BACKGROUND
Gastritis Cystica Profunda (GCP) is hyperplasia and cystic dilatation of the gastric glands, which extend into the submucosa. The first case was reported by Littler and Gleibermann in 1972.(1,2) The pathogenesis of GCP is hypothesized to be a trigger event resulting in injury to the muscularis mucosa. Trigger events are inciting factors such as prolonged chronic inflammation (Helicobacter pylori associated), ischaemia, previous gastric surgery or suture material. However, it can occur in patients without a history of gastric surgery. The injury to the muscularis mucosa thus initiates the entrapment of the gastric glands in the submucosa.(1) Mucosal prolapse and bile reflux in patients that have undergone gastric surgery are factors that propagate the cystic changes within the mucosa.(3) There are reports on the association of H. pylori as a risk factor for GCP, gastric cancer and gastrointestinal stromal tumour (GIST).(1,4,5)
Clinical presentation varies from non-specific abdominal pain, gastric outlet obstruction, gastrointestinal bleeding, anaemia, acid reflux and abdominal masses.(6,7) Oesophagastroduodenoscopy and endoscopic ultrasound (EUS) are imaging modalities of choice. Generally presenting as a submucosal growth, it has multiple differentials including malignant conditions such as GIST.(1,7,8) Due to its varied presentations endoscopically and the potential for malignancy as a differential, a definitive diagnosis of GCP can only be made on a histopathological specimen.(1) The pitfall with a preoperative histopathological diagnosis is the difficulty in obtaining a representative sample of the submucosa endoscopically and thus many patients undergo multiple invasive procedures for a mostly benign disease.(1)
CASE PRESENTATION
A 45-year-old female with no known co-morbidities or prior surgery presented with halitosis and gastroesophageal reflux disease. There was no associated loss of weight. At gastroduodenoscopy, a prepyloric submucosal mass was found measuring approximately 1.5 cm in diameter. It was smooth and elevated, clinically suspicious of a GIST. Biopsies of the lesion revealed gastric mucosa with a heavy load of H. pylori; however, no submucosa was found in the specimen. Repeat endoscopy and biopsies were performed 2 and 8 months later, which showed similar findings except that there was an increase in the size of the lesion and an absence of H. pylori and again no submucosa was represented in the specimen.
After three unsuccessful gastroduodenoscopy biopsies of the suspicious lesion, an EUS-guided biopsy was the next step in this diagnostic enigma. Thus, the patient subsequently underwent an EUS at a neighbouring academic hospital. The lesion was described as a 2.6×3 cm pedunculated mass in the antrum with central umbilication macroscopically. EUS showed a well-circumscribed hypoechoic mass in the antrum, located within the muscularis propria. Macroscopically, it was suspicious for a GIST, and an endoscopic resection was suggested. A fine needle aspirate (FNA) of the lesion performed at the time of EUS revealed groups of spindle cells, which were in keeping with a GIST. However, immunohistochemistry could not be performed on the sample at that time and thus other spindle cell lesions could not be excluded.
As previous gastroduodenoscopic biopsies and an EUS-guided FNA failed in clarifying the diagnosis, the patient was counselled on surgical excision. A preoperative CT scan of the abdomen revealed a thickening of the pylorus and no features of metastatic disease.
A month after the EUS study, the patient underwent a laparoscopic excision of the prepyloric lesion. However, due to difficulty in localizing the lesion, the procedure was converted to an open laparotomy. Intra-operatively, a 2 cm antral lesion was found. Excision of the lesion and a pyloroplasty was performed. All layers of the stomach were included in the surgically excised specimen. Features were in keeping with GCP in the background of mild chronic gastritis. Figures 1–4 demonstrate the histopathological findings. Six monthly follow-up visits after the surgery have demonstrated no recurrence of symptoms. The patient is scheduled for yearly endoscopy for screening for malignancy.
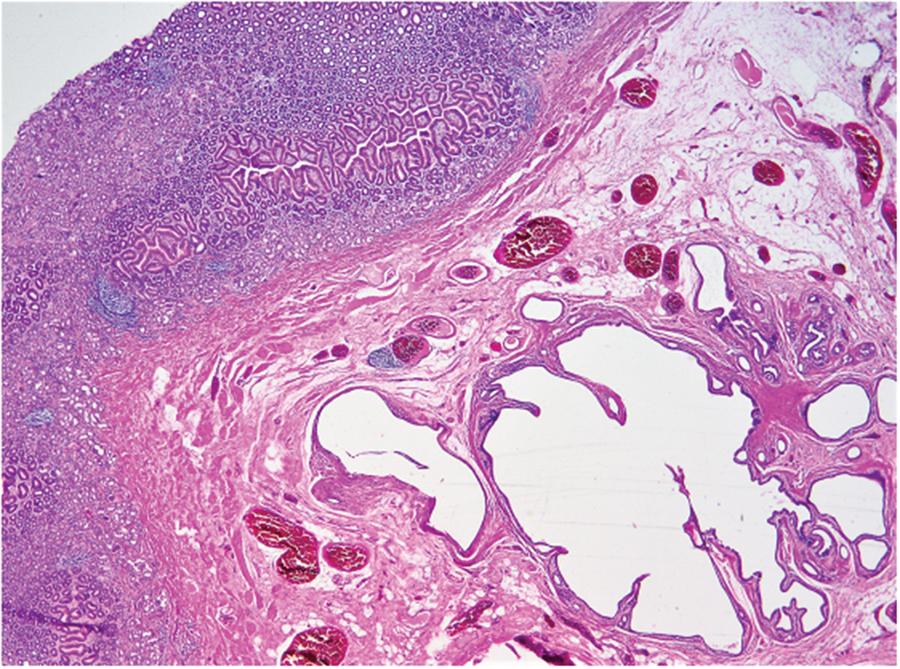
Low power view showing gastric mucosa and submucosa. Dilated epithelial-lined cystic spaces are present in the submucosa
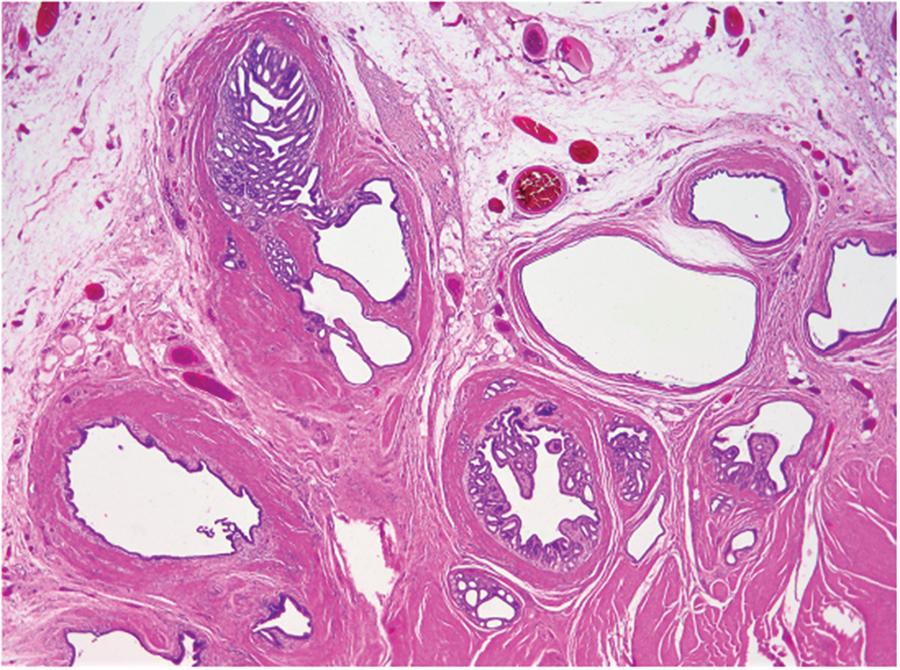
Low power view showing submucosa and muscularis propria. Epithelial-lined cystic spaces are present in the submucosa, extending into the muscularis propria
DISCUSSION
As noted from the literature and as the current case demonstrates, submucosal lesions pose a significant diagnostic dilemma. They often entail numerous invasive diagnostic procedures and at times unnecessary surgical intervention.(3,6) Our patient underwent four endoscopies with a biopsy each time and preoperative definitive diagnosis still could not be made. Each biopsy was not representative of the submucosa. Pre-operative marking of the lesion endoscopically could have facilitated a laparoscopic approach; however, due to the diagnostic dilemma, an open approach was justified for the advantage of tactile localization. Much of the literature available is based on case reports; hence, recurrence and long-term follow-up have not been adequately evaluated.(1–3,6–11)
Laratta et al. reviewed all published case reports on GCP from 1972 to 2011.(3) They found 37 cases, with a male:female ratio of 3, 6:1, with a mean age of 60.5 years. Of the published cases, 65% had a previous history of gastric surgery. Most commonly, patients present with abdominal pain (27%) followed by bleeding/anaemia (16%), sensation of fullness (8%) and anorexia/weight loss (8%). GCP was frequently found in the body of the stomach (62%) followed by the fundus (24%), antrum (8%) and the least frequent in the cardia (6%). The most common intervention was surgical resection (73%) followed by endomucosal resection (18%) and lastly polypectomies (4.5%). The malignant potential of GCP is unknown.(3)
Due to the diagnostic dilemma, many patients need to undergo invasive surgery to obtain a diagnosis of GCP. Many of the recent GCP case reports have suggested use of endoscopic, laparoscopic and open surgical techniques to obtain specimens when endoscopically obtained specimens were inconclusive.(1–3,6,7,9–11) H. pylori, which was found in this case, is not only associated with GCP but is also found in patients with GIST and gastric cancer.(1,4,5)
Thus, there is an obligation to eradicate H. pylori as it may potentially be a risk factor for malignancy in patients that are seropositive, as this closely approximates the true infection rate as opposed to a histological biopsy specimen.(4)
Less invasive surgery, namely, endoscopic techniques, should be highly considered when faced with a submucosal growth.(8) A retrospective study performed in a single institute in China of 34 histopathologically confirmed GCP cases showed that endoscopic submucosal dissection (ESD) and endoscopic mucosal resection (EMR) were able to achieve free margins in most cases, with shorter operating times and minimal intra-operative complications. All patients had no previous history of gastric surgery.(8) This centre achieved a complete resection rate of 92% in those undergoing ESD and 78% in those undergoing EMR.(8)
CONCLUSION
This case study highlights the diagnostic dilemma that is commonly associated with gastric submucosal lesions and the armamentarium of diagnostic and therapeutic options available. One should consider the use of EUS much earlier when a gastric submucosal lesion is suspected as its assists in characterizing the lesion and for retrieving a representative histopathological specimen.



