Background
Angiofibromas are the benign tumors of the blood vessels. The entity was named as angiofibroma first by Friedberg in 1940.This accounts for 0.5% of all the head and neck tumors and incidence is 1 among 150,000 individuals [1]. JNA was exclusively found in males in the second decade. This may not cause any symptoms for 6-12 months before the diagnosis is made. Extra nasalangiofibromas occur in elderly females and are comparatively less aggressive [2].
Case presentation
Case 1:
A 14 year old boy reported with complaint of slight swelling in right maxillary and orbital region of four months duration. There was no history of trauma or fever. There were complaints of right nostril obstruction and mild headache but no bleeding from the nose. On examination, the boy was of average built with slight prominence of the right malar region. The overlying skin was of normal coloration. Anterior rhinoscopy showed blockage of the right nostril by pinkish lobulated mass not adherent to the opposing surface. Posterior rhinoscopy revealed the mass occupying the right nostril with extension to the posterior nasopharyngeal space. Other systemic examination was unremarkable. Biochemical parameters were within normal limits. Plain X-ray of the neck with face had shown the soft tissue density opacity in the nasopharyngeal region (Figure 1).
The boy was subjected to plain and contrast computerized tomography after proper written consent. There was enhancing mass in the right nostril region extending postero laterally to the nasopharyngeal space. There was no destruction of bone but remodeling was noticed (Figure 2a,b,c, 3a,b and 4a,b).
The patient was finally diagnosed with that of nasopharyngeal angiofibroma stage IIb as per the NCCT and CECT findings with the background of clinical setting. Now the patient had been kept under observation and surgical intervention will be decided soon as the patient is not much symptomatic.
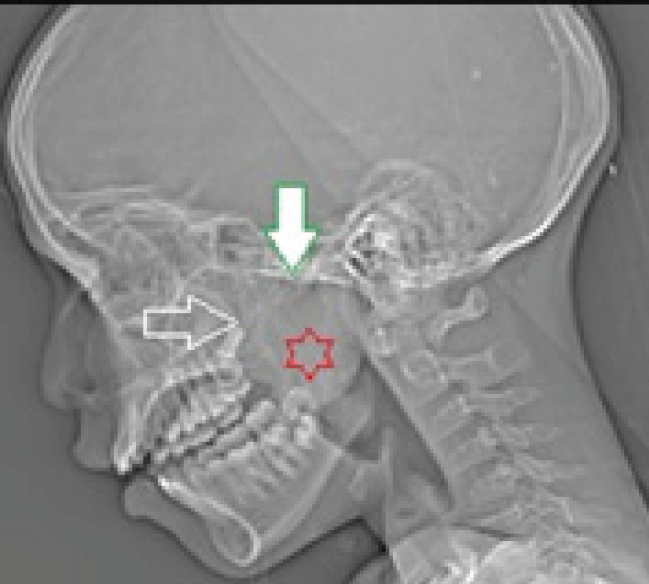
Plain X-ray lateral view of face and neck region.There is soft tissue density opacity in the nasopharynx (red star).Superior margins of the mass are retained by sphenoid sinus and basiocciput (white inverted arrow).

Non-contrast computerized tomography (NCCT) of the facial region axial sections. (a) there is an expansion of right nostril passage (blue arrow) and pterygomaxillary fossa(blue star) displacing the nasal septum to contralateral side. (b) inferior extension of the mass in nasopharyngeal space (blue star). (c) the bone window shows expansion of right nasal passage (red star) and pterygomaxillary fossa (green star) without bony destruction.

CECT facial region (a) axial section shows intense enhancement of the mass in the right nostril (red star) and in pterygomaxillary fossa (blue arrow).Right orbit shows slight prominence because of the mass expansion (white arrow). (b) sagittal reformatted image shows the superior extent of the mass (blue arrow) along with inferior extent (red star). (c) coronal reformat image shows enhancement of the mass extending from the medial aspect in the right nostril (red star) laterally to the pterygomaxillary fossa (white arrow) without involving infratemporal fossa (black arrow).
Case 2:
A 14 year old boy reported with the complaint of frequent episodes of epistaxis of one year duration. He also had complaint of headache of the same duration. There was slight protrusion of right malar prominence. He also had a history of recurrent episodes of sinusitis and right side nasal obstruction. On anteriorrhinoscopy there was a dull pinkish mass in the right nostril on the posterior aspect. Posteriorrhinoscopy had revealed the same mass going superiorly and inferiorly but was of more pinkish in coloration. Rest of the systemic examination was unremarkable. Blood investigations were normal except Hbas 9.5 g/dl. Plain lateral skiagram of the face showed the soft tissue density opacity in the nasopharyngeal space (Figure 5).
He underwent NCCT and CECT of the face for the nasopharynx and sinuses. There was intense enhancing mass in the right nostril extending to the pterygomaxillary and infratemporal fossa (Figures 6 a,b,c, 7a,b,c, 8a,b,c and 9).
The patient was diagnosed as that of nasopharyngeal angiofibroma stage II-c with the strong clinical setting. He had been planned for endoscopic surgical resection.

Plain X-ray of the head and neck region for nasopharynx. There is soft tissue density opacity in the nasopharynx (red arrow) with intact sphenoid sinus and bone (white arrow) and obstructing the nasal passage anteriorly (green arrow).

NCCT facial region axial sections. (a) soft tissue mass in the right nostril (blue arrow) and similar density in the right pterygomaxillary fossa (blue star). (b) section at slightly lower level shows the inferior extension of the mass (blue star). (c) The bone window does not reveal any bony destruction in the nasal passage (red star) and in the pterygomaxillary fossa (green star).
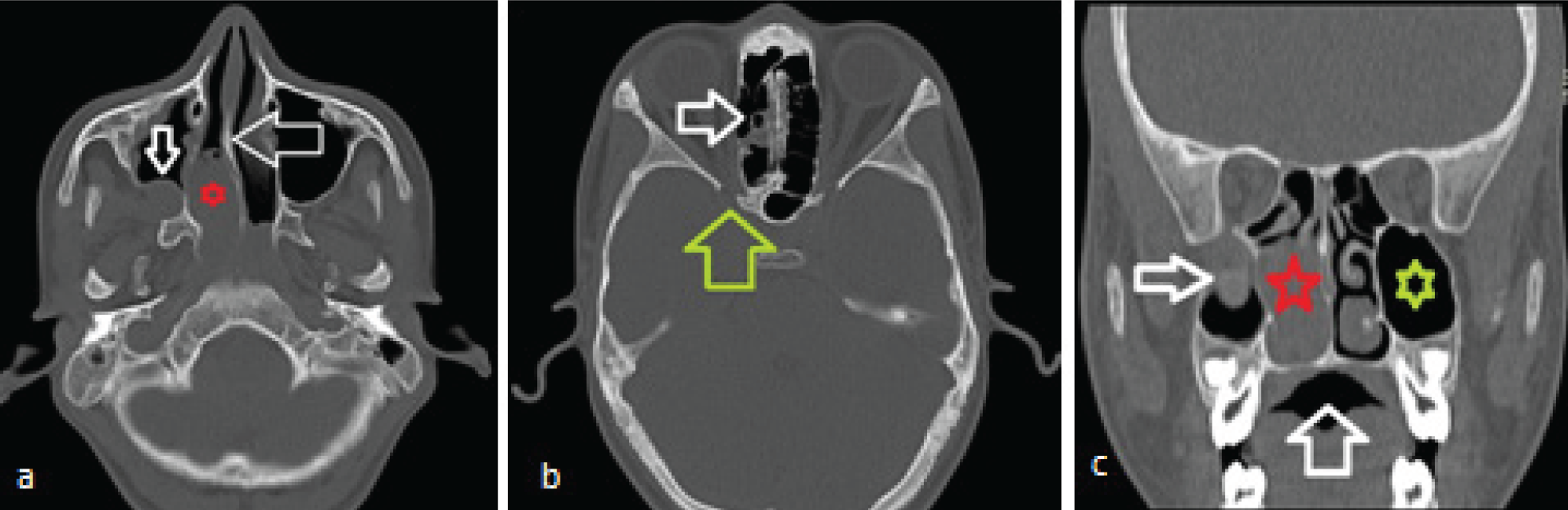
NCCT bone window. (a) axialsection shows the mass (red star) causing expansion without damaging the nasal septum (white horizontal arrow) and the posterior wall of the right maxillary sinus (white inverted arrow). (b) higher axial section shows partially opacified ethmoid air cells without destruction (white arrow) and with intact middle cranial fossa boundary (green arrow). (c) coronal reformat image shows the bony boundaries of the nasal mass (red star) with lateral extension (white arrow) with normal left maxillary sinus (green star).

CECT of nasopharynx show avid enhancing mass lesion. (a) axial ion shows the enhancing mass on the posterior aspect of the nostril (blue arrow) with partly non-obstructive anterior passage (arrow head). (b) sagittal reformat image shows the extent of enhancing mass from the basiocciput to the inferior margin (blue arrow). (c) coronal reformat image shows the same mass extending laterally towards the pterygomaxillary and infra-temporal fossa compromising the right maxillary sinus (blue arrow).
Discussion
JNA originates from the superior margin of spheno-palatine fossa and posterior part of the middle turbinate. The knowledge of the basic anatomy of the nasopharynx is very important to know the origin as well as expansion and extension of the tumor to various directions (Figure 10).
Though the exact etiology of JNA is uncertain but hormonal factors play a role in these tumors. Marten et al proposed this hormonal theory first time. This was because of decrease in androgen levels and increased activity of estrogen components. The nasal mucosa is having more hormonal receptors at the regions mentioned above with additional vascular endothelial growth factors (VEGF) responsible for the outcome [3]. Another theory of desmoplastic response of nasopharyngeal periosteum has also been suggested. Another version of nonchromafin paraganglionic cells of terminal branches of maxillary artery has been suggested. Girgis and Fahmy observed “Zell ballen” cell nests at the growing edge of the tumor making it similar to paraganglionoma. But the most accepted theory has been from the origin of sex steroid stimulus hamartomatous tissue. The tumor originates from the sphenopalatine foramen and extends to nasopharynx and pterygopalatine fossa. These are also called as bilobed tumors because of the pattern of their extension. It can extend superiorly to the sphenoid sinus and can invade the cavernous sinus. Around 10-20% of the tumor shows intracranial extension. Following are presenting features of the patients:
Nasal (80%) and orbital masses (15%) with proptosis (10-15%) are frequently encountered. Nasal obstruction frequently leads to the accumulation of the secretions leading to sepsis responsible for hyposmia or anosmia. Other associated features may develop depending upon the regional involvement like deafness, headache, diplopia and otalgia. These masses required to be differentiated from other pathologies causing nasal obstruction like polyps, teratoma, dermoid and encephalocele. Other factors responsible for epistaxis have to be ruled out. Sessions et al has categorized the tumor into the following three stages as per the sub mucosal extension [4,5]:
Stage I:
1-a: limited to the nasopharynx.
1-b: when there is an extension to one or more paranasal sinuses.
Stage II:
II-a: minimal extension to pterygomaxillary fossa through sphenopalatine foramina.
II-b: bowing of posterior wall of maxillary antrum (Holman-Miller sign) or extension to the orbit through inferior orbital fissure.
II-c: extension into infratemporal fossa (modified by Radomski)
Stage III: calabash like tumor
III-a: with minimal Intracranial extension.
III-b: extensive intracranial extension with or without cavernous sinus invasion.
Plain X-ray reveals the opacity because of the soft tissue mass. Computerized tomography (CT) is the best imaging modality to know the involvement of the bone with the extension of the tumor mass. The mass is well defined, non-encapsulated and lobulated with epicenter around sphenopalatine foramina. There is intense enhancement seen because of the high vascular contents [6,7]. There is resorption or remodeling of the bone rather than the destruction which makes it different from aggressive and malignant pathologies. The characteristic appearance of JNA may not warrant any biopsy confirmation which itself is not safe. Magnetic resonance imaging (MRI) is also helpful in tissue characterization and evaluation of the extent of the tumor. Pathologically, these are nodular masses which are white in color but the mucosal covered region is pink in coloration. Though there is no capsule but these are well demarcated from the surrounding tissue. This also suggests for first separating the mass and then to be removed surgically to avoid bleeding during surgery. Microscopically, these tumors are composed of fibrocellular stroma with spindle-shaped cells in interspersed collagen with irregular vascular pattern. Vascular component is more near the origin and has got two types of vessels, one without the muscular coat and other with thick muscular media. The vessels without muscular coats are responsible for bleeding as these fail to constrict. It is covered with squamous epithelium with underneath mucous glands along with infiltrates. Fibrous element increases with the age [8]. Treatment involves surgery, irradiation or hormonal therapy depending upon the staging and involvement. Spontaneous regression [9, 10] has been noticed in individuals who are > 20 years old. Surgical management [11, 12] is the primary treatment which is required in most of the cases. There is 46% recurrence rate after surgical excision. Angiography is helpful to find the feeder vessel and to embolize it 24-72 hours before surgery to minimize the bleeding [13]. Radiotherapy (30-46 Gy) is indicated in non-operable and recurring cases. This induces radiation vasculitis leading to many side effects. Small doses are ineffective and the large dose can cause damage to the healthy tissues. This also makes the surgery difficult if required later on [14]. Adjunctive hormonal therapy has been tried which block the testosterone receptors [15]. The tumor course may have to be watched with repeat imaging study in case there are signs of regression. The incidence of recurrence is high where there is antero-posterior extension.
Conclusion
JNA are the locally aggressive tumors which require complete work up before the decision of the management strategy. This can lead to great morbidity and mortality because of their aggressive nature and tendency to bleed. The correct staging is of paramount importance as there is great danger of the bleeding and residual tumors. CECT plays important role in staging. Pre-operative angiography is always helpful to minimize the bleeding during surgery.




