Background
Hypoglycaemia is more frequent among diabetic patients as a result of hypoglycaemic lowering therapies, making the diagnosis and management straightforward. However, when documented in non-diabetic people it becomes a diagnostic problem. Despite their seemingly benign behaviour, the aetiology and posterior treatment is of crucial importance, since recurrences are frequent and prolonged hypoglycaemia can cause serious brain lesions, making mortality higher among non-diabetic patients [1]
We present a case of a healthy-looking patient who presented to the emergency department with symptomatic hypoglycaemia.
Case Presentation
A 36-year-old man was admitted to our hospital due to an episode of altered mental status with documented hypoglycaemia, already corrected at home. The patient had been well until 6 years before admission, when he started various episodes of blurry vision and profuse sweating. During one of the episodes he had temporary loss of conscience with tonic-clonic movements of the limbs, according to his wife. The episodes were more frequent early in the morning while fasting or at night just before dinner. He was observed by a neurologist who did not start medication since it was an isolated occurrence. Five years later his wife noticed an episode of starry look, with profuse sweating and blurry vision described by the patient. She brought him to the emergency room and after doing computed tomography (CT) scan, which showed no alterations, he was discharged home. In the next day, he had a similar episode, this time with urinary incontinence and numbness of the arms. He was medicated with levetiracetam and referred to a neurology consultation, 4 months later, where his medication was altered to Zonidamide. Two days after the consultation, his wife called the pre-hospital emergency team because the patient described blurry vision followed by profuse sweating and loss of conscience with involuntary movements of the superior limbs. When the emergency team arrived at his home, they documented a capillary glycaemia of 54 mg/dl administrating 20 ml of intravenous 30% glucose and carried him to the emergency department. The patient had no medical conditions known and was only on zonidamide. He denied drinking alcoholic beverages and had no fever, chills, weight loss, abdominal pain, diarrhoea, dysuria, shortness of breath or chest pain. Polydipsia, pol-yphagia and polyuria were also denied. He lived with his wife and one young child. There was no family history of heart, pancreatic or autoimmune disease, and no one in the patient’s home used insulin or other diabetes medication. In the emergency department, the patient was asymptomatic. On the examination, he presented with a temperature of 36.7ºC, blood pressure of 128/75 mmHg, heart rate of 75 beats per minute, respiratory rate of 18 breaths per minute and oxygen saturation of 98% in ambient air. The patient was oriented and calm, with the neurologic and general exam completely normal. The complete blood count and levels of electrolytes were normal, as were results of renal-function tests. The patient was admitted to study the cause of hypoglycaemia. Due to the history of possible seizures, he was transiently admitted to an intermediate care unit to do the 72 hour fast test. During the fast, the patient consumed only water and, every 2 hours, a glucose level was obtained by fingerstick testing and an assessment was performed for subjective symptoms of hypoglycaemia or objective evidence of altered mental status. At 16 hours, blood glucose was 40 mg/dl and blood levels of glucose, C-peptide and insulin were obtained (Table 1).
Table 1 demonstrates the laboratory values during the 72-hour fast test. In the first row, one can see high levels of C-Peptide (68.7 ng/ml) and Insulin (7.07 uU/ml) in the presence of hypoglycaemia (40 mg/dl). The next three rows represent the response to the administration of one milligram of glucagon after 10, 20 and 30 minutes, demonstrating the expected increase of more than 25 mg/dl in blood glucose after that time. These results were compatible with endogenous hyperinsulinism.
The test for insulin antibodies was negative. One milligram of glucagon was administered with an increase of more than 25 mg/dl in blood glucose after 20 to 30 minutes. The results were compatible with endogenous hyper-insulinism and the team initiated the investigation of a possible insulinoma.
While waiting for the Magnetic Resonance Imaging (MRI), all the anti-epileptic drugs were discontinued, a personalised diet was initiated and medical therapy for the hypoglycaemia started initially with subcutaneous octreotide and posteriorly, when available, with diazoxide, titrated up to a dosage of 900 mg/day divided in three daily intakes in order to keep euglycemia.
The abdominal MRI revealed a nodular lesion of approximately 14 mm, hyperintense on diffusion-weighted imaging with contrast hypercaptation, indicating a possible primitive pancreatic lesion. The Positron Emission Tomography (PET) scan with 68Ga-DOTA-NOC demonstrated hyper-captation of the nodular lesions described above.
The diagnosis of insulinoma was assumed, supported by all the exams above, and the patient was referred for surgical resection of the insulinoma by laparoscopic enucleation with no immediate complications. Since then it was possible to keep euglycemia. He was transitioned from a liquid to a normal diet and surgical drain was removed after confirming output of serous fluid with a low amylase. He was discharged home 7 days after the surgery. The pathological exam revealed a well-differentiated pancreatic neuroendocrine tumour, grade 1 with positive insulin receptors, corroborating the diagnose of insulinoma.
| PARAMETERS | 0´ | 10 ´ | 20´ | 30´ |
|---|---|---|---|---|
| Glucose (60–100 mg/dl) | 40 | 54 | 75 | 88 |
| C-Peptide (1.1–4.4 ng/ml) | 68.7 | - | - | - |
| Insulin (2.6–24.9 uU/ml) | 7.07 | - | - | - |
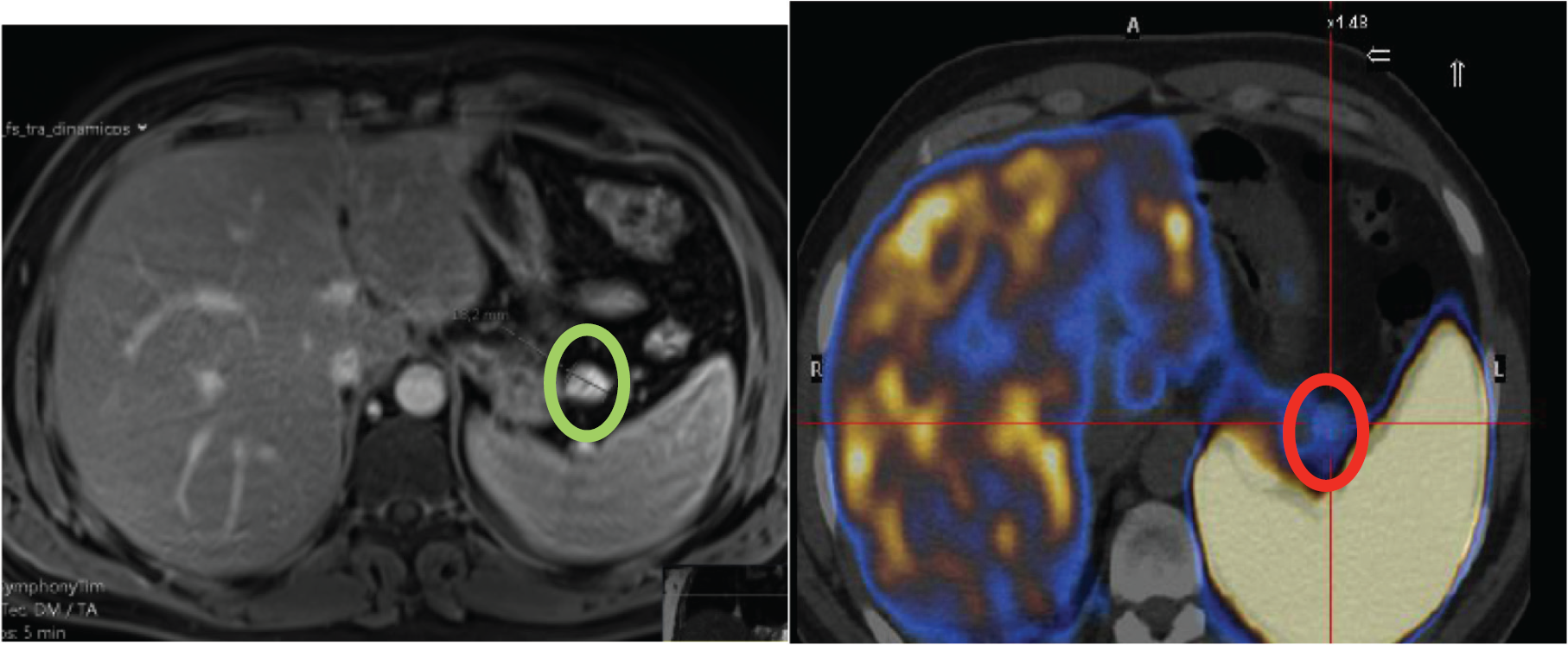
MRI showing a nodular lesion of 14 mm (green circle), hyperintense on diffusion-weighted imaging and the PET scan with 68 GA-DOTA-NOC demonstrating the hypercaptating nodule (red circle) compatible with an insulinoma.
After discharge, the patient was observed in a routine postsurgical follow-up. He was recovering well, without further symptoms of hypoglycaemia. After group reunion, he was counselled for follow-up regarding the 6% risk of recurrent insulinoma within 10 years.
Discussion
In clinical practice, hypoglycaemia is mainly caused by insulin or insulin secretagogue used to treat diabetes mellitus. Hypoglycaemia is rare among non-diabetic persons and its investigation should only be made when it is accompanied by Whipple triad-symptoms or signs consistent with hypoglycaemia, a plasma glucose level less than 55 mg/dl, measured with a precise method such as a venous blood sample, and resolution of symptoms after raising plasma glucose level. The first physiological response to a decreasing plasma glucose level is a down-regulation of insulin secretion, followed by a heightened glucagon secretion. Even though most of the times hypoglycaemia presents with typical neuroglycopenic symptoms, sometimes seizures occur, making essential to rule out hypoglycaemia as its cause, as occurred in this case report. Its aetiology, while generally straightforward in an ill patient with medical conditions, such as sepsis, end-stage liver failure, chronic renal failure or another, may be a quest in the healthy looking patient, requiring detailed medical history and laboratory evaluation. A detailed medical history with past medical conditions, pharmacologic habits and alcohol use is essential to this investigation [2–4].
Endogenous hyperinsulinism is a rare cause of hypoglycaemia in healthy patients and should be considered when all other causes of hypoglycaemia have been excluded. It can be caused by autoimmune diseases, use of medications which alter insulin secretion, altered secretion of insulin and gut hormones after gastrointestinal surgery and insulinoma. Insulinoma is the classic cause of endogenous hyperinsulinism, despite being a rare neuroendocrine tumour. Its incidence is so low that little information is known about the demographic distribution. According to Mayo Clinic, its incidence is around 0.4 cases per 100,000 habitants [5]. Since it is a tumour with autonomous capacity of producing insulin, its diagnose is made when high serum insulin concentrations are measured during a spontaneous or induced episode of hypoglycaemia. After diagnosis, imaging techniques are needed to localise it. The location must be accurate since most of the tumours are too small to be palpable during surgery.
Transabdominal ultrasonography, CT, MRI, Pentetreotide scintigraphy and PET are non-invasive procedures available for tumour location. Attention is needed when interpreting these results, since up to 40% of insulinomas do not express enough subtype 2 somatostatin receptors [6,7]. The treatment of choice is surgical resection of the tumour, but in case of high surgical risk chemical ablation is possible with ethanol injection [8]. While waiting for definitive treatment, hypoglycaemia must be managed with pharmacological therapy such as octreotide, diazoxide or verapamil.
The overall survival rate does not differ from that expected in the general population. However, caution is needed since the cumulative incidence of recurrence was 6% at 10 years and 8% at 20 years.
Conclusion
With this clinical case, the authors want to remind the main steps in the investigation of hypoglycaemia in non-diabetic patients, emphasising all the clinical problems in managing the hypoglycaemic episodes before the surgery.
What is new?
Hypoglycaemia is rare among non-diabetic patients and its aetiology is not straightforward. Endogenous hyperinsulinism is a rare cause of hypoglycaemia and rarely is the result of an insulinoma. Insulinoma treatment of choice is surgical resection and the prognosis is good since overall survival rate does not differ from that expected in the general.

