Background
Eosinophilic gastritis is a rare disease that is characterized by the eosinophilic infiltration of the various layers of the gastrointestinal tract in the absence of any definite causes of eosinophilia. Patients with eosinophilic gastritis have diverse symptoms, including abdominal pain, emesis, abdominal distension, and weight loss [1]. Up until now, the epidemiological and pathophysiological studies of eosinophilic gastritis remain unclear. The severity of the presentation varies broadly from regurgitations to hematemesis. We present two cases with eosinophilic gastroenteritis caused by milk protein allergy, manifested with severe fresh blood-strained vomiting.
Case Presentation
Case 1
A 4-month-old female patient, born at full term and weighing 3,200 g, received full course of vitamin K prophylaxis in the first month of life. She was the fourth normal pregnancy of her mother. The patient was fed formula milk since birth. According to her mother, her symptoms started at 3 months of age. Prior to admission, she was suffering the common flu with a runny nose, fever, and cough. Full recovery of her flu was reported after 5 days. She was taking paracetamol continuously at home (dose of 80 mg/kg) for more than 2 weeks, due to an attempt by the mother to help aid the child’s recovery.
For approximately 1 month, the child continued vomiting after each feed, and for a period of 6 days, there was presence of fresh blood in the vomit. She had already been hospitalized three times at the Pediatric and Surgery Department in multiprofile hospital for active treatment Burgas, for evaluation of vomiting and hematemesis. The child was later referred to a university hospital for further evaluation.
On physical examination, the child presented with a satisfactory general condition – alert with no signs of respiratory or cardiac failure. She had normal vesicular breathing and clear heart sounds with no additional murmurs. The abdomen was soft and non-tender on palpation. Bowel sounds were regular, and no pathological sounds were auscultated. There was no hepatosplenomegaly and the neurological exam was normal.
On her blood results, she presented with initial mild anemia, which continued to worsen throughout her 5-day stay. The patient had thrombocytosis on admission because of the stress from blood loss. She had an elevated erythrocyte sedimentation rate which worsened. She presented with eosinophilia at the end of her stay.
No significant results from her blood biochemistry were received. Her coagulation profile was normal. Arterial blood gases and urine tests were within normal ranges. The skin prick test of milk protein and used formula was negative. The abdominal ultrasound examination was normal.
A decision was made to conduct an endoscopy for further evaluation of hematemesis. Gastroscopy revealed acute erosive gastropathy with diffuse hyperemia of the gastric mucosa and multiple erosions in the gastric body and antrum. Endoscopic findings in the esophagus and duodenum were normal.
An endoscopic biopsy from the antrum was taken and examined histologically. The morphological result showed the involvement of the entire thickness of the mucosa with findings suggestive of chronic active gastritis. Large amounts of eosinophilic cells (>20 cells/hpf) were observed among the inflammatory cells (see Figures 1 and 2). Helicobacter pylori were not found.
Initial antacid treatment with H2 blockers and infusions was started. There was no improvement of the patient’s condition. After receiving the histological reports, the patient was diagnosed with allergic gastritis, who had gone through erosive gastropathy resulting from allergy to cow’s milk protein. Elimination test with hydrolyzed milk was given. After this elimination, there was absence of new bleeding episodes, indicating a positive response. She suffered no further complications.
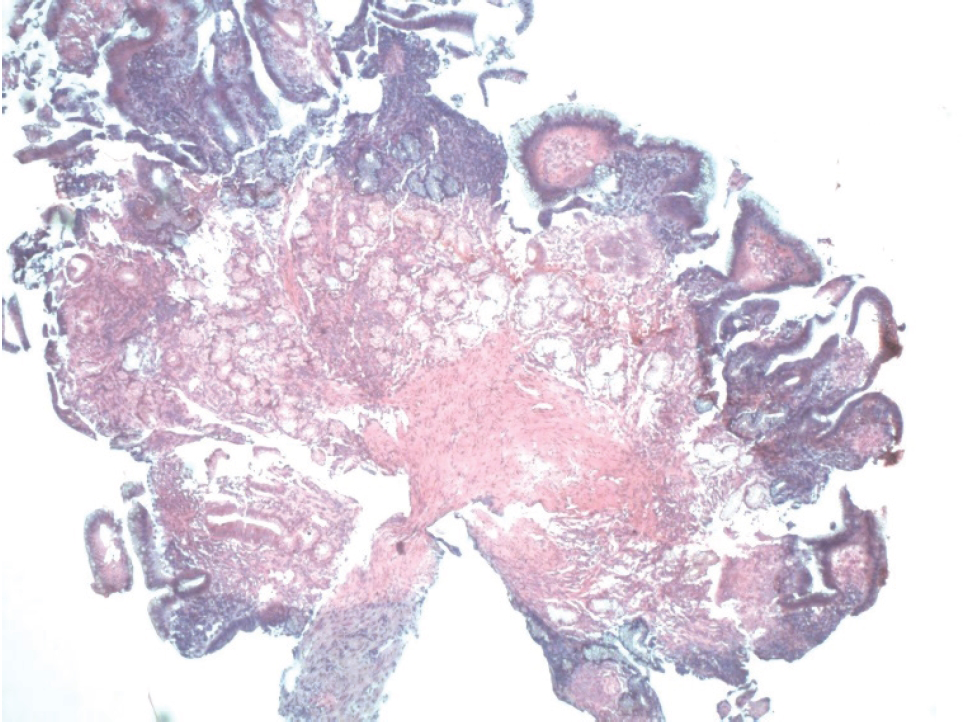
Histological biopsy of stomach of case 1 Involvement of the entire thickness of the mucosa. Eosinophilic cells (>20 cells/hpf) observed among the inflammatory cells.
Case 2
A 6-month-old female, born of a third normal pregnancy, who was breast fed from birth. At birth, her weight was 3,560 g and height was 53 cm. She had no remarkable complaints up to 3 months of age. From 3 months of age, she began suffering from a fever, runny nose, cough, and three episodes of hematemesis. This was treated with antibiotics and non-steroidal anti-inflammatory drugs (NSAIDs) for a period of 10 days. Three weeks later, the patient presented with another episode of coryzal symptoms, but in the absence of fever. One month later, an episode of hemoptysis occurred. Blood tests were done, and hemoglobin was significantly low at −65 g/l with no icterus and normal urine analysis. It was reported that she often took paracetamol as an “anti-viral drug” for the past 2 months.
At 6 months of age, the child once again began coughing with fever up to 38.5°C. After review, by the general practitioners, she was given amikacin intravenously and symptomatic treatment for 5 days. Her symptoms began to improve. However, 2 weeks later, she was admitted to our clinic with another episode of hematemesis after feeding, and the next day had stools of slight green consistency.
On clinical examination, she was in good general condition with a weight of approximately 7,000 g and a height of 67 cm. She was alert with normal skin turgor and elasticity. The child had reduced nasal patency with no secretion. Respiratory and cardiovascular examination demonstrated no pathology. The abdomen was soft and non-tender with regular bowel sounds. No tumor masses or hepatosplenomegaly was found.
Significant blood results were recorded on the day of admission and 4 days later. Initially, her Hb was low and later demonstrated no improvement. She had leukocytosis, which remained high. Platelet count was high, but then it dropped down.
The only significant result from her biochemistry tests was an elevated lactate dehydrogenase (LDH) of 687.0 U/l. Coagulation tests were within normal ranges. Arterial blood gases were normal. Immunological stool test for occult bleeding was negative and urine tests were normal.
Radiography of the lung demonstrated bilateral white lungs without pathological lesions. A broad network drawing appeared which was treated as an interstitial inflammatory process. There were clear costophrenic sinuses, but diaphragmatic restrictions were present. The abdominal ultrasound showed normal findings.
Consultations with the Ears, Nose, Throat specialist were done. No bleeding sites in the upper respiratory ways were reported.
Gastroscopy showed normal esophageal findings. The stomach mucosa showed diffuse hyperemia within the corpus and fundus. There were multiple small erosions and one large one with dimensions up to 3 mm at the gastric corpus. There was no active bleeding during the study. In D1-part of the duodenum, lymphatic nodular hyperplasia was observed.
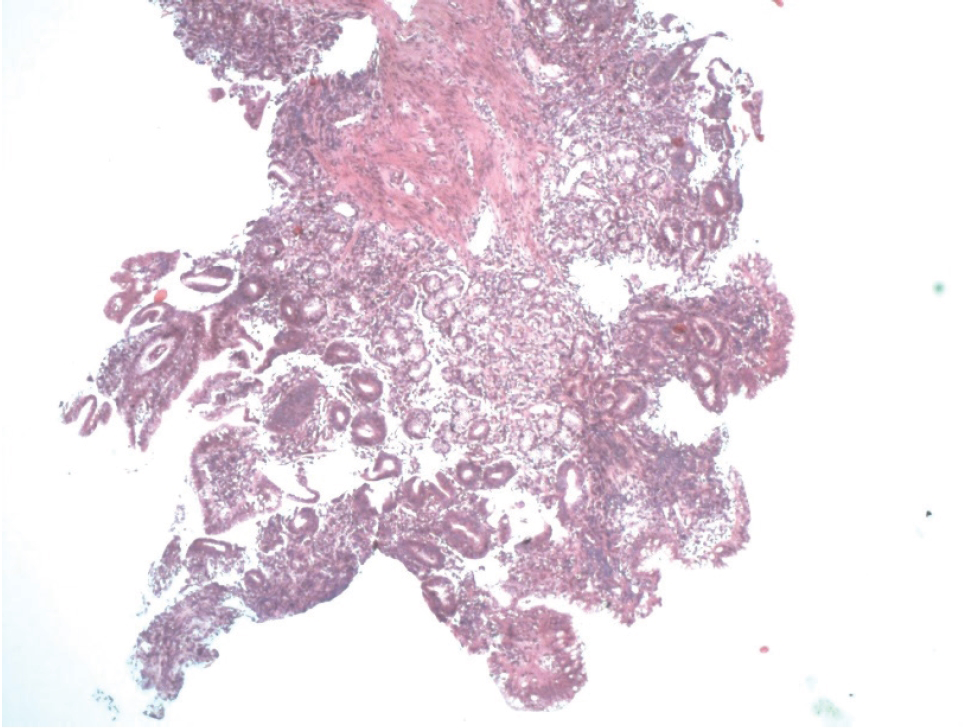
Histological biopsy of stomach of case 2.
Inflammatory infiltrate (lymphocytes, plasma cells, eosinophils) in the stomach and penetrating the entire mucosa (>25 cells/hpf).
Histology revealed an abnormal mucosal finding – inflammatory infiltrate in the stomach which was moderately pronounced and penetrating the entire mucosa. The infiltrate was represented by lymphocytes, plasmatic cells, and eosinophils (>25 cells/hpf) (see Figures 3 and 4). Helicobacter pylori were not found.
Initial treatment with H2 blockers and fluid showed no improvement. The patient was later diagnosed with acute erosive gastropathy as an allergy to cow’s milk protein, and elimination test with hydrolyzed formula was initiated. Her condition improved during her stay and there was gradual reduction in episodes of hematemesis. There were no further complications.
| Complete blood count (CBC) | Hemoglobin (HGB) 117 114 |
| Mean corpuscular volume (MCV) 72 71.4 | |
| Hematocrit 0.385 0.392 | |
| Platelets (PLT) 548 632 | |
| Erythrocyte sedimentation rate 33 38 | |
| Eosinophils 5.8% 6.1% | |
| LDH 687 | |
| Endoscopy | Acute erosive gastropathy with diffuse hyperemia of the gastric mucosa. Multiple erosions in the gastric body and antrum. |
| Histology | Involvement of entire thickness of the mucosa (suggestive of chronic active gastritis). Eosinophilic cells (>20 cells/hpf) observed among the inflammatory cells. |
| CBC | Initial After 4 days |
| HGB 106.0 109 | |
| WBC 18.59 18.08 | |
| PLT 643 459 | |
| MCV 71.7 71.4 | |
| LDH 687.0 | |
| Fecal occult blood test | (−) negative |
| Endoscopy | Stomach mucosa showed diffuse hyperemia in the corpus and fundus. Multiple small erosions. One larger erosion (dimensions up to 3 mm) at the gastric corpus. Presence of lymphatic nodular hyperplasia in D1-part of duodenum. No active bleeding |
| Histology | Inflammatory infiltrate (lymphocytes, plasma cells, eosinophils) in the stomach, which was moderately pronounced and penetrating the entire mucosa (>25 cells/hpf). |
| Other Imaging | Radiography of the lung demonstrated bilateral white lungs without pathological lesions. A broad network drawing appeared (interstitial inflammatory process). Clear costophrenic sinuses but diaphragmatic restrictions were present. |
In both our cases, we initially suspected erosive gastroscopy because of NSAIDs treatment. A lack of improvement from acid-suppression therapy and recurrent episodes of hematemesis were the main indications to conduct endoscopy investigations. Other diagnoses considered in our patients were late onset of hemorrhagic disease of newborn, acute infection gastroenteritis, bleeding disorders, and congenital structural abnormalities. All of these were ruled out with laboratory and imaging investigations. Our management was assessed as appropriate, based on the absence of further episodes of hematemesis and other presentations of food protein intolerance.
Discussion
Eosinophilic gastritis is an uncommon benign inflammatory condition which can be present in infants. It often leads to hematemesis along with other non-specific symptoms, such as abdominal pain, nausea, weight loss, and malabsorption. Clinical manifestations of milk protein allergy related to the digestive tract are very similar to those of lactose intolerance, which can easily lead to misdiagnosis. However, there are some crucial differences. For example, cow’s milk protein allergy (CMPA) can cause skin lesions (atopic eczema) and respiratory symptoms which are absent in lactose intolerance [2]. In addition, the presence of respiratory manifestations is further supported by a case report conducted by Simoniuk et al. [3]. Here, a patient presented with eosinophilic gastroenteritis which was associated with atopic symptoms, such as asthma, bilateral pleural effusion, and even ascites due to protein malabsorption (subserosal type). All of this suggests that the two atopic conditions of eosinophilic gastritis and asthma are closely linked. This information supports the diagnosis of eosinophilic gastritis in our second patient who presented with concomitant respiratory symptoms, including hemoptysis.
Unfortunately, the pathogenesis of eosinophilic gastritis is not well understood, but studies do suggest the role of T-helper cytokines and other mediators, such as interleukins. Tissue eosinophilia, peripheral eosinophilia, and elevated IgE levels are common in the literature [4,5]. A study by Tien et al. [6] supports this finding in a cohort of 14 patients. However, it is important to note that one-third of the patients did not have peripheral eosinophilia. In a cohort study involving 24 children, by Choi et al. [7], conducted at Seoul National University, 91.7% of the patients had peripheral eosinophilia. Our patients presented with eosinophilia and anemia on their blood results. The positive stool occult blood test is very important in diagnostics. The next steps in diagnosis are endoscopic examination and histology. It is important to obtain multiple biopsies since eosinophilic infiltration is often extensive [4–6]. This was supported by our second case where duodenitis was also present.
For an accurate diagnosis, the criteria required are clinical symptoms, histological evidence of eosinophilic infiltrates in the stomach, and the exclusion of other diseases which can mimic similar findings. The disease is divided into three subtypes according to Klein’s [5] classification: mucosal, muscular, and serosal. Often, this inflammation extends to the duodenum. Choi et al. [7] highlighted how many studies show predominance of mucosal layer disease (43%–100%), and from his study, 18 out of the 24 patients (75.0%) were classified with mucosal layer disease.
As mentioned earlier, the exclusion of other diseases is critical and diagnosis through a biopsy is not straightforward. This is because scattered intramucosal eosinophils can often be a normal finding of the gastrointestinal tract. Findings which should trigger concern include the collection of eosinophils which are not associated with other inflammatory cells, groups of cells associated with architectural injury (e.g., crypts), collections of eosinophils extending to the muscularis mucosae, or when mucosal involvement is patchy. However, numerous other diseases that present with high numbers of intramucosal eosinophils need to be ruled out, which include parasitic disease, inflammatory bowel disease, and non-Hodgkin’s lymphoma of the gut. It is important to note that allergic gastroenteritis is never associated with chronic architectural changes or metaplasia [8].
There is no standard treatment for eosinophilic gastroenteritis, and corticosteroid remains the main regime for suppressing eosinophilic-induced inflammation. Other choices that have been used include leukotriene antagonists or anti-interleukin 5 monoclonal antibodies. The treatment of choice is variable according to clinicians and depends on the severity and age of the patient [4]. A cohort study concluded that the lack of universal response to corticosteroids may bring into question the true effectiveness of this medication, especially considering potential adverse effects. Additionally, it may suggest non-allergic or non-immune-mediated pathogenic mechanisms in some patients [6]. Furthermore, the cohort study of Choi et al. [7] also supports this: 21.1% of the patients showed dependency and the remaining showed resistance to steroids.
Since eosinophilic gastritis is an atopic condition, avoidance of the allergen is an important part of the treatment. A review conducted by Lucendo et al. [9] regarding the efficacy of dietary regime for treatment of eosinophilic gastritis found that children who took an elemental diet showed a clinical improvement of 75.8%. However, there was lack of histological evidence to support this. Elimination of allergic foods was the most common option. With regard to CMPA, the exclusion of cow’s milk protein and its derivatives proves valuable. Alternatives include formulas made of hydrolyzed protein or amino acids. Other options include the implementation of soy-based products. This is supported by many reports in the literature, but it is important to note that up to 40% of the patients may present cross-sensitivity reactions [10]. In our patients, formula milk was implemented which proved useful.
Conclusion
Our cases are unique since both patients had their gastritis develop into an acute erosive gastropathy because of delayed diagnosis. This is because the symptomatology of allergic gastritis is unspecific, and therefore, hematemesis should be investigated properly. This includes investigations such as the provoking test of hydrolyzed milk, endoscopy of the GI tract, and histological biopsy. Furthermore, this report highlights the importance of early and extensive diagnostics in preventing further damage and a worse prognosis for patients.



