Background
Acute necrotizing encephalopathy of childhood (ANEC) is a rare but distinctive type of encephalopathy predominantly involving infants and young children, which was first described by Mizuguchi et al. [1]. It is commonly found in Asian populations with most of the cases reported in Taiwan, Japan, and Korea. It was initially thought to be unique to Asian populations, but an increasing number of cases had been found in Western countries too [2]. The etiology and pathogenesis of ANEC are still unknown, but viral infections appear to be the causative agents in most cases, commonly influenza A, mycoplasma, and herpes simplex virus. ANEC is known to have a fulminant clinical course, with affected patients having high mortality and neurological sequelae.
Case Presentation
An 11-year-old boy with no known medical history presented to his general practitioner with a 2-day history of high-grade fever (39oC), headache, myalgia, and sore throat. There was no neurological symptom at the time of presentation. He was treated as having upper respiratory tract infection and was discharged home with symptomatic treatment. On the following day, he deteriorated rapidly with multiple episodes of tonic-clonic seizures and subsequently developed impaired consciousness. On arrival to the hospital, his GCS was only 5/15. He was, therefore, intubated and loaded with intravenous (IV) phenobarbitone. On clinical examination, he had hypertonia and increased deep tendon reflexes in bilateral upper and lower limbs. His cranial nerves examinations were unremarkable, and there was no urinary incontinence. On chest auscultation, the lungs were clear.
The laboratory investigations showed thrombocytopenia, coagulopathy, deranged renal profile, and metabolic acidosis. CSF analysis and culture were normal. Influenza A H1N1 virus was isolated from the child’s endotracheal secretion sample which confirmed the diagnosis. In view of the rapid neurological deterioration, ANEC was suspected, and magnetic resonance imaging (MRI) brain was performed. The MRI showed symmetrical T2 hyperintensities at bilateral thalami with associated hemorrhagic component (Figure 1). T2 hyperintensities with hemorrhages also noted at the tegmentum of midbrain and pons, cerebellar vermis, and bilateral cerebellar hemispheres (Figure 2). Overall radiological findings are in keeping with ANEC.
He received a course of IV methylprednisolone (30 mg/ kg/day), IV immunoglobulin (2 g/kg), and oral tamiflu. His condition improved initially, and a trial of extubation was performed after 2 weeks of ventilation but failed. He was reintubated due to worsening respiratory distress. Subsequently, he underwent tracheostomy in view of prolonged ventilation and was then discharged back to the district hospital for supportive treatment.
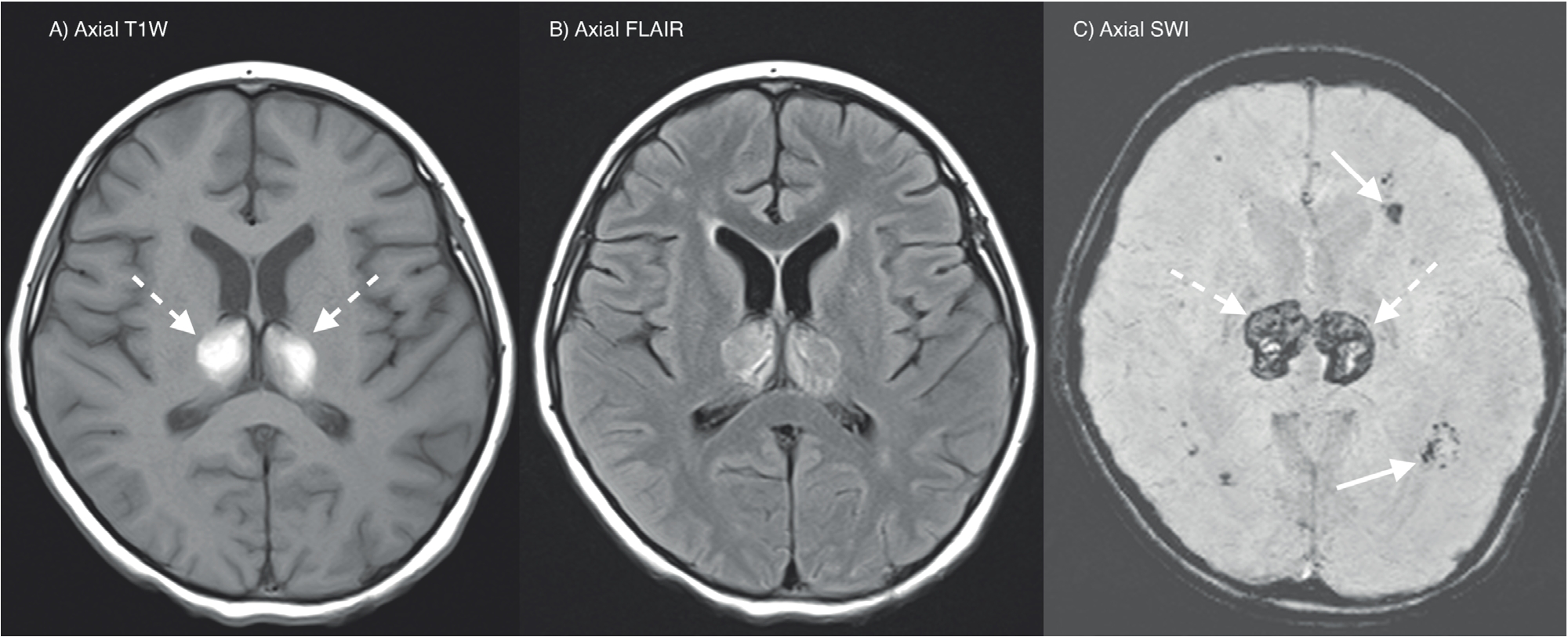
A and B: T1W and FLAIR axial MRI brain shows symmetrical hyperintensities at bilateral thalami (dashed arrows). C: SWI axial MRI brain shows abnormal susceptibility at bilateral thalami (dashed arrows) and periventricular white matter (straight arrows) in keeping with hemorrhages.
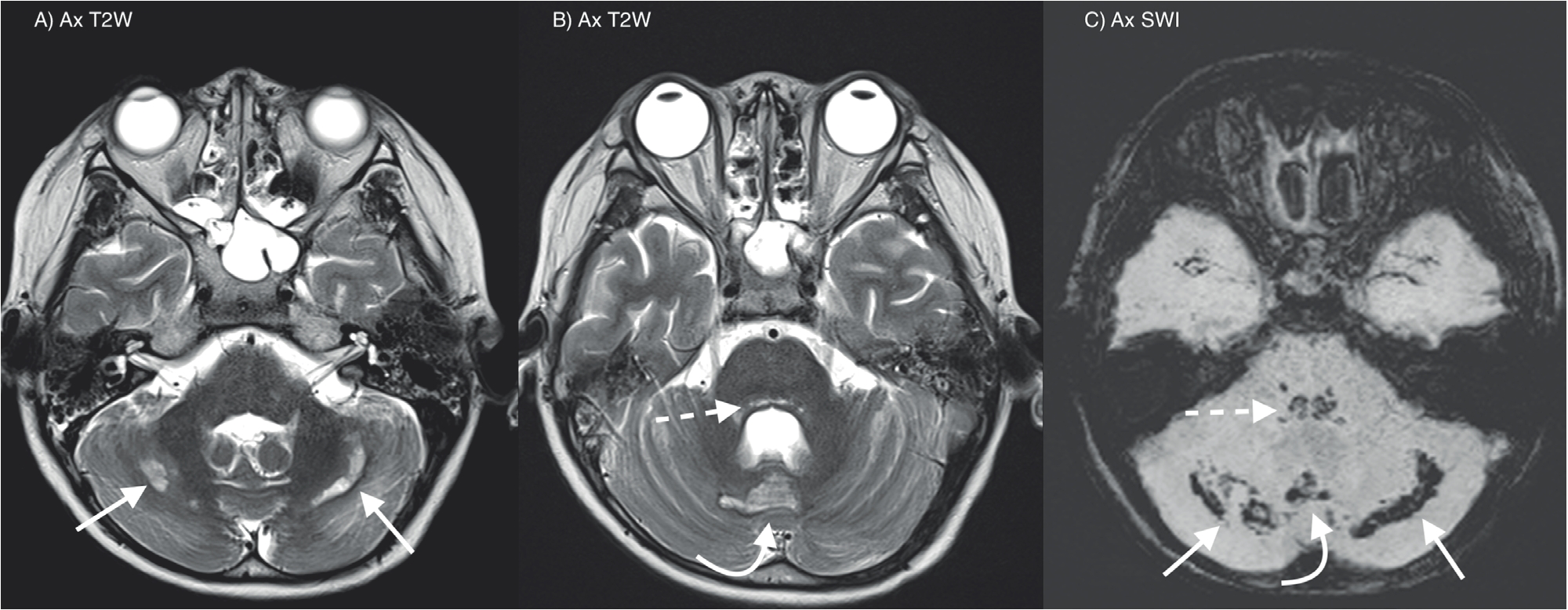
A and B: T2W axial MRI brain shows abnormal hyperintensities at the tegmentum (dashed arrow), cerebellar vermis (curved arrow), and bilateral cerebellar hemispheres (straight arrows). C: SWI axial MRI brain shows corresponding abnormal susceptibility at these regions in keeping with hemorrhages.
Discussion
Both environmental and host factors may contribute to the development of ANEC. Commonly, ANEC develops secondary to viral infection, and in this case, it is caused by influenza A virus. The exact mechanism of how ANEC develops is still unknown, but the most prevalent hypothesis for this condition is secondary to hypercytokinemia or cytokine storm [3–5]. Patients suffered from ANEC were found to have an exaggerated immune response to viral infection, causing an increased level of proinflammatory cytokines. This will subsequently result in systemic inflammatory response syndrome, and the patient can present with liver and renal dysfunction and disseminated intravascular coagulation (DIC) as seen in this case. In the brain, the increased level of cytokines results in alteration permeability of the intracranial vessel walls, causing disruption of the blood-brain barrier, and subsequently results in damage to the central nervous system. This hypothesis was supported by many studies showing elevated levels of serum and CSF cytokine in patient suffering from ANEC. Interleukin 6 and tumor necrosis factor-a are believed to be the possible culprits as both can cause damage to the central nervous system when in high levels [4,6,7].
There is no sign or symptom which is specific for ANEC; therefore, diagnosis based on clinical information alone is usually not possible. Patients commonly present with prodromal of viral infections, such as upper respiratory tract infection or gastroenteritis. With progression to ANEC, patients will have sudden deterioration with the development of convulsions, neurological deficits, and impaired consciousness. In addition to that, there will be signs of multiorgan failure and DIC. The patient will usually go through three phases of clinical course, which include prodromal stage, acute encephalopathy stage, and recovery stage [2,8]. The clinical course of ANEC can be fulminant with high mortality and morbidity rates, and most of the patients will develop severe neurological sequelae [2,9,10].
Neuroimaging plays a vital role in the diagnosis of ANEC with its characteristic imaging findings as shown in this case. With careful clinicoradiological correlation, the diagnosis of ANEC can be made. A symmetrical involvement of the thalami is the most common and consistent findings in all reported cases. Brainstem, cerebellum, and periventricular white matters are also commonly involved [10,11]. Depending on the timing of the neuroimaging, the radiological findings may vary. Neuroimaging shows dynamic changes from edema in the early stage to petechial hemorrhages and necrosis in the later stage. At the onset of ANEC, the affected regions of the brain will demonstrate T2 prolongation and mass effect, which indicate swelling and edema. Subsequently, edema will resolve, and hemorrhage with necrosis will ensue. MRI will show T1 shortening with corresponding susceptibility artifacts on gradient echo imaging in keeping with hemosiderin deposits [2,10,11]. Depending on the location of involvement, the differential diagnosis will include acute disseminated encephalomyelitis, hemolytic uremic syndrome, and viral encephalitis.
Even though there is no specific treatment for ANEC at the moment, early recognition and diagnosis are still important. In most of the cases, the management involves intensive care, symptomatic treatment, immunomodulatory agent, and antiviral therapy. As discussed earlier, the development of ANEC is likely due to hypercytokinemia; therefore, immunomodulatory therapy which suppressed cytokine production has the potential to improve the clinical outcome of ANEC. Early steroid treatment is also shown to improve the clinical outcome of the patient without brainstem lesion as reported by Okumura et al. [12]. Nevertheless, the prognosis of ANEC remains poor, varying from complete recovery to death. The reported mortality rate is about 30%, and merely less than 10% of the patients recovered with neurological sequelae [2,9,10].
Conclusion
Acute necrotizing encephalopathy of childhood (ANEC) is a progressive and devastating disease, in which there is no proper treatment at the moment. The prognosis of ANEC can be devastating with high mortality rate and severe neurological sequelae. The diagnosis of ANEC is usually made based on the clinical and characteristic neuroimaging findings as described in this case. Therefore, early neuroimaging is indicated and should be performed early in patients with suspected ANEC. Prompt diagnosis is essential as immunomodulatory therapy can be started, which might potentially improve the outcome.

