Introduction
Linezolid is the first new synthetic oxazolidinone with a spectrum of activity against a variety of organisms including the multidrug-resistant microbes like methicillin resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus spp (VRE) and multi-drug resistant (MDR) Mycobacterium tuberculosis [1]. Optic neuropathy has been reported previously as an adverse effect in patients using linezolid [2-7].
Here we report a case of 28 years old lady receiving treatment with linezolid for extensively drug resistant tuberculosis (XDR-TB) with reversible bilateral optic neuropathy.
Case presentation
A 28-year-old lady presented with painless, progressive diminution of vision in both eyes for previous two months. This was associated with insidious onset reduced sensation of both feet and ankle. Medical history included long standing XDR-TB under multiple drugs. Current medications included pyrazinamide, moxifloxacin, ethionamide, para-aminosalicylic (PAS) granules, linezolid, amoxicillin and clavulanic acid. She had been on 600 mg of oral linezolid once a day for the previous 14 months when she presented to us.
On presentation, best corrected visual acuity (BCVA) in both eyes was 6/60. Color vision recorded with Farnsworth Munsell D15 test showed protanopia. Contrast sensitivity was reduced in both eyes when recorded with Pelli-Robson chart (1.03). Visual field test done with Humphrey visual field analyzer revealed bilateral central scotoma (figure 1 and 2). Pupils were round and briskly reactive to light with no relative afferent pupillary defect. The anterior segment examination was unremarkable. Fundus evaluation after pharmacological mydriasis demonstrated bilateral, symmetrically swollen optic discs with hyperemia and peripapillary retinal nerve fiber layer (PRNFL) edema (figure 3 and 4). Optical coherence tomography (OCT) of optic nerve head revealed increased retinal nerve fiber layer thickness in both eyes (figure 5).
A clinical diagnosis of linezolid induced optic neuropathy (LION) was established. Linezolid treatment was discontinued and dose of ethionamide was optimized after discussing with the treating physician.
One week later, BCVA improved to 6/18 in both eyes. Though color vision and contrast sensitivity testing revealed no changes compared to initial findings, she could experience subjective improvement in brightness in both eyes. Fundus examination also revealed that the optic disc edema was resolving (figure 6 and 7).
Five weeks after discontinuation of treatment, BCVA improved to 6/6 in both eyes. Color vision was normal when tested with both Ishihara color plates and D-15 test. Contrast sensitivity testing with Pelli-Robson chart showed normal contrast sensitivity. Visual field analysis revealed decreased size of central scotoma (figure 8 and 9). Examination of optic discs showed markedly decreased optic disc hyperemia, absent optic disc swelling and reduced PRNFL swelling (figure 10 and 11). PRNFL scan showed decrease in 360˚ average retinal nerve fiber layer thickness measurement [40 μm in the right eye (RE) and 45 μm in the left eye (LE)]. Marked decrease in thickness were detected in temporal (18 μm RE and 43 μm LE), nasal (16 μm RE and 17 μm LE), superior (57 μm RE and 73 μm LE) and inferior (53 μm RE and 48 μm LE) quadrant (figure 12).
Causality assessment of the adverse drug reaction
In the last decade, there has been an increase in the reporting of adverse drug reactions (ADRs). Policy makers have been focusing on a robust pharmacovigilance system for the provision of safer drugs, however, causality assessment has been the greater challenge for academicians and even industry [8].
Causality assessment has now become a common routine procedure in pharmacovigilance. The Naranjo probability scale is one of the generally accepted and most widely used methods for causality assessment in clinical practice as it offers a simple methodology [9]. The scale is shown in table 1.
Adverse reactions are rarely specific for a drug. In practice, few adverse reactions are ‘certain’ or ‘unlikely’ due to lack of specific diagnostic tests and a rechallenge being rarely ethically justified. Most ADRs are therefore somewhere in between these extremes, i.e. ‘possible’ or ‘probable’ [10].
Structured causality assessment methods for individual cases have been used to facilitate case processing rather than to reach definitive conclusions. However, individual case reports may also provide compelling evidence of causality even though definitive decisions about causality (leading to risk management plan/label changes etc.) usually incorporate evidence from various sources [8].
Here we assessed the likelihood of linezolid causing this case of optic neuritis.
| Score | Yes | No | Don’t Know | |
| 1. | Are there previous conclusive reports on this reaction? | +1 | 0 | 0 |
| 2. | Did the adverse event appear after the suspected drug was administered? | +2 | −1 | 0 |
| 3. | Did the adverse reaction improve when the drug discontinued or a specific antagonist was administered? | +1 | 0 | 0 |
| 4. | Did the adverse drug reaction reappear when the drug was re-administered? | +2 | −1 | 0 |
| 5. | Are there alternative causes (other than the drug) that could solely have caused the reaction? | −1 | +2 | 0 |
| 6. | Did the reaction reappear when a placebo was given? | −1 | +1 | 0 |
| 7. | Was the drug detected in the blood (or other fluids) in a concentration known to be toxic? | +1 | 0 | 0 |
| 8. | Was the reaction more severe when the dose was increased, or less severe when the dose was decreased? | +1 | 0 | 0 |
| 9. | Did the patient have a similar reaction to the same or similar drugs in any previous exposure? | +1 | 0 | 0 |
| 10. | Was the adverse event confirmed by objective evidence? | +1 | 0 | 0 |
Definite ≥ 9, Probable 5-8, Possible 1-4, Unlikely ≤ 0
We have used this tool for causality assessment in our case i.e. to assess the likelihood of linezolid causing LION, the result of which is shown in the table below
| Score | Yes | No | Don’t Know |
| 11. Are there previous conclusive reports on this reaction? | +1 | ||
| 12. Did the adverse event appear after the suspected drug was administered? | +2 | ||
| 13. Did the adverse reaction improve when the drug discontinued or a specific antagonist was administered? | +1 | ||
| 14. Did the adverse drug reaction reappear when the drug was re-administered? | 0 | ||
| 15. Are there alternative causes (other than the drug) that could solely have caused the reaction? | +2 | ||
| 16. Did the reaction reappear when a placebo was given? | 0 | ||
| 17. Was the drug detected in the blood (or other fluids) in a concentration known to be toxic? | 0 | ||
| 18. Was the reaction more severe when the dose was increased, or less severe when the dose was decreased? | 0 | ||
| 19. Did the patient have a similar reaction to the same or similar drugs in any previous exposure? | 0 | ||
| 20. Was the adverse event confirmed by objective evidence? | +1 |
Hence, the score came to be 7 i.e. the adverse drug reaction (ADR) is probably caused by linezolid. However, we have not done the rechallenge test due to ethical reasons and had it been done, it might have shown a positive result and hence the score would have been 9 i.e. it would have been a definite case of ADR. Some pharmacologist also choose to give a positive score for this point irrespective of the rechallenge test when there is no any alternative etiologies to the reaction and where other factors suggest a strong causal association and the adverse effect is a well-known effect of a drug [9]. We also failed to detect the blood levels of linezolid due to lack of specific test which could have further strengthened the causality .
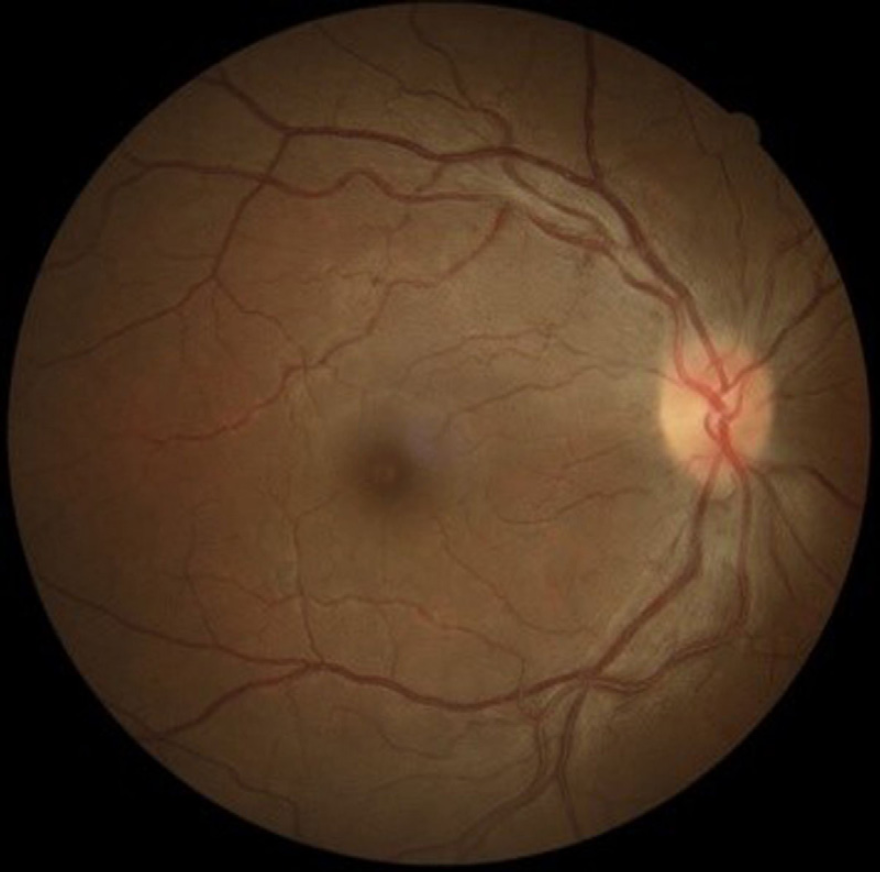
Fundus photographs showing resolving disc edema in right eye one week after discontinuing linezolid.
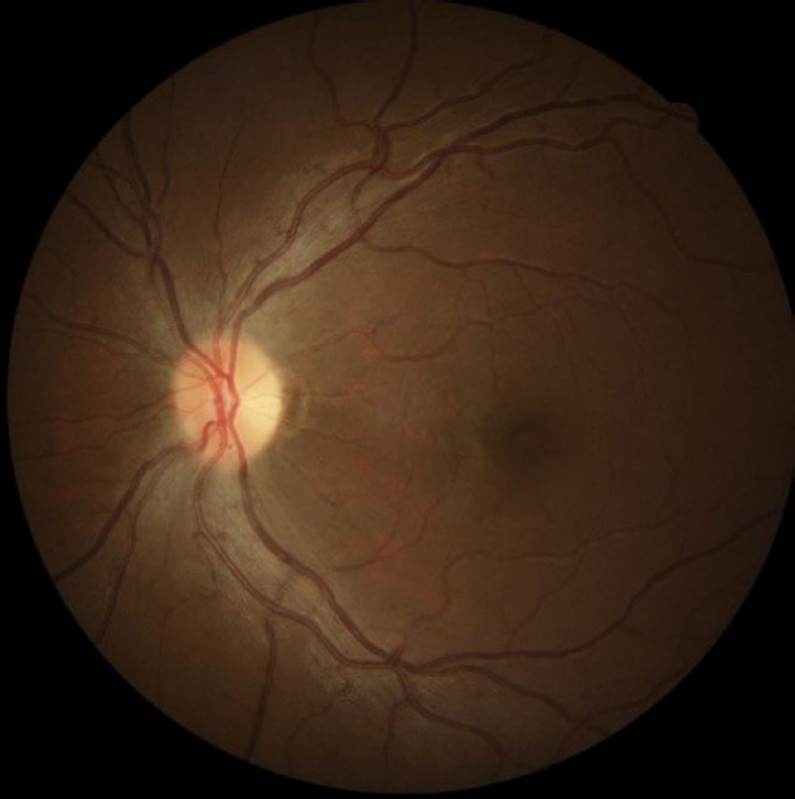
Fundus photographs showing resolving disc edema in left eye one week after discontinuing linezolid.
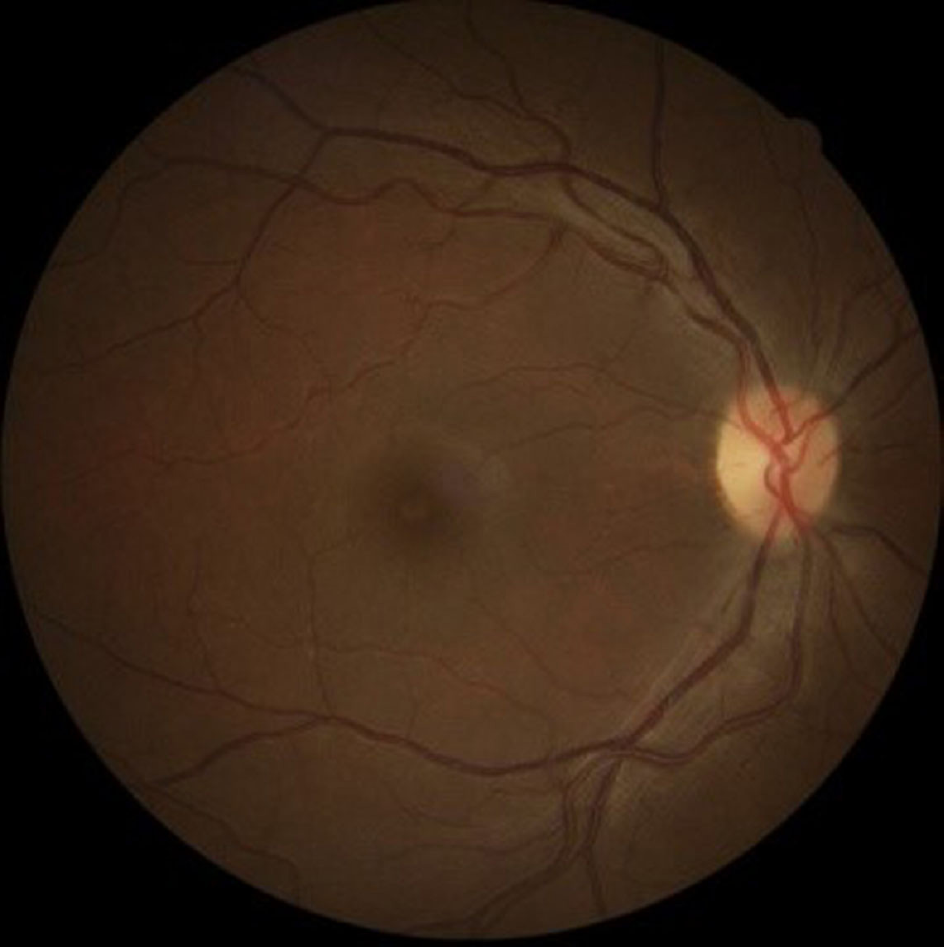
Fundus photographs showing resolved optic disc edema in right eye five weeks after discontinuing linezolid.
Discussion
XDR-TB is caused by Mycobacterium tuberculosis which is resistant to isoniazid and rifampicin along with further resistance to any fluoroquinolones and at least one injectable second-line drug (amikacin, capreomycin or kanamycin).
Linezolid, the first new synthetic oxazolidinone, acts through inhibiting bacterial protein synthesis by binding to bacterial rRNA, specifically to the 23S rRNA of the 50S ribosomal subunit, and thereby inhibiting the formation of the protein synthesis initiation complex. [3] It has a spectrum of activity against a variety of multidrug-resistant organisms including MRSA, VRE and MDR Mycobacterium tuberculosis.
Linezolid in XDR-TB is highly efficacious. A systematic review based on data from various studies done globally showed treatment success rate of linezolid in MDR cases over 80% [11].
Linezolid is a well-tolerated and relatively safe drug with a recommended duration of therapy for no more than 28 days. [12,13] Prolonged use outside the recommended safe period is often undertaken but has previously been associated with optic and peripheral neuropathies. [2,4-7] In this case the patient had already been on linezolid 600 mg once daily for fourteen months before she developed symptoms due to its toxicity.
A systematic review and meta-analysis of cases of MDR-TB revealed toxic optic neuropathy as an ADR in 13.2% of patients using linezolid exceeding 600mg per day for a mean duration of 9 months [11]. The ribosomal similarity between the mitochondria in mammalian nerve cell and that of bacteria is the basis of neurological adverse effects in human [3].
Our case was a young lady who presented with progressive loss of vision in both eyes. She had been on multiple second line anti-tubercular drugs along with linezolid for over a year. Tests including color vision, contrast sensitivity, visual field and optical coherence tomography (OCT) supported the diagnosis of toxic optic neuropathy.
With the use of a causality assessment tool in our case, the diagnosis of toxic optic neuropathy seemed even more likely and hence discontinuing the inflicting drug appeared rational.
With increasing use of linezolid, optic neuropathy as its specific side effect is increasingly being reported.
Though many cases of LION have been reported, none have used a causality assessment tool like in our study to evaluate the probability of linezolid as an inflicting agent for optic neuropathy.
In a developing country like Nepal where infectious diseases such as tuberculosis are still a major problem, awareness among ophthalmologists and physicians regarding visual monitoring in patients receiving long-term linezolid therapy is important. This is because early identification of toxicity of this drug and its discontinuation can result in complete visual recovery.
Conclusion
Ophthalmologists and physicians should be aware of the ocular complications associated with prolonged linezolid therapy. This is a potentially reversible adverse effect if picked up early. Use of causality tool like the one used in our case can provide additional support in establishing diagnosis and thus timely intervention is possible.










