Background
Littoral cell angioma (LCA) is a rare and unique clinicopathologic entity, which was firstly reported in 1991 by Falk et al. [1]. LCA is within the scope of primary vascular tumours of the spleen. The tumour originates from ‘littoral’ or ‘seashore’ cells lining the sinuses located in the splenic red pulp. Intriguingly, these cells express both endothelial (CD31, FVIII-related antigen) and histiocytic markers (CD68, lysozyme) which give its unique immunohistochemical phenotype [1].
The incidence and pathogenesis of LCA remain unknown. There is no gender or age predilection although most of the patients are diagnosed in their 50s [1]. More than half of the patients are asymptomatic. Nevertheless, 45% of these patients may suffer from abdominal pain, symptoms associated with hypersplenism, or constitutional symptoms such as fever of unknown origin [2]. LCA is usually revealed by imaging studies as an incidental finding. The radiologic features are not specific, but well-delineated nodular lesions consisting of anastomosing vascular channels can be detected [3,4]. Histopathological and immunohistochemical staining following splenectomy is mandatory to confirm the diagnosis [1]. Although a majority of cases had a benign clinical behaviour [1], some others were linked with various malignancies, gastrointestinal associations, or immune system dysfunction [5–7]. Based on these findings, it is generally advised to accept LCA as ‘a benign lesion with malignant potential’. Close clinical follow-up is recommended.
In this report, we would like to present a case with LCA who was diagnosed after open splenectomy and histopathological examinations, along with the current literature search.
Case Presentation
A 49-year-old non-smoker Turkish woman was admitted to the general surgery outpatient clinic with a 1-month history of intermittent upper abdominal pain. Her past medical history was remarkable for cerebral palsy and chronic constipation. The severity of constipation increased for the past 1 month. A significant weight loss (~10 kg) attributed to a 6-month period was noted by her relatives. The physical examination showed skin pallor and a distended, meteoric abdomen. Traube’s space was dull to percussion. There was no palpable lymphadenopathy. Haemogram revealed Hb = 8.9 g/dl (normal range: 12–15.1 g/dl), white blood cell (WBC) = 4.74 × 103/µl (normal range: 4–10 × 103/µl) and platelet (PLT) = 94 × 103/µl (normal range: 150–450 × 103/µl). Serum iron and ferritin levels were 22.4 µg/dl (50–175) and 6.3 ng/ml (13–150), respectively, which were indicative of an iron deficiency anaemia. On peripheral blood smear, red cell morphology showed anisopoikilocytosis, platelet number was in accordance with moderate thrombocytopenia and white cell differential count revealed moderate lymphopenia (20%, absolute lymphocyte count = 0.948 × 103/µl). Other routine serum chemistries were within normal levels. Her past medical history also revealed that she had been transfused four units of packed red blood cells before an orthopaedic surgery on June 2018.
The radiogram of the lungs and abdomen showed extensive meteorism at the upper quadrants due to enlarged bowel segments (Figure 1). A computerised tomography (CT) scan of the abdomen revealed a massively enlarged spleen (22 cm in length) with numerous nodular hypointense lesions (the largest one is 2 cm in diameter) scattered throughout the organ parenchyma. Furthermore, the CT scan showed hepatomegaly (24 cm in length) and extremely enlarged upper bowel loops in conjunction with extensive luminal gas distention.
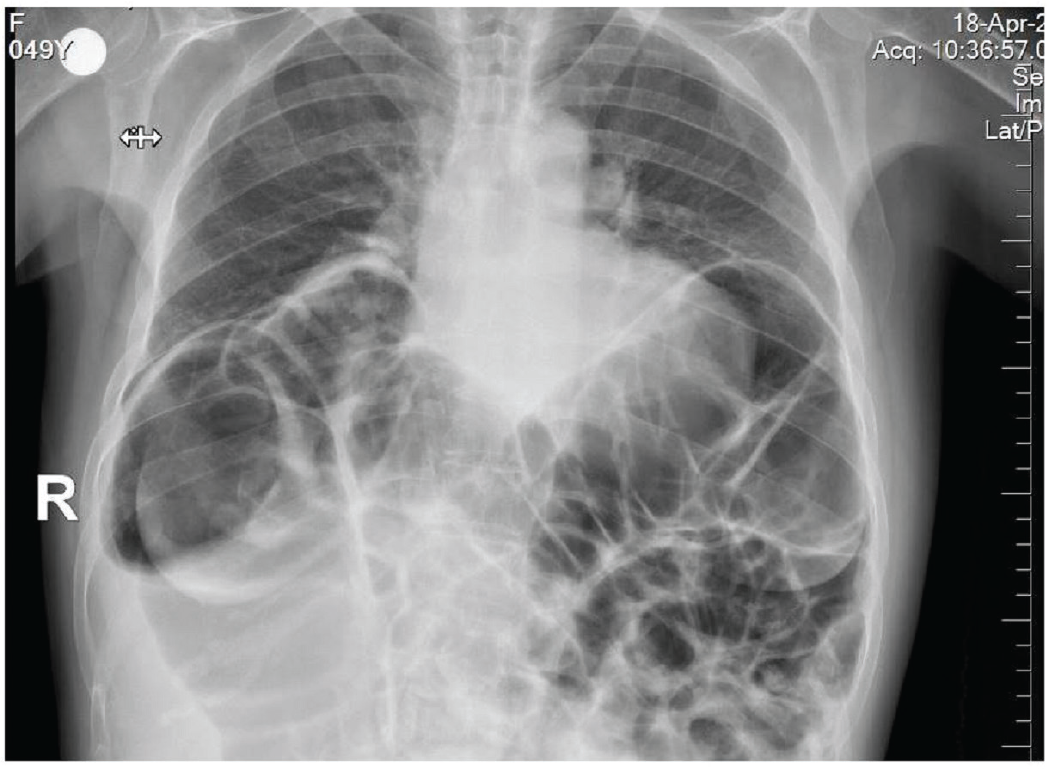
X-ray of the lungs and abdomen showed extensive meteorism at the upper quadrants of the abdomen due to enlarged bowel segments.
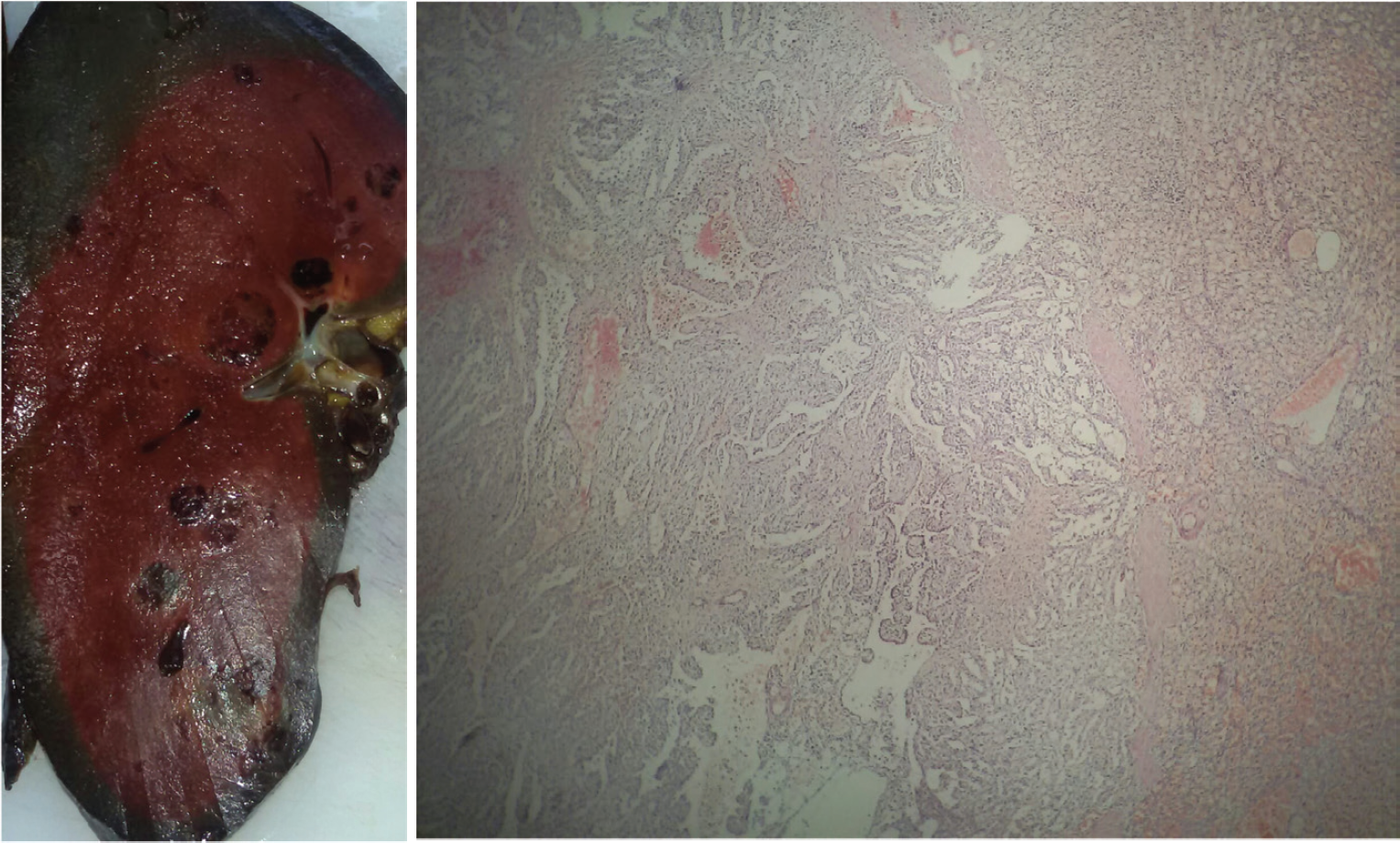
(A) Cut surface of the spleen revealed a dark maroon organ showing multiple well-delineated nodules without an evident surrounding capsule.These nodules are dark-red, dark maroon, or black (due to blood clots with different ages) with a maximum diameter of 2.0 cm × 2.2 cm located 0.8 cm away from the hilus. (B) The normal splenic parenchyma is replaced by cyst-like areas consisting of vascular anastomosing channels.
After these examinations, iron deficiency anaemia, mild thrombocytopenia, hepatomegaly, massive splenomegaly, multiple hypointense lesions in the spleen parenchyma and enlarged upper bowel segments were detected. To rule out possible infectious and malign aetiologies, extended laboratory tests were planned. Brucella agglutination with Coombs antisera, hepatitis screen, tumour markers including CA-125, CA-19-9 and carcinoembryonic antigen (CEA) and alpha-fetoprotein were found to be negative.
Despite the absence of lymphadenopathy, the provisional pre-operative diagnosis was lymphoma of the spleen. For diagnostic and therapeutic purposes, ‘total splenectomy and sigmoidectomy’ was jointly performed on this patient. The patient had an uneventful post-operative recovery and discharged on the 3rd day. Her Hb and PLT levels improved following splenectomy. The patient did not require any blood transfusion during the post-operative period. Control blood count at day 8 revealed a mild anaemia and thrombocytosis (Hb = 11.4 g/dl and PLT = 463 × 103/µl). Unfortunately, we have recently learned that, by a telephone call with her relative, she was expired due to sepsis in the university hospital after about 5 weeks from surgery.
Gross examination of the spleen revealed an 870-g greyish-brown nodular organ measuring 20 cm × 12 cm × 6 cm. The cut surface of the spleen showed well-circumscribed, locally cystic lesions throughout splenic parenchyma, which ranged from 0.7 cm to 2.2 cm in diameter (Figure 2A). The histopathologic evaluation identified multiple and anastomosing vascular channels lined by tall columnar endothelial cells. The lacunae were filled with oedematous fluid and blood (Figure 2B). There were no cytologic or nuclear abnormalities as well as mitotic figures. The histopathological findings were consistent with LCA. To confirm the histological diagnosis, immunohistochemical staining was also performed. Immunohistochemical staining was positive for both endothelial (FVIII-related antigen) and histiocytic (CD68) markers (Figure 3A and B), and CD34 was found to be negative as expected (Figure 3C). Based on both histologic and immunohistochemical findings, the patient was diagnosed as LCA.
On the other hand, the gross examination of sigmoid colon showed a 150 cm × 8 cm yellowish-brown and massively extended organ. Inside the bowel segment, effaced and greyish-coloured folds were observed. Histologically, the bowel specimen was negative for neoplasia and showed chronic inflammation.
Discussion
A 49-year-old female admitted to the hospital with abdominal pain, changes in bowel habits and significant weight loss. An abdominal CT scan revealed massive splenomegaly and multiple nodular lesions coinciding with enlarged upper bowel segments. In addition, the presence of anaemia and mild thrombocytopenia subsequently led us to perform an open splenectomy plus sigmoid colectomy, instead of a laparoscopic procedure. The histopathology revealed an incidental diagnosis of LCA (diffuse nodular form), along with a sigmoid volvulus. One week after surgery, she was in a very good condition without any symptoms. Thrombocytopenia was completely resolved, and Hb value was 11.4 g/dl.
Littoral cell angioma remains to be a rare clinic entity. Two forms of LCA have been defined: ‘diffuse nodular’ form and rarely seen ‘solitary’ form [8]. Most of the cases present with splenomegaly of unknown origin and diagnosed in their 50s’ post-splenectomy [1].
A definite diagnosis can only be made after histopathologic studies. To confirm the dual differentiation potential of littoral cells, an immunohistochemical panel, including both endothelial and histiocytic markers, is required. Littoral cells express FVIII, CD31, CD21, CD68, CD136, lysozyme and CD4, but they are negative for CD8, CD34, factor XIIIa, D2-40 and WT-1 [1,5]. Focal S100 positivity has also been reported [1]. Hodgkin and non-Hodgkin lymphomas should also be taken into consideration in the differential diagnosis of splenic tumours. Clinical, pathological and radiological features of LCA and lymphomas with splenic involvement are briefly shown in Table 1.
Diffuse nodular involvement of LCA makes total splenectomy a first-line treatment option. Open splenectomy or hand-assisted laparoscopic total splenectomy [9] is recommended for this purpose. In the selected cases, such as patients with a single and localised lesion, a partial splenectomy can also be preferred. To date, there has been only one report of partial splenectomy. A 58-year-old man who had 5.5-cm lesion was successfully treated with laparoscopic partial splenectomy [10]. This approach may enable to preserve immune functions of the spleen and, especially, be convenient for child or immunocompromised patients.
Based on the clinical case reports, the results of splenectomy are quite satisfactory [11,12]. The only mortal case after splenectomy was reported by Ben-Izhak et al. [13]. This patient had undergone splenectomy for a splenic tumour showing large areas of typical LCA. The splenic tumour recurred 8 years after with an abdominal mass and multiple hepatic metastases, resulting in the patient’s death. The patient died due to sepsis after 5 weeks following surgery. To prevent post-splenectomy infections, pre-operative vaccinations against Streptococcus pneumoniae, Haemophilus influenzae type B and Neisseria meningitidis should be administered.
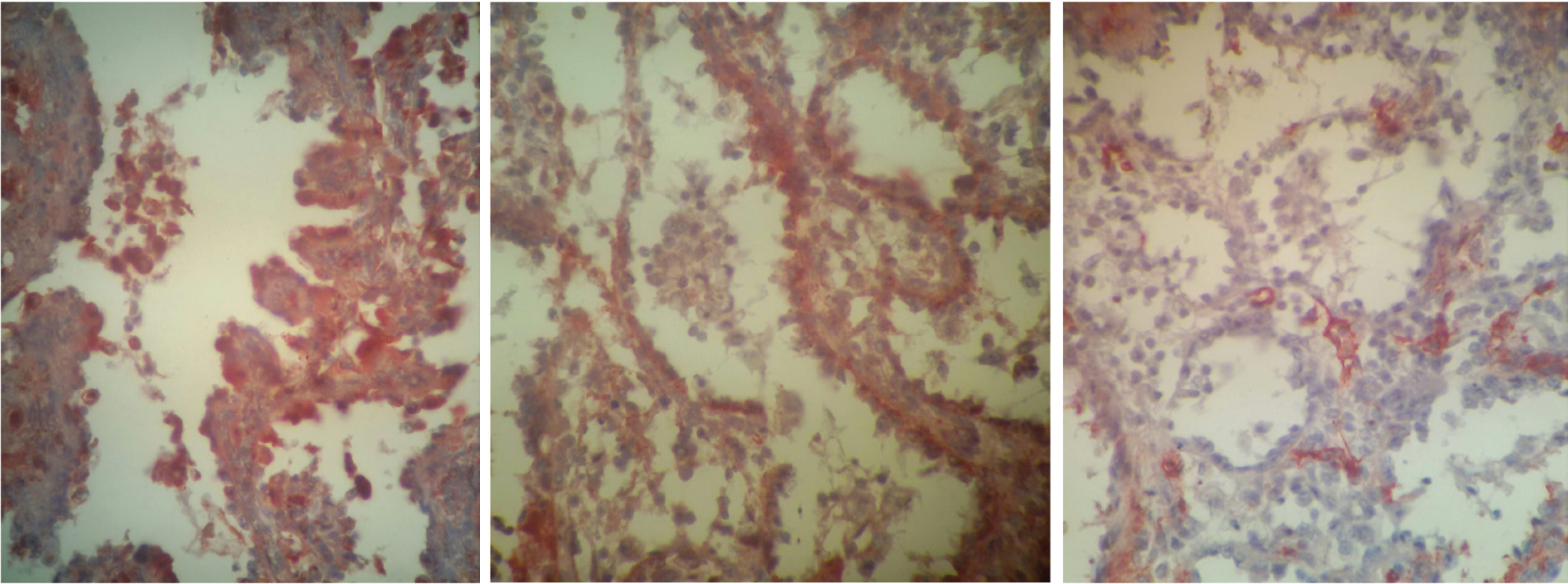
Immunohistochemical staining of splenic nodules including vascular anastomosing channels lined by tall endothelial cells (neoplastic littoral cells). (A) Littoral cells are strongly and diffusely positive for CD68 (CD68 immunohistochemistry, 200×), (B) these cells are strongly reactive to FVIII antigen (FVIII immunohistochemistry, 200×), (C) endothelium of the peripheral vessels are reactive to CD34 but not the littoral cells (CD34 immunohistochemistry, 200×).
| TYPE | CLINICAL FEATURES | PATHOLOGICAL FEATURES | RADIOLOGICAL FEATURES |
| LCA | Asymptomatic (>50% of cases), a benign lesion with malignant potential | Well-circumscribed nodules of anostomosing vascular channels lined by tall columnar endothelial cells | US: Hypoechoic to hyperechoic; CT: Iso to hypoattenuating with contrast enhacement; MRI: Low intensity lesions |
| Hodgkin lymphoma | Fever, night sweats, weight loss >10% (B symptoms) is noted. The spleen is rarely the primary site. A highly curable malignant disorder. | Characterized by typical giant cells with multiple or bilobed nuclei (Reed-Stenberg) | CT: Hypodense nodules with nodular sclerosis (in nodular sclerosis subtype) |
| Non-Hodgkin lymphoma | Fever, night sweats, weight loss >10% (B symptoms) are common. Splenic marginal zone lymphoma can present with massive splenomegaly. | Derived from B or T cells, lymphoproliferative disorder | CT: Hypodense nodules; MRI: Isotense on precontrast images, hypotense on postcontrast images |
Abbreviations: LCA: Littoral cell angioma; US: Ultrasonography; CT: Computed tomography; MRI: Magnetic resonance imaging.
LCA may also be the underlying cause of long-standing anaemia [11], including resistant iron-deficient type [12]. In most cases, hypersplenism is the leading cause of anaemia. Very rarely, the persistence of hypochromia and microcytosis (despite an appropriate iron deficiency treatment) may also be a predictive sign of LCA [12]. The case was admitted to our hospital with grade 2 (moderate) anaemia associated with iron deficiency, and her anamnesis revealed four units of packed blood cell transfusion history. Total splenectomy resulted with a significant increment in Hb value. The improvement of anaemia in this patient without any additional therapy is probably due to the removal of the enlarged spleen rather than iron deficiency.
A considerable amount of patients with LCA (31.1%) had second primary cancer [6]. More recently, a clinicopathologic study with extended follow-ups (mean: 11.6 years) has shown that 60% of patients with LCA had concomitant visceral malignancies, including mesenchymal, epithelial and hematologic [5] origin. Massive splenomegaly (>1,500 g), which correlated with a pre-operative spleen size of 20 cm, was significantly associated with the presence of malignity [14]. Given the malignant potential of these vascular lesions, close clinical follow-up is recommended to detect synchronous visceral or metastatic tumours. In the present case, despite a preoperatively large size of spleen (20 cm × 12 cm × 6 cm), no synchronous tumour was detected.
Volvulus is the rotate of a colon segment around its blood supply, whereas sigmoid volvulus is the most common type. From the clinical point of view, chronic constipation or immobilisation may cause sigmoid volvulus as was seen in the patient [15]. This report is the first case of LCA together with sigmoid volvulus although this association is likely to be entirely coincidental.
Conclusion
Clinicians should be aware of this rare clinicopathologic entity whilst evaluating the differential diagnosis of splenomegaly with solitary or multiple nodules. Malignant potential of LCA renders clinical follow-up mandatory.
What is new?
Littoral cell angioma (LCA) is a very rare clinicopathologic entity. Patients with LCA often present with splenomegaly and the laboratory signs of hypersplenism, including anaemia and/or thrombocytopenia. The radiologic appearance of splenic nodular lesions is nonspecific. Histologic and immunohistochemical examination of splenic specimens is mandatory for correct diagnosis. The case was diagnosed as LCA and sigmoid volvulus after open splenectomy, along with sigmoidoscopy. To the best of our knowledge, this coincidence has not been reported yet. We would like to share our experience and emphasise this rare disorder in the differential diagnosis of splenomegaly with multiple nodular lesions.

