Background
Drug-induced sleep endoscopy (DISE) is a useful method to evaluate multiobstructive sites simulating sleep with drugs, with a greater reliability compared to traditional examination in the waking state [1]. It has become a widely used method in ENT practice, since the effectiveness of surgical interventions in OSA syndrome largely depends on the accurate preoperative identification of the sites of obstruction.
Several pharmacological agents could be used to induce sleep during DISE, such as propofol, dexmedeto-midine and midazolam. In our clinical practice, we use propofol, as a short-acting hypnotic and amnestic agent, widely used for its favorable characteristics, such as the lack of accumulation and the relatively short recovery time [2].
In many cases, different adverse effects have been reported, such as pain on injection, arterial hypotension, bradycardia, including asystole and respiratory depression. It could also set off an epileptic crisis favored by hypoxia in patients with OSA, causing muscle relaxation and decrease in ventilatory drive [3, 4].
Several clinical reports [5, 6] have suggested a drug-induced excitation of the CNS after the administration of propofol, showing generalized tonic–clonic seizures, focal motor seizures, opisthotonos and involuntary movements, collectively termed as seizure-like phenomena (SLP). Electroencephalographic evidence to support such claims is variable; in some cases, where the EEG has been monitored during administration of propofol, the evidence of epileptiform activity has not been accompanied by motor manifestations, while in other cases, the EEG recordings after the events were reported as normal.
In our hospital, we usually carry out DISE in the operating room in the presence of an anesthesiologist, neuroelectrophysiology technician and otolaryngologist. All patients are prepared for polygraphic intraoperative recording, with electroencephalographic (EEG), electrooculographic (EOG), and electromyographic (EMG) monitoring, which allow sleep staging. The cardiorespiratory parameters (oronasal flow, thoracoabdominal movements, respiratory noise, pulse oximetry, heart rate, and ECG monitoring) are carefully checked during the procedure. All patients receive sedation with target-controlled infusion (TDI) of propofol (3 mg/kg/h) to simulate the NREM sleeping phase. The degree of sedation is under continuous monitoring by bispectral index (BIS) monitoring (Apect Medical Systems, Newton, MA). When the BIS is between 50 and 70, generally during snoring, we introduce a 3.4-mm flexible nasopharyngoscope into the nasal cavity to visualize and record the pattern and degree of obstruction.
The complete polygraphic intraoperative recording during DISE, inclusive of brain electrical function monitoring, allowed us to record the proconvulsive effect of propofol, a rare but possible adverse event also in non-epileptic patients. The tonic and clonic phases can be observed and documented on the EEG during the procedure, associated with the seizure movements of the trunk and four limbs.
Case Presentation
A 44-year-old male patient visited the Department of Head and Neck Surgery of our Institution (Otorhinolaryngology, Fondazione Policlinico Agostino Gemelli-IRCCS, Catholic University of Sacred Heart, Rome), reporting night snoring, daytime sleepiness, sporadic cloning of the four limbs during sleep, one episode of nocturia, bilateral nasal respiratory obstruction, and no headache on waking. He also reported previous facial trauma during adolescence. His history was not suggestive of any other comorbid illnesses, including hypertension, diabetes or seizure disorder, no febrile seizures during childhood or familiarity of neurological diseases.
Nocturnal home-based polysomnography confirmed a mild case of OSAS (AHI 5.5 e/h; ODI 2.3 e/h); he totalized 9 on the Epworth Sleepiness Scale and 39 on the sino-nasal outcome test (SNOT-22). The ENT evaluation, with endoscopic video-recording pharyngolaryngoscopy, showed deviation of the nasal septum and hypertrophy of the inferior turbinates, intravelic palatine tonsils (grade 1 using Brodsky’s tonsillar grading), mild hypertrophy of the base of the tongue and normal larynx. Müller’s maneuver induced a collapse of 50% of the airway diameter at the level of the soft palate and 25% at the level of the base of the tongue
The patient was posted for DISE, septoplasty, and turbinectomy. On preanesthetic evaluation, the patient was assessed by the American Society of Anesthesiologists physical status Class I, weighing 82 kg (BMI 24 kg/m2). His Mallampati classification (MPC) grading was 2 and his blood pressure was 130/80 mm Hg. Routine investigations, including hemogram, electrocardiogram (ECG), and chest X-ray (CXR), and coagulation studies were normal.
After securing the intravenous line, we induced sedation by increasing the doses of propofol (maximum dose was 300 mg). Two minutes after the introduction of the flexible nasopharyngoscope in the right nostril, the patient had three close episodes characterized by generalized tonic–clonic seizures of short duration (15–20 seconds), without the loss of sphincter control.
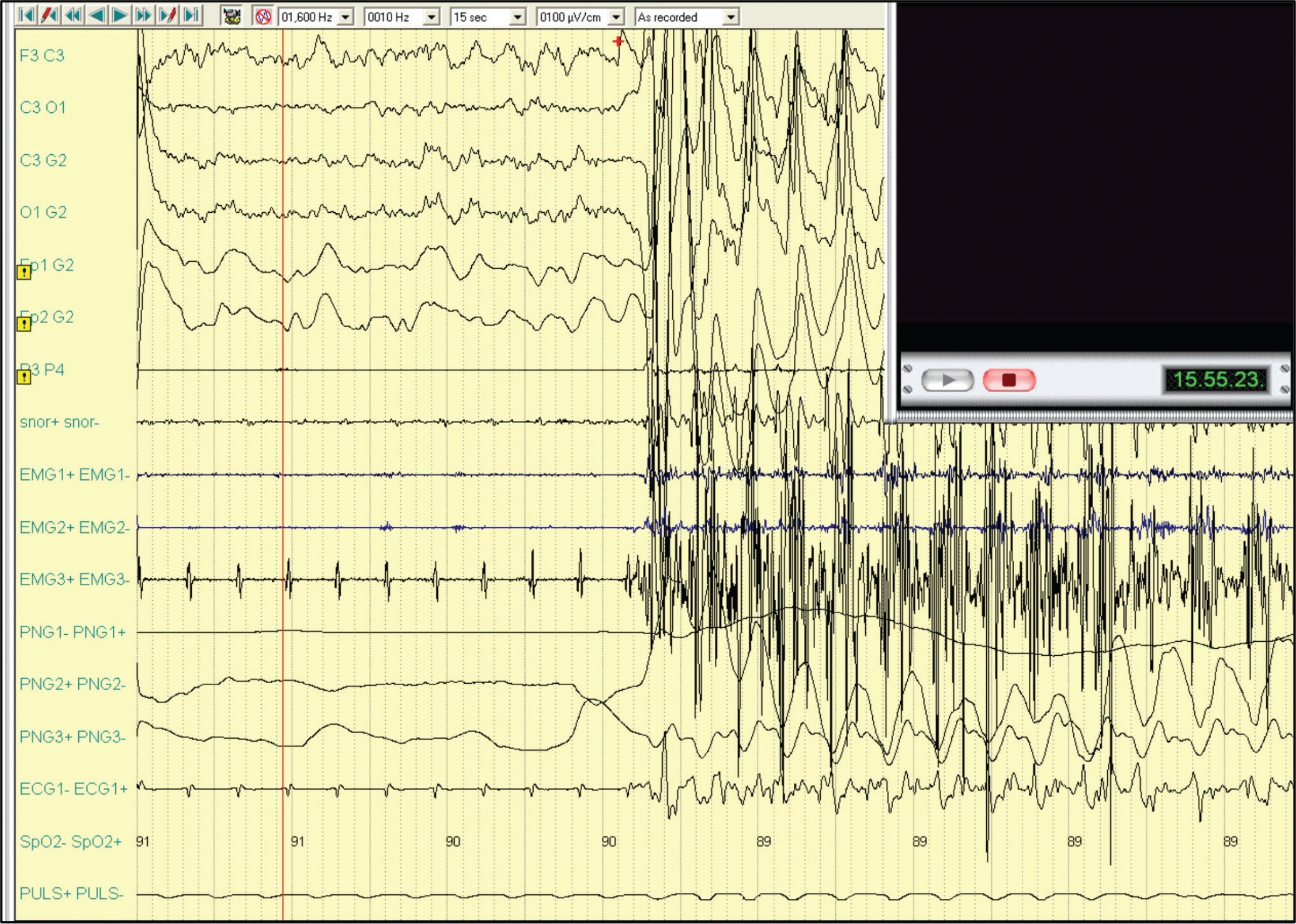
Polygraphic intraoperative recording with electroencephalographic (EEG) registration, and onset of convulsions after the administration of propofol.
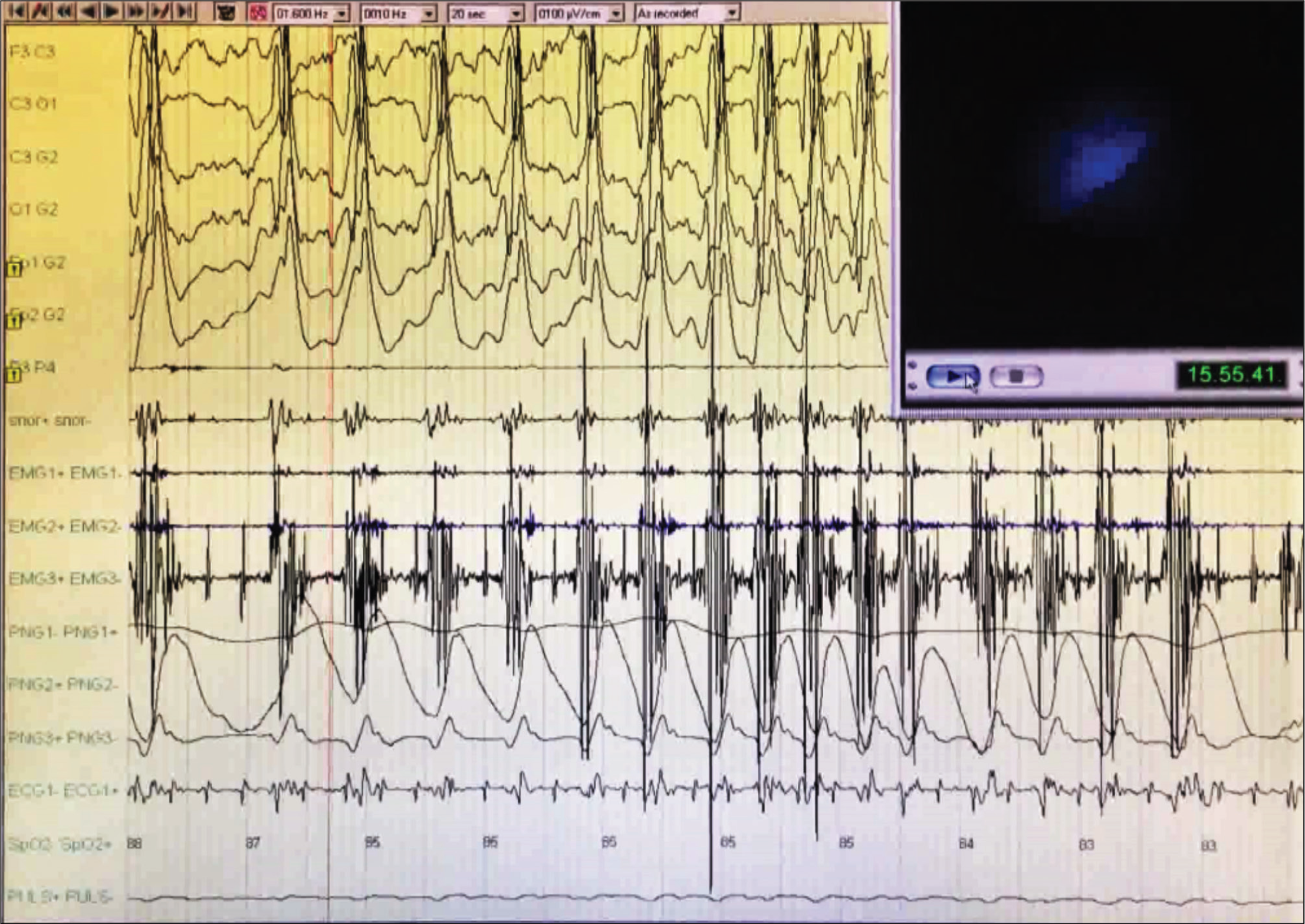
Polygraphic intraoperative recording with electroencephalographic (EEG) registration; spike-and-slow-wave complexes, medium-wide voltage, diffuse, with mixed very short sections of depression of electrical brain activity can be seen.
The electroencephalographic (EEG) trace recorded with derivations only from the left hemisphere showed the appearance of spike-and-slow-wave complexes, medium-wide voltage, diffuse, with mixed very short sections of depression of electrical brain activity (Figure I and Figure II). The seizures subsided completely after the intravenous administrations of diazepam (10 mg). The patient remained hemodynamically stable. The ongoing procedure was interrupted and the patient was awakened and hospitalized for diagnostic tests.
Serum electrolytes and postoperative computerized tomographic (CT) scanning of the head were normal. Neurology consultation revealed no neurological abnormality. A 24-hour cardiological evaluation with ECG-Holter and tilt-test showed no signs of alteration. Laboratory polysomnography after sleep deprivation did not show epileptic events on EEG monitoring. Magnetic resonance imaging (MRI) of the brain was normal.
After six months from DISE, the patient underwent surgery of septoplasty and turbinectomy without intra or postoperative complications.
Discussion
Propofol is a hypnotic drug with doubtful proconvulsant and anticonvulsant actions, believed to act by activating the GABA receptors and enhancing transmission like other drugs, such as diazepam and thiopentone [7].
Despite the evidence that propofol may have proconvulsant activity, many authors state its anticonvulsant effect. Propofol has been shown to be effective in controlling status epilepticus; continuous intravenous infusions of midazolam, pentobarbital, thiopental, or propofol represent the third line treatment against refractory and superrefractory status epilepticus [8].
The pathophysiological mechanisms of the neuroexcitatory activity of propofol are unknown. Spontaneous movements induced by propofol are not related to cortical activity and subcortical origin [9]. Several mechanisms have supposed and several studies have proposed the biochemical and biophysical origin of the excitatory phenomena using propofol. The most accredited hypothesis is that high doses of propofol depress both the cortex and subcortex with an anticonvulsant effect; while in low doses, it may act as a proconvulsant by inhibiting the inhibitory subcortex, resulting in the 'release' of the normal hyperexcitability of the cortex [10].
Some works explored the subcortical effect of propofol. Borgeat et al. [11] noted that its antiemetic effects, attributable to subcortical action, last much longer than its hypnotic effects, supposing that the pharmacodynamics of propofol may differ at various levels, with mechanisms that cause the excitatory effects on induction. If the cortical epileptic foci were inhibited by subcortical activity, then it would be possible that an accumulation of propofol at the subcortex level during the induction or the emergence phases could release the epileptic seizure activity in susceptible patients [12].
In a recent review, Walder et al. [13], according to the time point of their occurrence, showed that most of the phenomena happened during the induction or emergence phases, suggesting that a change in cerebral concentration of propofol may be causal. In our case, we observed the crisis for few seconds after the drug injection confirming this hypothesis. Moreover, the TDI method used during DISE implies the use of increasing doses of the drug until BIS between 50 and 70 is reached, thus making the method sensitive to this adverse event.
Few minutes after the administration of propofol, our patient developed opisthotonus with extension of the upper and lower limbs, tonic–clonic movements, muscle rigidity and hyperextension of the neck lasting for about 15–20 seconds; it stopped for one minute and then other two episodes of the same duration took place. During EEG registration, we observed the onset of spike-and-slow-wave complexes, diffuse, with short sections of depression of electrical brain activity.
The event that we observed in the operating room led us to doubt the initial diagnosis of OSA, since sleep apnea syndrome and nocturnal seizures are often associated and a misdiagnosis can lead to the aggravation of both diseases [14]. In our case, the patient had no history of epilepsy, the post-operative awakening was normal, brain CT scan and MRI were normal, EEG registration and laboratory pol-ysomnogram with deprivation of sleep were also normal. We think it may have been an adverse event related to the use of propofol.
Another consideration is that the hypoxia caused by the apnea, added to propofol-induced respiratory depression, could be the cause of seizures in patients with OSA [15].
Our current opinion is that the dose and TDI method used for propofol during DISE can cause seizures in sensitive subjects.
Conclusion
Propofol is a hypnotic drug with doubtful proconvulsant and anticonvulsant actions; electroencephalographic evidence to support such claims is variable. The complete polygraphic intraoperative recording during DISE, inclusive of brain electrical function monitoring, allowed us to record the proconvulsive effect of propofol, a rare but possible adverse event in sensitive subjects.
What is new?
Propofol is a hypnotic drug with doubtful proconvulsant and anticonvulsant actions. Electroencephalographic evidence to support such claims is variable; the complete pol-ygraphic intraoperative recording during DISE, inclusive of brain electrical function monitoring, allowed us to record the proconvulsive effect of propofol – a rare but possible adverse event.

