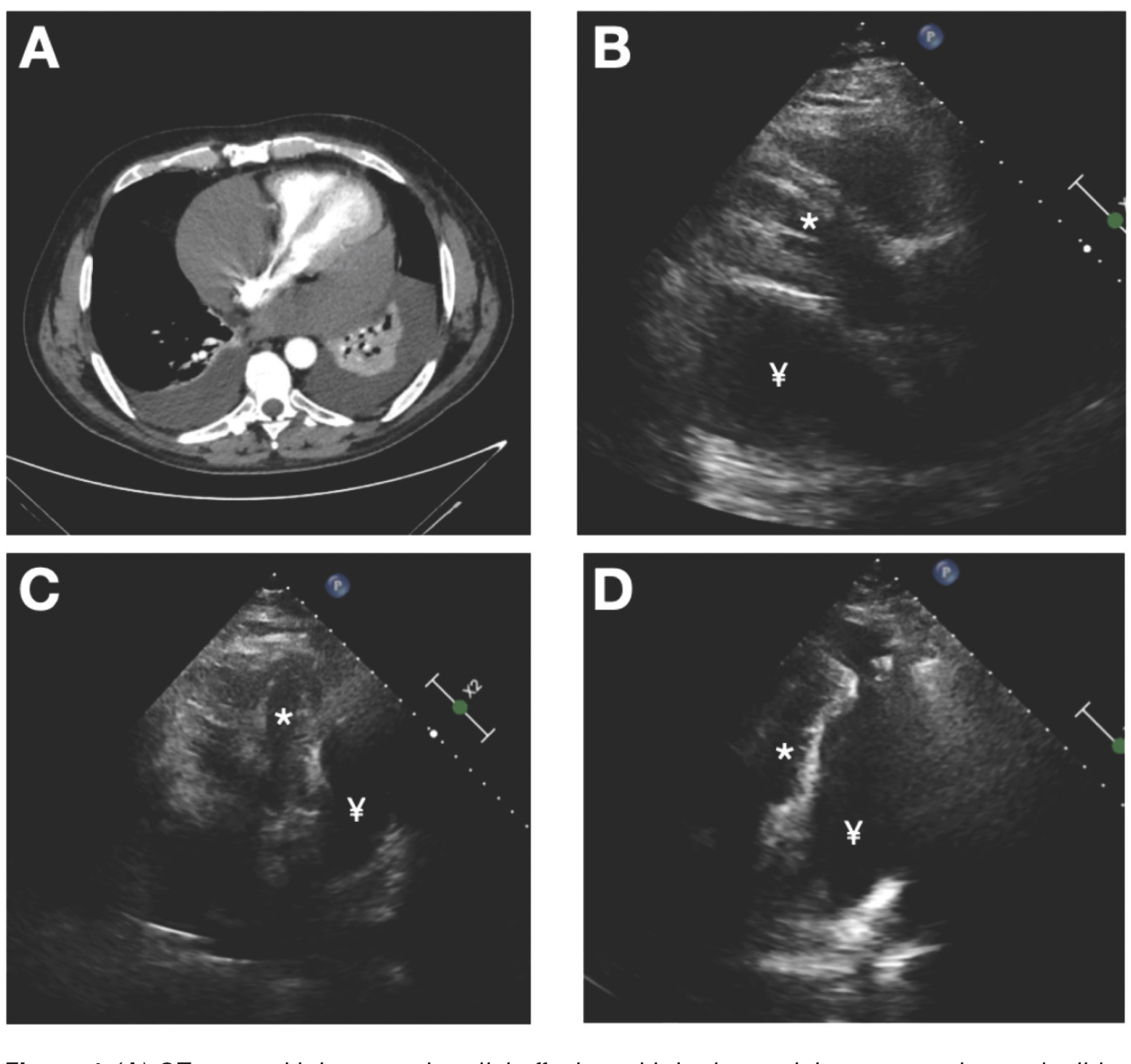
(A) CT scan with large pericardial effusion with both ventricle compression and mild bilateral pleural effusion. (B) Parasternal echocardiographic view with left ventricle compression. (C) Apical 4 chambers view with predominant posterior and lateral diffusion of the pericardial liquid. (D) Adherences between cardiac apex and parietal pericardial layer (arrow) with subsequent distortion of basal and mid left ventricular wall. Legend: * = left ventricle; ¥ = pericardial effusion.
However, although the viral genome in organs and tissues other than the lung has been demonstrated [1,7], the mechanisms underlying the cardiac involvement in COVID-19 patients remain only partially understood.
Regarding cardiovascular complications in COVID-19, Varga et al. [9] recently suggested that SARS CoV-2 infection facilitates endotheliitis as a direct viral action and the consequences of the host inflammatory response with impaired microcirculatory function.
Similarly to what happens in other viral infections, it is plausible that COVID-19 triggers an exaggerated systemic inflammatory response leading to acute pericarditis and increased production of pericardial fluid.
The presence of polyserositis (concomitant moderate pleural and peritoneal effusion detected by CT scan) in our patient supports this hypothesis.
Viral infection is a common cause of acute pericarditis, typically associated with exudative effusion [10] and if the pericardial fluid is rapidly accumulating its distribution is usually circumferential.
Although our case describes an acute cardiac involvement with rapid expansion of the effusion in a few days, similar to what previously described [4], it presents some features that are usually typical of chronic pericardial effusion, such as adherences and located distribution.
Main limitations of this report are:
a) the incomplete exclusion of other viral causes of pericarditis, except for the Enterovirus; b) the sero-hem-orrhagic feature of the fluid could be the consequence of the anti-platelet and anti-coagulant therapy that the patient assumed for the previous NSTEMI diagnosis. The negative cytologic analysis reasonably excluded a malignant etiology of the effusion.
It cannot be excluded that the sero-hemorrhagic fluid may be the direct consequence of cardiac virus localization, since the pericardial effusion had similar features in all previous published cases of cardiac tamponade [3-5].
Lastly, although in left lateral decubitus, an echo assisted parasternal approach pericardiocentesis was an effective and safe procedure.
Finally, colchicine treatment was associated with the therapeutic success of this case.
The question about SARS-CoV-2 RNA detection in the pericardial effusion and its role in the peculiar clinical and echocardiographic presentation is still an unsolved issue.
Conclusion
SARS-CoV-2 RNA can be detected in pericardial effusion during the acute phase of COVID-19 infection.
Concomitant presence of pleural and peritoneal effusion could confirm the hypothesis of an exaggerated systemic inflammatory response triggered by the virus.
What is new?
SARS-COV-2 RNA has been found in many tissues and fluids but only once in pericardial effusion after cardiac surgery and not in the acute phase of COVID-19. At the best of authors’ knowledge, other three cases of cardiac tamponade during COVID-19 have been described but no information about the RNA detection in the pericardial fluid was available. The authors present a case of cardiac tamponade secondary to a sero-hemorrhagic effusion with SARS-COV-2 RNA detection.


