Background
Superior mesenteric artery syndrome (SMAS), also known as Wilkie’s syndrome, is a rare cause of upper intestinal obstruction. It happens due to the partial or complete extrinsic compression of the third portion of the duodenum between the superior mesenteric artery and the aorta [1–3]. SMAS is caused by the loss of the retroperitoneal fat pad resulting in narrowing of the aortomesenteric angle [2]. This syndrome is more often diagnosed in young individuals, after acute weight loss [1,4,5].
The surgical therapy is indicated for the cases of failed clinical treatment [1,6]. There are two surgical procedures described in the literature: duodenojejunostomy and release of the Treitz’s ligament (Strong’s procedure). Strong’s procedure consists of division of the Treitz’s ligament, thereby releasing the duodenum distal to the superior mesenteric artery by dissecting the surrounding peritoneum. The duodenojejunostomy consists of anastomosing the third portion of the duodenum to a jejunal loop to bypass the obstruction [7].
The current case report describes in detail the use of laparoscopic duodenojejunostomy combined with Treitz’s ligament release for the treatment of SMAS in a young male. There is a lack of literature about the combination of both techniques, and few articles have reported this combination, however, with a lack of technical detail.
Case Presentation
A 19-year-old male was admitted to the emergency department with intense, diffuse abdominal pain, mainly in the upper abdomen, for 3 days. Pain was colicky in nature, worsened after food intake, and associated with severe nausea and vomiting. He had a history of intense physical training and acute weight loss after joining the Brazilian Army recently. Symptoms were recurrent since he was 10 years old. He had several hospital admissions and was diagnosed with acute gastroenteritis.
Routine blood tests were performed, and no infection or significant changes were seen. A computed tomography (CT) of the abdomen and pelvis showed a slow progression of oral contrast medium in transition beyond the third portion of the duodenum. Reduced aortomesenteric distance (0.5 cm) causing compression between the third and fourth portions of the duodenum was also observed (Figure 1). In addition, angiographic magnetic resonance imaging also showed a shortened distance between the superior mesenteric artery and the aorta (4 mm). The angle between the two arteries was of approximately 18° (Figure 2).
The patient’s food intake was suspended, and a nasogastric tube was placed for biliary drainage, relieving his symptoms. The preoperative nutritional support was provided through parenteral nutrition. Surgical treatment was indicated due to the failure of conservative treatment and the risk of recurrence of symptoms with an intense military routine.
The patient was positioned on the table with his lower and upper limbs open. The laparoscopy tower was placed next to the patient’s head, on the right-hand side. The surgeon stood between patients’ lower limbs. Two assistants helped the surgeon, and the instrument table was placed on the patient’s left-hand side (Figure 3).
After the induction of general anesthesia, pneumoperitoneum was performed with a Veress needle up to the pressure of 12 mmHg. A 10-mm trocar was inserted at the patient’s umbilicus for the camera introduction. After exploring the abdominal cavity, the other two trocars were inserted under direct view: the first one was a 5-mm trocar inserted in the patient’s right flank, whereas the other one is a 12-mm trocar inserted in his left flank (Figure 3).
Transverse colon was lifted to visualize retroperitoneal duodenum, pancreas, and mesenteric vessels. The peritoneum was dissected on the left of the middle colic artery using ultrasonic scissors to expose the head of the pancreas and the mesenteric vessels and also to release the third duodenal portion to enable a broader, tension-free, and safe anastomosis.
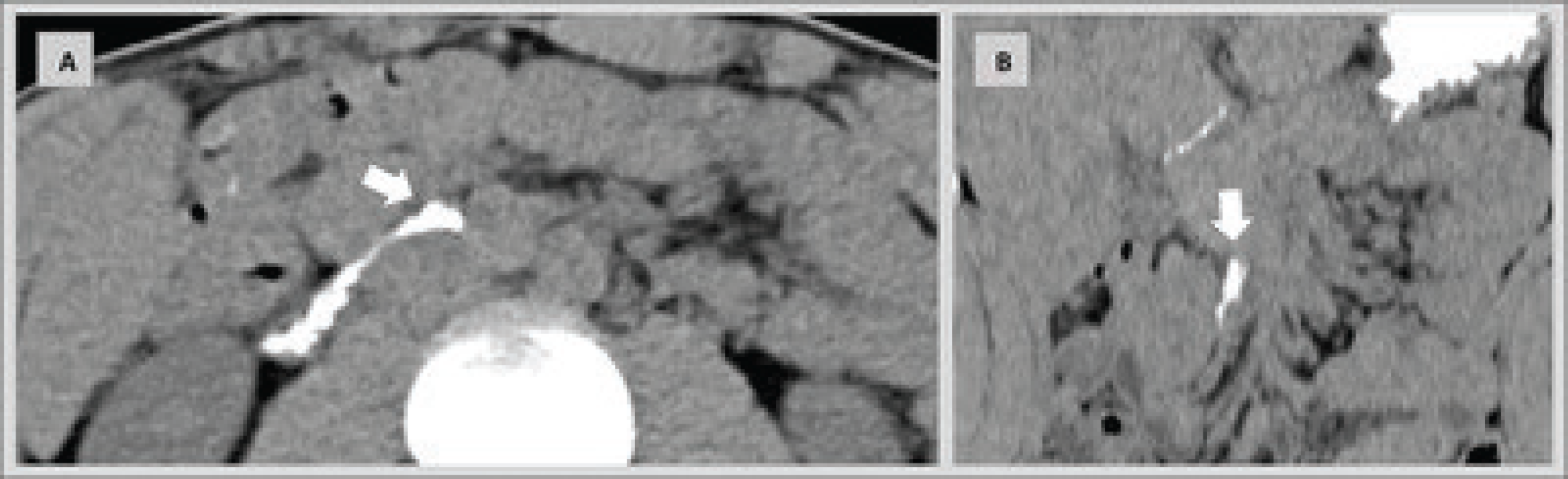
CT of the abdomen: slow progression of the oral contrast (arrows) beyond the third portion of the duodenum due to superior mesenteric artery compression; viewed in axial (A) and coronal (B) sections.
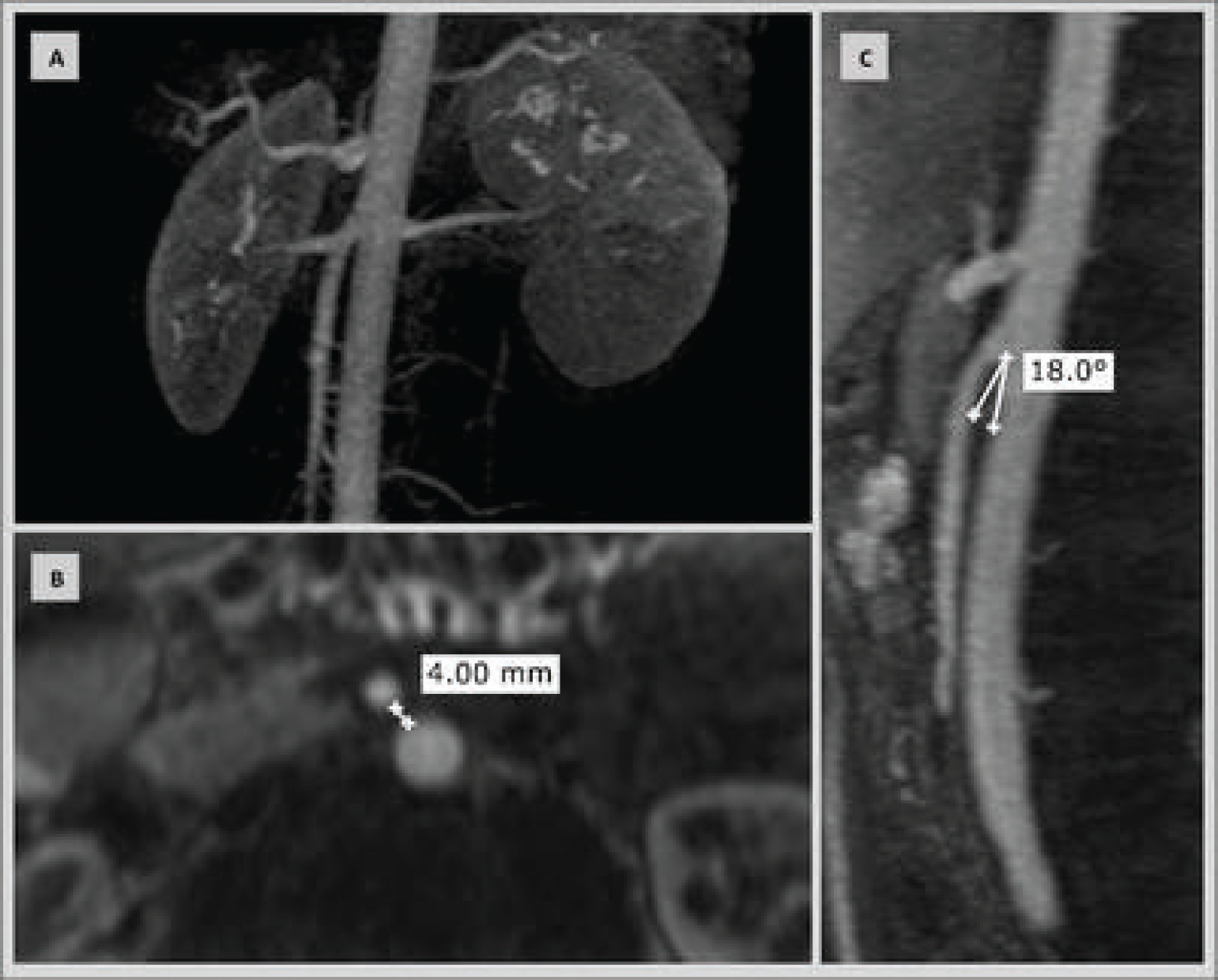
Angioresonance of the abdomen. (A) Relation between superior mesenteric artery and aorta in 3D reconstruction. (B) Distance between the superior mesenteric artery and the aorta in the axial section. (C) Angle between the superior mesenteric artery and the aorta in the sagittal section.
The Treitz’s ligament was divided, and the duodenum was released by dissecting the surrounding peritoneum to have it distally mobilized to the superior mesenteric artery. A jejunal loop located approximately 15 cm from the Treitz’s ligament was positioned next to the third portion of the duodenum for anastomosis. Duodenal-jejunal anastomosis was performed using two 45-mm loads of endoscopic stapler to enable a wider bypass. The stapling gap was closed with PDS 3-0 suture (Figure 4). Anastomosis patency was tested with methylene blue administrated through a nasogastric tube.
Trocars were removed under vision after hemostasis review. The 10- and 12-mm trocar apertures in the deep fascia and skin were sutured. The procedure was completed with no complications, and no abdominal drain was placed.
The surgery lasted for 54 minutes, and there was no considerable bleeding. The nasogastric tube was removed during surgery. Liquid food was initiated 2 days postoperatively, and solid food was started 2 days later. The patient recovered well, with no complications, and was discharged 5 days later.
In postoperative follow-ups, the patient remained well after more than 90 days after surgery, presented good tolerance to the food intake without vomiting or new pain episodes, and also added 4 kg of weight (6%).
Discussion
SMAS manifests itself with non-specific clinical features, easily misinterpreted as common conditions such as acute gastroenteritis, dyspeptic syndrome, gastroesophageal reflux disease, and some causes of upper intestinal obstruction; therefore, the diagnosis is often made after the other causes of symptoms have been ruled out. Symptoms can be acute or insidious and depend on the upper intestinal obstruction level [1]. Mild obstruction is associated with postprandial epigastric pain and early satiety, whereas intense obstruction is associated with nausea, vomiting, eructation, and weight loss. Therefore, most of the patients have a low body mass index [1]. The diagnosis of this syndrome can be confirmed through imaging methods such as angiotomography, magnetic resonance angiography, and arteriography, which is performed only when less invasive examinations are not conclusive [5]. There is no consensus in the literature about reference values associated with the diagnostic criteria of this syndrome. It is estimated that the most sensitive measurements for such diagnosis are related to an aortomesenteric angle smaller than, or equal to, 25°, as well as to distance shorter than, or equal to, 8 mm [3–6].
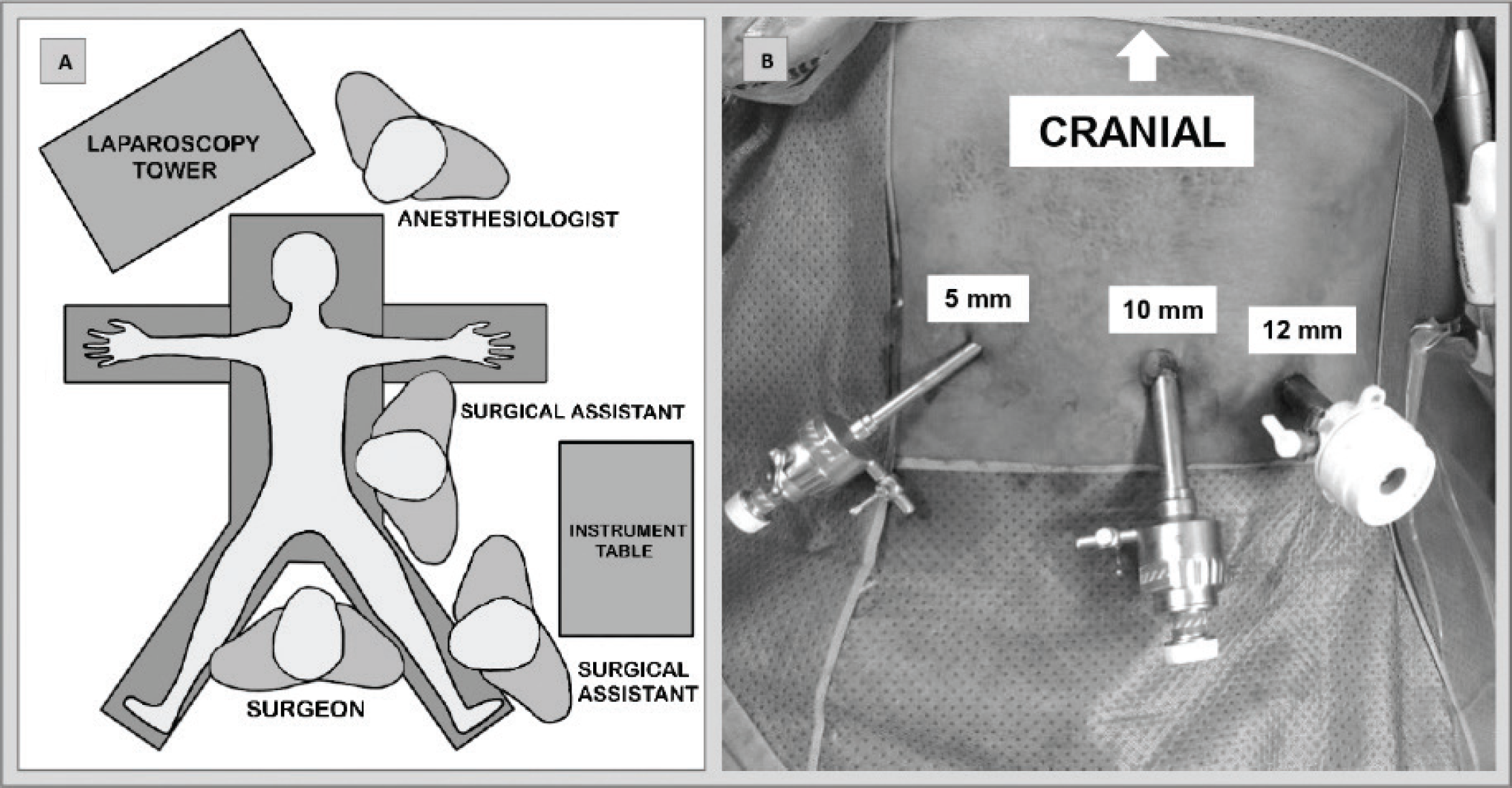
Positioning of the patient and trocars. (A) Positioning of the patient, surgical team, and video monitor in the operating room. (B) Positioning and size of introduced trocars.
The initial treatment of this syndrome is conservative, based on supportive measures such as total parenteral nutrition, nasogastric tube placement, intravenous hydration, and electrolytes disorder correction [1]. If possible, diet adaptation to a small amount of food in short intervals should be attempted, as well as a change to left lateral decubitus position. Conservative treatment has reasonable prospects for success in patients presenting acute conditions, reducing the need for a surgical treatment from 70% to 14% [8]. However, when it comes to chronic patients, conservative measures are often associated with prolonged therapy and low success rates [3].
Surgical treatment is often indicated in SMAS after conservative treatment failure [1,6], as well as in chronic cases with progressive weight loss and duodenal dilation. The most common techniques used are Strong's procedure and duodenojejunostomy-both of which can be performed through an open or laparoscopic approach. However, given the rare nature of this condition, the literature lacks the comparative studies focused on the benefits of either of these two techniques, and also, there are no established protocols favoring the use of one procedure over the other.
Laparoscopic duodenojejunostomy appears to be a safe and effective procedure [7]. However, the literature lacks a deeper analysis in the long-term outcomes and incidence rate of complications such as late obstruction, internal hernia, blind loop syndrome, bacterial overgrowth, and recurrence of the syndrome. On the other hand, the advantage of Strong’s procedure lies in the fact that it does not require anastomosis and can also be performed through laparoscopy. However, the recurrence rate associated with this technique can reach 25% [9].
In this case, we combined both techniques to reduce the rate of recurrence and long-term complications and increase anastomosis safety.
A wide duodenal-jejunal anastomosis is a fundamental step for a successful surgery [10]. Distal duodenum mobilization to the superior mesenteric artery emergence in order to undo the point of obstruction of the loop, to be excluded, is expected to reduce the relapse rate. The anastomosis was performed between the jejunum loop (15 cm away from the Treitz’s ligament) and the third portion of the duodenum, which was released from the surrounding peritoneum. This procedure reduced the tension and, consequently, increased anastomosis safety.
Blind loop syndrome is one of the possible longterm complications due to intestinal loop exclusion from the gastrointestinal transit after the third portion of the duodenum (obstruction point). It might increase the risk of bacterial overgrowth, mainly in the duodenal region [11].
In addition, the combination of both techniques was preferred due to the patient’s strenuous work activity (causing abrupt weight loss), a fact that increases the risk of recurrence of the syndrome. Moreover, the laparoscopic approach was chosen based on the surgeon’s experience because the patient was young and thin [8,10].
Conclusion
The combination of the techniques did not compromise surgery time or costs. The patient recovered well and remained hospitalized for a short period of time. The surgery fully improved the patient’s abdominal pain and food tolerance.
It may be concluded that the combination of both techniques is safe, does not compromise surgery time or costs of the procedure, and enables positive outcomes, as long as it is performed by an experienced team. Despite the good initial results, more studies with larger series and longer post-operative follow-up are needed to help establish the evidence for the indication of each technique.
What is new?
The literature describes two techniques (duodenojejunostomy and laparoscopy-based Strong’s procedure) for the treatment of Wilkie's Syndrome, but it lacks the studies focused on comparing benefits deriving from either of these. The herein reported case combined both techniques to reduce relapse rates, increase anastomotic safety, and reduce long-term complications.


