Background
Pneumothoraces are either primary (occurring without any evident lung disease) or secondary (with pre-existing lung diseases, or in those over the age of 50 years and with a significant smoking history) [1]. The management of either is subject to much debate in the literature. Treatment options include observation, admittance to hospital oxygen therapy, and/or intervention with either needle aspiration or a small-bore chest drain, and/or ambulatory management with portable devices [1-3].
Pleural medicine is increasingly recognized as a subspecialty of respiratory medicine [4].Guidance in the United Kingdom suggests that every hospital requires cover 24 hours a day, 7 days a week to provide a safe, effective, and timely pleural service [4,5]. Complications from pleural interventions are rare, but carry significant morbidity and mortality [6].
Case Presentation
Patient A is a 78-year-old male patient with chronic obstructive pulmonary disease (COPD) (ratio of the forced expiratory volume in the first one second to the forced vital capacity - FEV1/FVC 0.93/2.43 ratio 38%, FEV1 35%), who initially presented with hemoptysis. He was diagnosed with a right lower lobe and hilar squamous cell lung cancer staged at T4N2MX through endobronchial ultrasound sampling. He received four cycles of gemcitabine and carboplatin in August 2019 with palliative intent. He had received no radiotherapy or immunotherapy. A computed tomogram (CT) staging in September 2019 showed stable appearances. He underwent a repeat staging scan in late February 2020. This showed a right pneumothorax, a small effusion, and a probable tethered lung (Figure 1). The radiographers referred him to Accident and Emergency (A&E) for urgent assessment. In the department, at 8 o’clock in the evening, he was normotensive, had a heart rate of 77 beats per minute, a respiratory rate of 18 breaths per minute, and oxygen saturations of 91% on room air. He described 5 days of gradually increasing breathlessness but had attributed this to a possible non-infective exacerbation of his COPD. There was no chest pain. On examination, he had reduced air entry in the right hemithorax and his trachea was central. He was talking in full sentences and looked well.
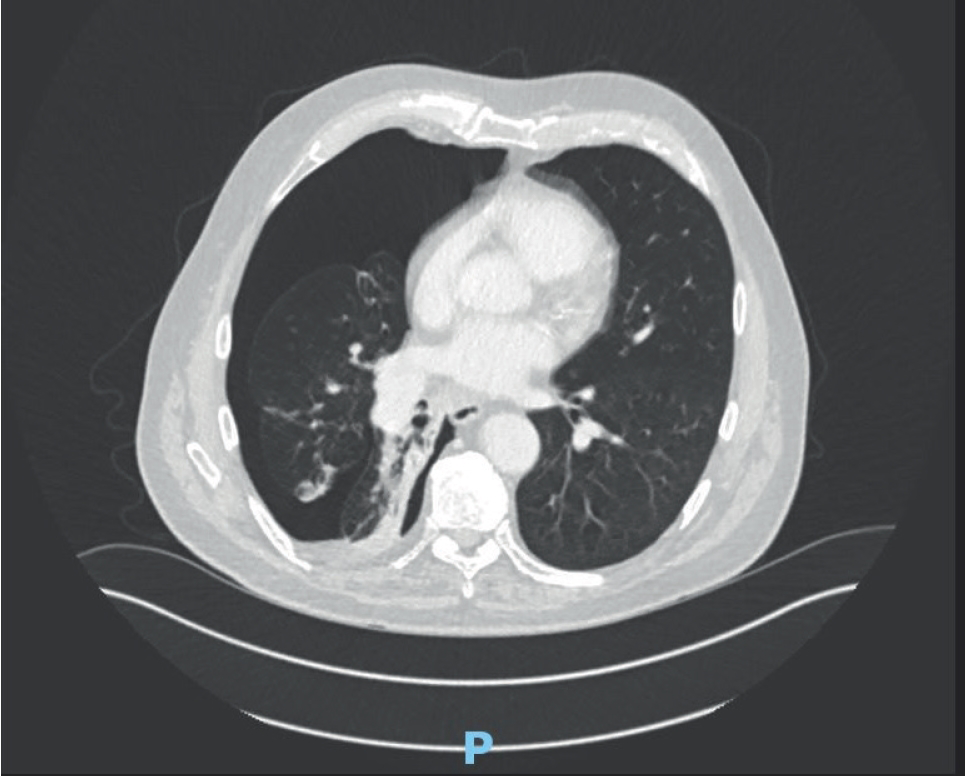
Lung window of CT scan showing right pneumothorax, a small effusion and a probable tethered lung.
Patient B is an 81-year-old male patient with COPD (FEV1/FVC 1.02/2.43 ratio 42%, FEV1 35%), ischemic heart disease, diet-controlled type 2 diabetes, hypertensions, and T4N2M1a small cell lung cancer (right lower lobe with pleural involvement) diagnosed via endobronchial ultrasound sampling in January 2020. He was commenced with carboplatin and etoposide chemotherapy late January 2020 with palliative intent and attended for a routine staging CT scan in late February 2020. He had received no radiotherapy or immunotherapy. Figure 2 shows the interval development of a hydropneumothorax. The right lower lobe mass was unchanged and there was a mild increase in the subcarinal lymphadenopathy. The radiographers referred the patient to A&E for further assessment. In the department, at 4 o’clock in the afternoon, he was normotensive, had a heart rate of 67 beats per minute, a respiratory rate of 17 breaths per minute, and oxygen saturations of 95% on room air. He also described a few days history of increasing breathlessness but no chest pain. On examination, he had normal air entry bilaterally and his trachea was central. He also was talking in full sentences and looked well.
Patient A was reviewed by the A&E physicians and was consented for a small bore 12 French gauge Rocket® chest drain. This was placed by a trainee, under consultant supervision, at 9 o’clock in the evening and was uneventful. The drain was secured by adhesive dressings and connected to an underwater bottle. Bubbling and swinging were observed. The patient felt slightly better. Figure 3 shows a chest radiograph (CXR) with the drain in situ and the pneumothorax reducing in size. Oxygen saturations remained at 91% on air and the patient was admitted overnight. The next radiograph was performed the next day (Figure 4) which shows distal migration of the drain and development of surgical emphysema. Before any action could be taken, the drain fell out. Figure 5 shows the next CXR which is almost a complete resolution of the pneumothorax but progressive surgical emphysema.
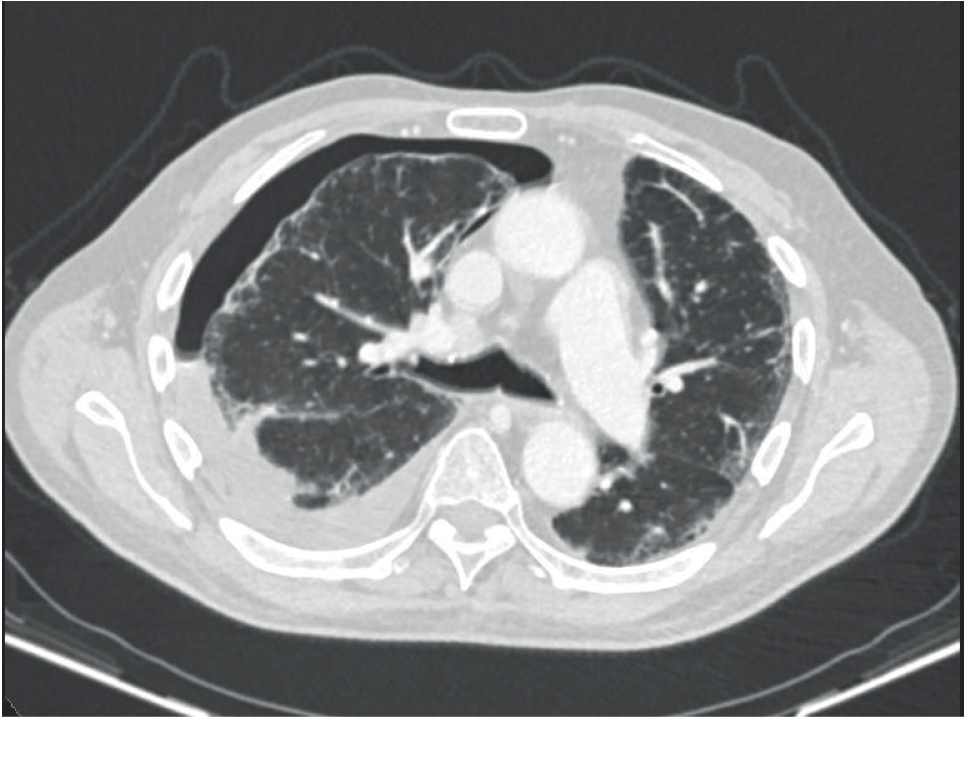
Lung window of CT scan showing right pneumothorax and a pleural effusion (hydropneumothorax).
Patient B was discussed with a member of the pleural team and it was decided that no intervention was required given the clinical and radiological picture.
On the day patient’s A drain fell out, specialist pleural advice was sought. Observations had remained normal and symptoms were not worsening. It was hence decided not to intervene. He was discharged to come back to Medical Ambulatory Care (MAC) in 48 hours and given open access to MAC to present if he felt more unwell. At his planned review, he showed continued clinical and radiological improvement (Figure 6) and was discharged from the inpatient service back to oncology with appropriate safety nets in place. Total inpatient length of stay was 5 days.
Patient B was booked to come for review on MAC on the same day as patient A. His symptoms had improved; his observations had remained normal; and his repeat CXR (Figure 7) showed improvement. As such, there was still no need to intervene and he was thus also discharged back to oncology with safety netting.
Discussion
Spontaneous pneumothorax has a yearly incidence of 14.1 admissions per 100,000 population aged more than 15 years. The incidence is higher in men (20.8 per 100,000) than women (7.6 per 100,000). From 1968 to 2016, age-standardized admission-based rates in England was on the rise for both men [annual percentage change (APC) 0.79, 95% CI 0.66-0.92; p < 0.001] and women (APC 1.54, 95% CI 1.25-1.84; p < 0.001). Pneumothorax also has a bimodal distribution with one peak in patients aged 15-34 years and another in those aged more than 55 years [7].
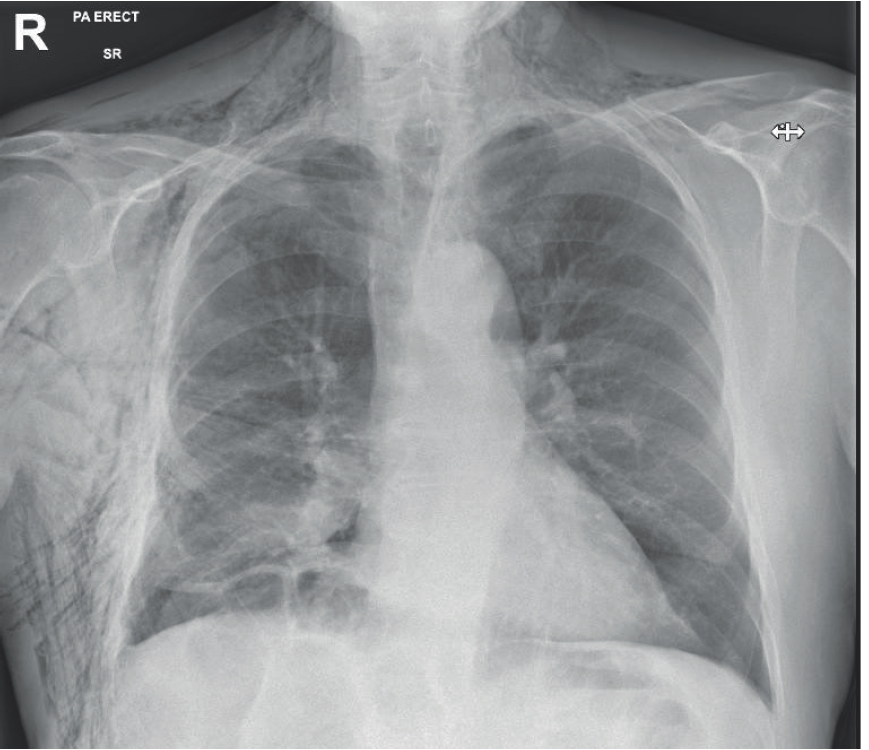
CXR showing almost a complete resolution of the pneumothorax but progressive surgical emphysema
Pneumothorax is air in the pleural space and is traditionally defined as primary spontaneous pneumothorax (PSP) or secondary spontaneous pneumothorax (SSP) as described earlier, but the definitions are not absolute. There is increasing evidence that the lungs in patients with PSP are not normal: findings of increased apical pleural porosity, significant blebs, emphysema-like changes, and generalized inflammation have been described and play a role in the etiology of PSP. Smoking is the biggest risk factor, causing a 12% risk of any type of pneumothorax in a healthy male, as compared to approximately 0.1% in a non-smoking male. However, while the commonest underlying lung condition in SSP is smoking-related COPD with emphysema in the Western world, SSP has been described in patients with bullous disease, cystic fibrosis, tuberculosis, lung cancer, HIV-associated pneumocystis jirovecii pneumonia, lymphangioleiomyomatosis and histiocytosis X [7,8].
Pneumothoraces can be treated conservatively with therapeutic aspiration or with chest drain insertion. The exact modality used can depend on the patient’s symptoms, choice, the size of the pneumothorax, as well as the treating physician’s capability and resources [1]. A 2008 National Patient Safety Agency Report in the United Kingdom recorded 27 cases of mortality or serious morbidity caused by chest drains [1,5,6]. The 2015 British Thoracic Society (BTS) pleural procedure audit found that 40% of drains for pneumothoraces were inserted out of hours and that 9.2% of drains fell out, among other complications [6].
The question is why was the drain in Patient A? Feedback collected from the A&E physicians showed that reasoning was based on the BTS 2010 guidelines. The resultant ventilation-perfusion mismatch in a pneumothorax is less likely to be tolerated in patients with already existent underlying lung disease and the air leak is less likely to settle spontaneously. The guidance also suggests that all patients with SSP should be admitted to the hospital for at least 24 hours and should receive supplemental oxygen in line with their oxygen guidelines. This is based on D level evidence, the lowest level of the Scottish Intercollegiate Guideline Network framework of rating recommendations. The BTS guidelines are currently being updated and due to be published in late 2020. The current guidance also suggests that most patients will require intervention in the form of a small-bore chest drain (B level evidence) [1]. There is evidence to support that surgical procedures done out of hours have increased complication rates [9]. This has not been replicated in pleural medicine but makes common sense and is advocated in the BTS guidance that the vast majority of pleural interventions do not need to be performed as an emergency and in the absence of cardio-respiratory compromise, should not be performed overnight [10].
There is some evidence to support observation in SSP, although the numbers in the retrospective studies were small and recurrence rates and selection bias into the studies were high [11]. The drain was not sutured in despite local and national guidance suggesting that because of the perception that the dressing is enough to keep a drain in place [1]. A retrospective analysis has also showed that the routine use of suturing reduces fall out rates [12].
Both patients A and B had normal observations and were referred from the radiology department. Both presented to A&E and were found to have mild increase in dyspnea. They had good family support. Patient A had a small-bore chest drain inserted with the complications described earlier and patient B was simply observed. We are of the firm opinion that patient A should have been admitted and observed in a place of safety, rather than have a chest drain placed out of hours in A&E by a supervised doctor in training, and without stitches to hold it in place. While the pneumothorax is bigger, the symptoms were minimal and there was no cardio-respiratory compromise. A point must be noted that the 2010 BTS pleural disease guidance suggests to measure the pneumothorax size on a CXR at the level of the hilum, but that patient A only had a CT scan and that the treating team were more concerned with his relatively low oxygen saturations, which were in hindsight, not out of keeping with his known severe obstructive lung disease. Inserting a chest drain under supervision is not normally an issue and is important for training, but junior doctors lack confidence and competence when it comes to chest drains [13]. There is a growing trend to use ambulatory devices in primary and secondary pneumothoraces [14,15] and although the Randomised Ambulatory Management of Primary Pneumothorax trial has shown safety and efficacy in primary pneumothoraces [16], the Hi-Spec trial has not been published yet to show safety in secondary pneumothoraces. Conservative management of primary pneumothoraces has also been suggested, but no clear evidence exists [17].
Conclusion
Secondary pneumothoraces occur in patients with pre-existing lung disease. Pleural interventions should be done at the right time, for the right patient, in the right setting - the vast majority should not be done overnight. Chest drains should be sutured to reduce fall out rates but some secondary pneumothoraces can be observed as long as adequate safety nets are in place.





