Background
Post-ischemic ventricular septal rupture (VSR) is a rare event, complicating about 0.17-0.31% of patients with acute myocardial infarction (AMI) [1]. VSR is traditionally considered as a late event, occurring 3-5 days after AMI, but latest studies show a trend in decreasing of this time interval [2], which could be due to increasing availability of bedside echocardiography and to greater familiarity with VSR. Targeted research of the defect in anterior or inferior/lateral AMI and differences in pathophysiological change in myocardial tissue repair process secondary to earlier reperfusion and fibrinolysis could also reduce this interval. The mechanism leading to VSR is related to coagulative necrosis of the ischemic tissue in a transmural infarction, while precarious ruptures are frequently secondary to intra-parietal hemorrhagic complications of AMI suck hematomas or to dissection of the septal wall [3]. Clinical presentation is widely variable, but a rapid hemodynamic deterioration is frequently found and correlates to mortality of 41-80% in last decades [4]. When VSR is diagnosed (usually through accurate clinical evaluation and trans-thoracic or trans-esophageal echocardiography), a multidisciplinary management of the patient is mandatory to reach the best outcome. Medical therapy for VSR consists in afterload reduction to minimize left-to-right shunt, while surgical repair is definitive intervention. Best timing and modality of surgery are controversial and should be determined according to clinical condition of the patient, comorbidities, morphological and functional characteristics of septal defect [5]. During last years, percutaneous VSR closure demonstrated a good efficacy and improved outcome in patients with high surgical risk or as a bridge-therapy to surgical repair in unstable patients [6].
Case Presentation
A 74-year old man presented to the emergency department (ED) at 6:03 p.m. complaining about asthenia, malaise, lack of appetite, and occasional mild nausea for the last 3 days. He also reported some episodes of vomiting and diarrhea about 10 days before, with complete spontaneous resolution within 48 hours. His medical history was significant for well-controlled hypertension and chronic headache responsive to paracetamol. He was given a “green” priority code and was conduced to waiting room. At first medical examination (10:43 p.m.) he was asymptomatic except for moderate asthenia, his vital signs were within normal ranges, he was afebrile and had no respiratory symptoms. Physical exam revealed mild pallor, some pulmonary crackles and a significant systolic murmur. Blood samples were sent to laboratory and heart murmur was assessed by performing ECG and bedside echocardiography. ECG (Figure 1) showed an inferior STEMI with ST-depression in anterior and lateral leads (V1, V2, I, and aVL) and Q waves in inferior leads. Cardiac enzymes analysis was added to blood texts previously performed, cardiology consult was required and patient was monitored (11.28 p.m.). Bedside echocardiography showed right ventricle enlargement and color-doppler evaluation in apical view demonstrated a significant left-to-right flux across a septal defect of a maximum diameter of about 9 mm (Figure 2). This data were confirmed with echocardiography performed by cardiologist and when laboratory informed ED that cardiac specific high-sensibility Troponin of the patient was 7,978 pg/ml (normal values 0-14 pg/ml), admission to Coronary Unit was already confirmed. It was 1:05 a.m. During 7 hours spent in ED, the patient was persistently asymptomatic, though anxious: he did not complain of chest pain, heartburn, dyspnea, nausea, palpitation nor other symptoms. Since Cardiac Surgery department was absent in the hospital of admission, following management was agreed with consultants from Cardiothoracic department of closest hospital: given hemodynamic stability of the patient, the transfer was planned for the following day and reperfusion was considered a priority. Emercengy coronary angiography revealed a complete occlusion of proximal right coronary artery and percutaneous coronary intervention was performed restoring blood flow with balloon angioplasty. Delayed surgery was reputed the best option for this patient and surgical repair was performed at day 8 from first access to ED. Left ventriculotomy was preferred by surgeon to access the VSR and Daggett technique allowed to easily close the defect by placing a double patch over the VSR (Figure 3). The patient had an uncomplicated course and was discharged at day 19 from first access to ED.
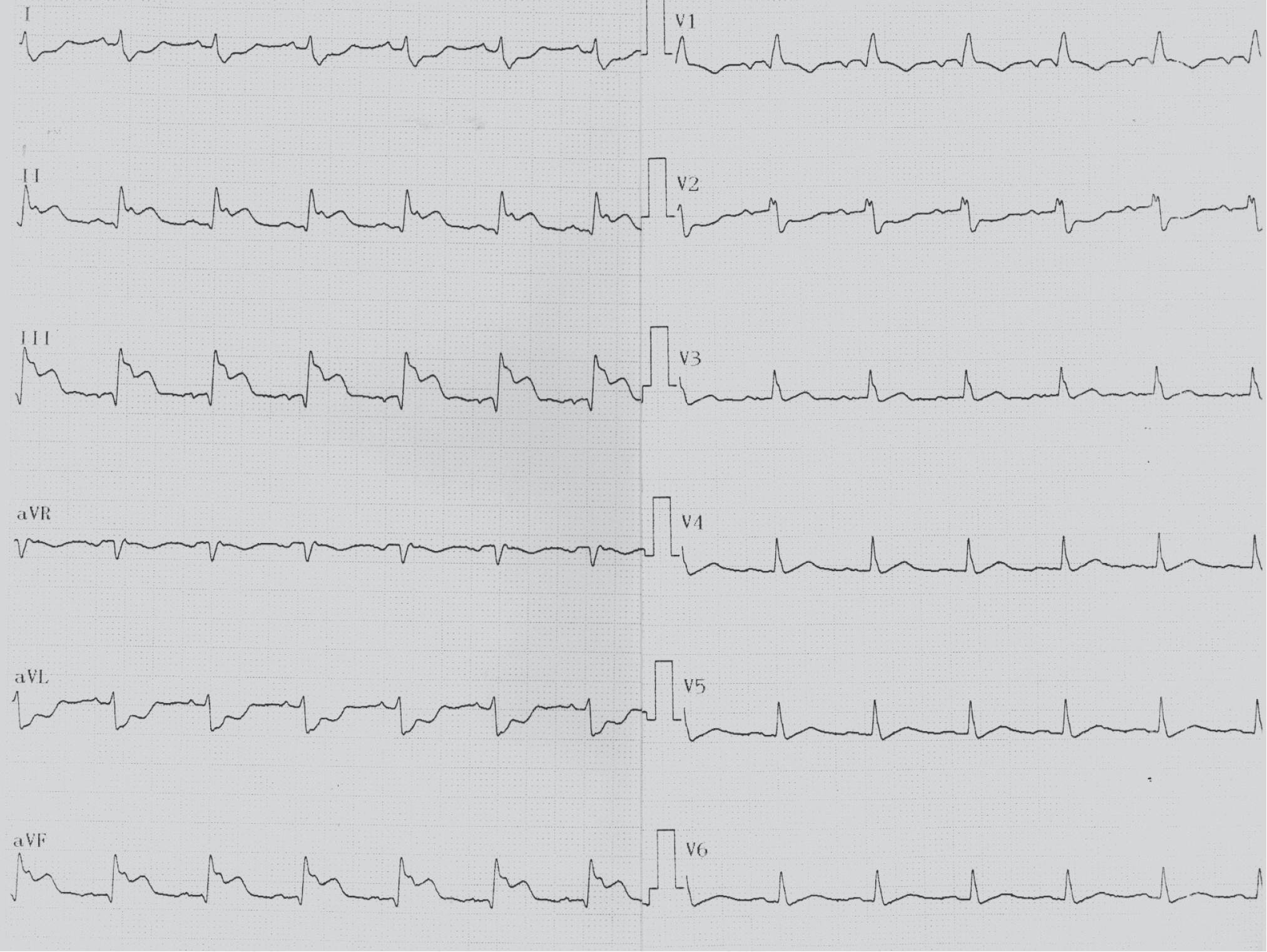
ECG showed a sinus rhythm with a ST elevation and Q waves in inferior leads with specular ST-depression in leads V1, V2, I and aVL.

Echographic images of the defect in parasternal view: on the left and lower portion of images, left ventricle is sectioned along its major axis and appear slightly hypertrophic. The right ventricle, in the upper and left portion of the images, appears dilated. (a) The entire course of the interventricular defect. (b) The defect in the projection that highlights its larger diameter. (c) Color-Doppler showing flux along the rupture. (d) Color-Doppler showing the left-to-right shunt in the right.
Conclusion
Early revascularization in AMI progressively reduced the prevalence of any complication of ischemic event, even more frequent like hypokinesis or arrhythmia. Although it is a rare complication of AMI, VSR is a life-threatening event and should be suspected in any patient with AMI, particular anterior and inferior/lateral ones, when a rapid hemodynamic deterioration is observed or when physical examination reveals a heart murmur or signs suggestive for right ventricle overload. Echocardiography is always needed and should be immediately performed in the ED. When VSR is diagnosed, a multidisciplinary team should collaborate to plan following management of patient, to obtain best outcome possible, according to high mortality related to this complication.

