INTRODUCTION
The most common causes of changes in the immune response are stress factors that lead to redistributive reactions of blood leukocytes, changes in hormonal levels, and cytokine production. These can be factors of an infectious nature as well as different variations of physical or emotional stress. Even though neutrophilia is one of the classic manifestations of the stress response, the biological meaning of this phenomenon remains unclear. Research is carried out mainly in the direction of studying stress-induced changes in the functional activity of these cells [1,2]. However, we can assume that antimicrobial proteins that are secreted from neutrophils, particularly lactoferrin (LF), not only act as antibiotics but also can influence the development of stress and immune responses according to the principles of regulation of physiological reactions.
Experimental models simulating some aspects of the infectious process, such as inducing the changes in cytokine production by means of LPS administration, are often used to evaluate the immunomodulatory properties of lactoferrin. With the help of such models, it was shown that LF can inhibit the LPS-induced increase in TNFα, IL1β, and IL8 secretion by the human monocyte cell line THP-1 [3]. Experiments on mice have also demonstrated that LF has a protective effect against LPS-induced intestinal inflammatory injury [4]. The effect of stress on this aspect of the immunomodulatory action of LF has not been previously studied, although the combination of stress and infection is a reallife situation.
Previously, we found that LF exhibits stress-protective and immunomodulatory effects in an experimental model that combined emotional and physical stress in rats. Thus, it was shown that preventive LF administration significantly diminishes the stress-induced increase in both corticosterone concentration in the blood (observed 30 min after stress exposure) [5] and the Tlr4 gene expression in rat splenocytes (observed 3 h after stress exposure) [6].
In this study, we investigated the effect of LF on corticosterone level and on the expression of the IL4 cytokine gene and the TLR4 pattern-recognition receptor gene under stress and upon LPS administration in rats.
MATERIALS AND METHODS
Animals
Experiments were performed in male Wistar rats weighing 150-200 g. The animals were kept under vivarium conditions at room temperature with a 12-h light/dark cycle and free access to water and food (the standard diet in accordance with the current requirements for keeping laboratory animals (European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes, https://rm.coe.int/168007a67b).
Reagents
Human milk lactoferrin (purity ≥ 98%, iron saturation 10-15%) was isolated by ion exchange chromatography and gel filtration according to the method described earlier [7]; LPS from E. coli serotype 055: B5 (Sigma, USA) was used.
Protein samples were evaluated for the presence of LPS using the Limulus test (quantitative chromogenic Limulus amebocyte lysate (LAL); Lonza Walkersvile, USA). Incubation with polymyxin-agarose (Sigma, USA) was used to remove LPS contamination. The final concentration of LPS in the samples did not exceed 0.2 UE/ml.
Experimental model
Swimming in cold water (1-4°C) for two minutes was used to expose rats to the combined emotional and physical stress in our experimental model. Immediately before the stress exposure, 500 μl of aqueous solutions of the studied preparations were injected intraperitoneally at the following concentrations: human LF – 200 μg/kg animal weight, LPS – 500 μg/kg animal weight. In the case of a joint test, LF (in a volume of 250 μl) was administered 5 min after the LPS injection (in a volume of 250 μl).
Blood and spleens were collected for analysis 30 min and 3 h after the end of the stress exposure. All experiments were carried out at the same time interval (11:00-14:00) in order to prevent circadian fluctuations in the corticosterone levels.
Animal groups (5-7 animals in each group): (1) intact (control 1); (2) water injection, euthanasia in 30 min after stress (control 2); (3) water injection, euthanasia in 3 h after stress (control 3); (4) LPS injection, euthanasia in 30 min after stress; (5) LPS injection, euthanasia in 3 h after stress; (6) LPS injection, stress, euthanasia in 30 min after stress; (7) LPS injection, stress, euthanasia in 3 h after stress; (8) LPS&LF injection, stress, euthanasia in 30 min after stress; (9) LPS&LF injection, stress, euthanasia in 3 h after stress.
Corticosterone determination
The determination of corticosterone concentration in blood serum was performed by enzyme immunoassay using the Corticosterone EIA-4164 kit by DRG according to the manufacturer’s instructions.
Measurement of gene expression
RNA isolation from spleen cells was performed using a commercial Gene Elute Mammalian total RNA kit (Sigma-Aldrich, USA) according to the manufacturer’s protocols.
A Bio-Rad cDNA synthesis kit (iScript cDNA Synthesis Kit, USA) was used to obtain cDNA. The procedure was performed according to the manufacturer’s protocol. The obtained cDNA was stored at -20°C.
Real-time PCR was performed using the Bio-Rad CFX96 Touch™ Real-time system. Maxima SYBR Green qPCR Master mix (2X) from Thermo Scientific was used for PCR. The glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene was used as a reference gene. Primers for PCR were selected using the Primer3 software (http://bioinfo.ut.ee/primer3-0.4.0/primer3/) and literature data [8, 9]. Primer sequences (5’-3’) were as follows: IL4 sense (CGGTGAACTGAGGAAACTCTGTAGA), IL4 antisense (TCAGTGTTGTGTGAGCGTGGACTC), TLR4 sense (CCTGAAGATCTTAAGAAGCTAT), TLR4 antisense (CCTTGTCTTCAATTGTCTCAAT), GAPDH sense (CCTGCACCACCAACTGCTTAGC), GAPDH antisense (GCCAGTGAGCTTCCCGTTCAGC).
PCR protocol: 95.0°C – 10 min; 94.0°C – 15 sec; 60.0°C – 1 min; 72.0°C – 1 sec (fluorescence reading). Steps 2-4 were repeated for 40 cycles. Melting curves were acquired using 0.5°C steps from 60.0 to 94.0°C; 10.0°C to 10 min. Reaction products were evaluated and identified by means of the melting curves method. The results were processed using the CFX Manager™ software version 2.1.
RESULTS AND DISCUSSION
Without stress, gene expression of IL4 in rat splenocytes significantly increased 3 h after LPS administration compared to intact animals, while gene expression of TLR4 decreased (Fig. 1). There were no significant changes in the expression of either gene 30 min after LPS administration.

Exposure to stress together with the LPS injection led to an increase in the expression of both studied genes 3 h after the exposure (Fig. 2).
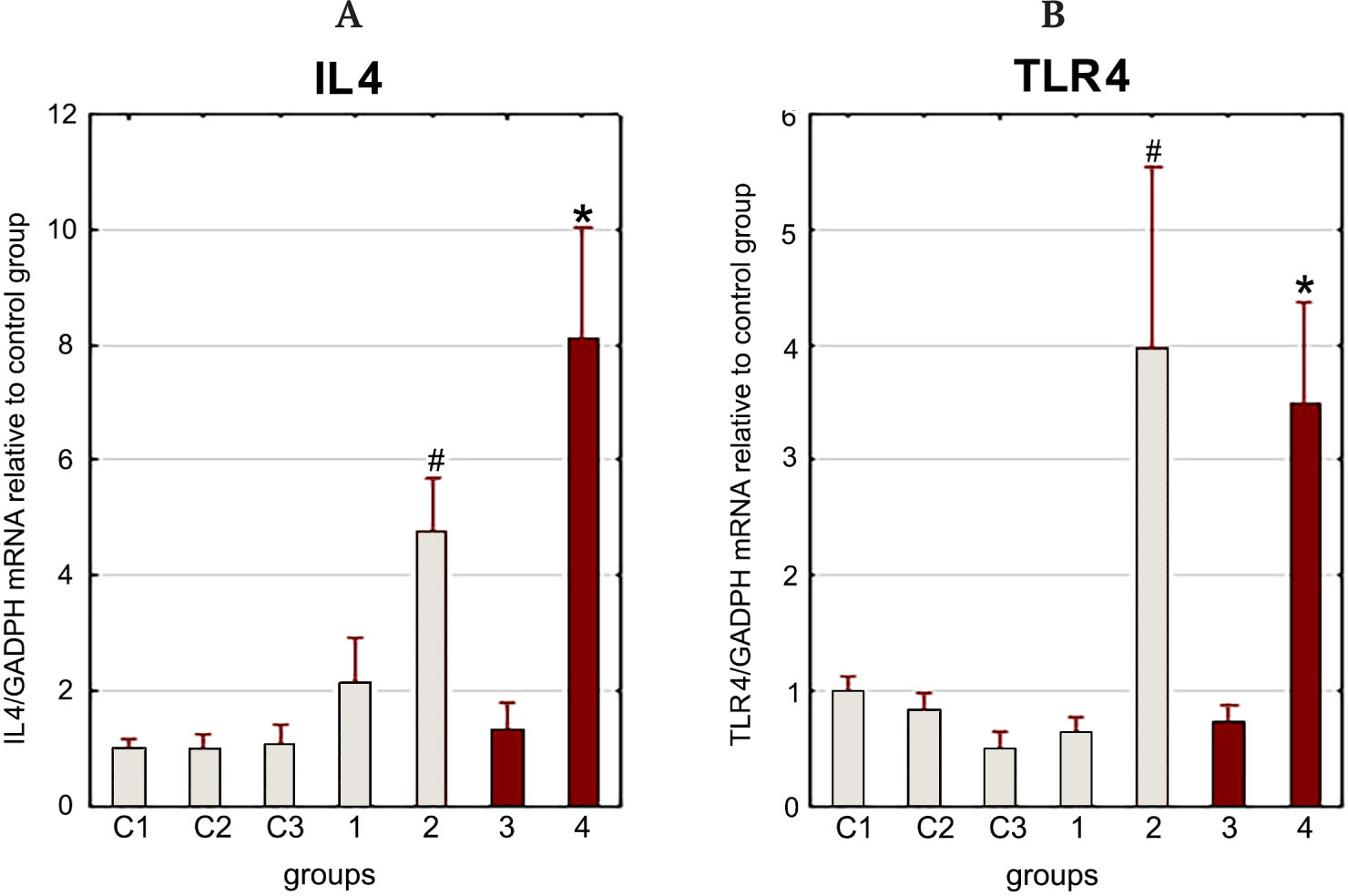
LF injection reduced gene expression of IL4 and TLR4 3 h after LPS injection and stress exposure (Fig. 3).
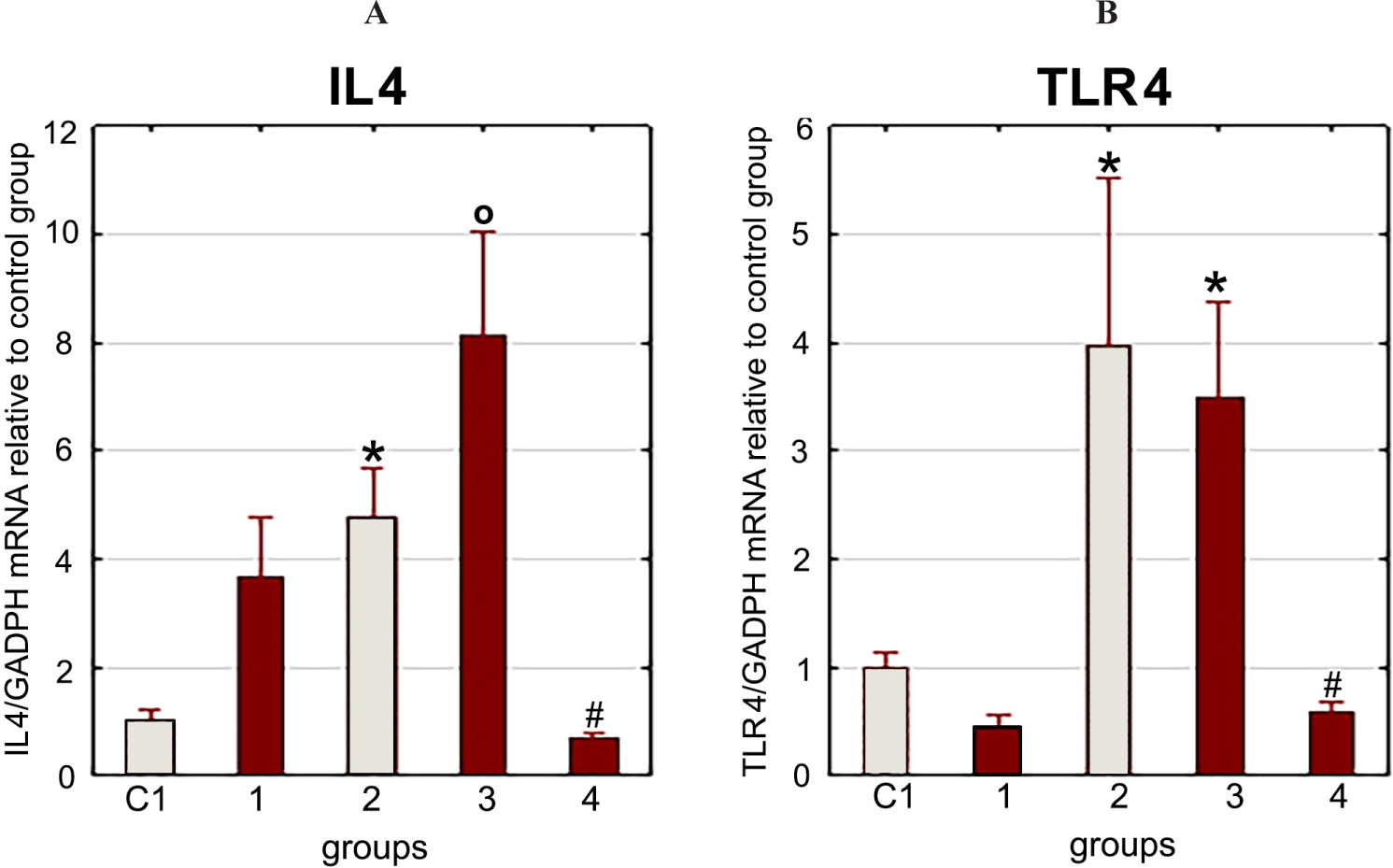
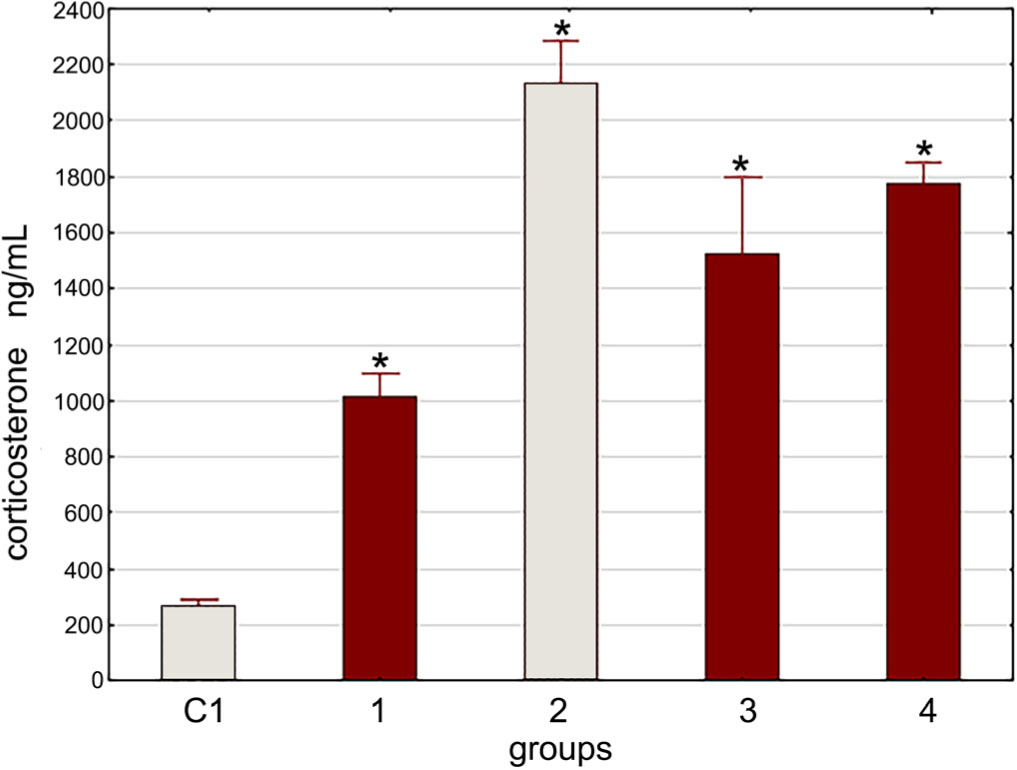
It should be noted that the administration of LF did not lead to a decrease in the elevated corticosterone blood level caused by the combined effect of stress and LPS (Fig. 4, 5).
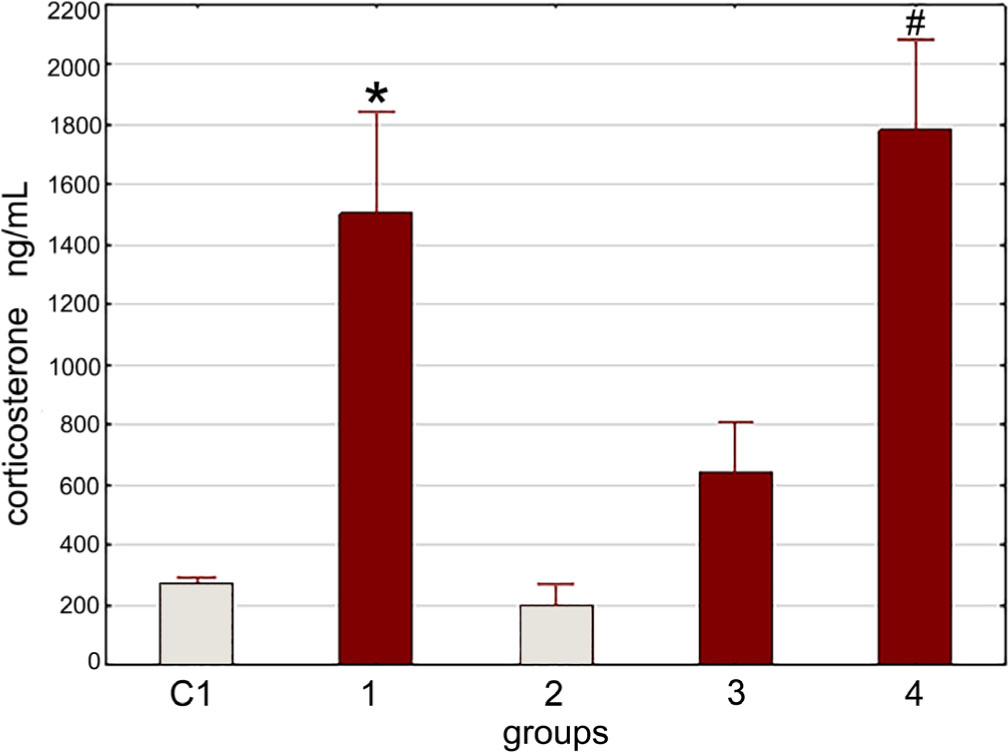
As pointed out earlier, under conditions of combined stress exposure (swimming in cold water), preventive administration of LF reduces the stress-induced increase in blood corticosterone concentration and the Tlr4 gene expression in rat splenocytes. LF administration has the same effect on the stress-induced increase in the Il4 gene expression in splenocytes (Fig. 6).
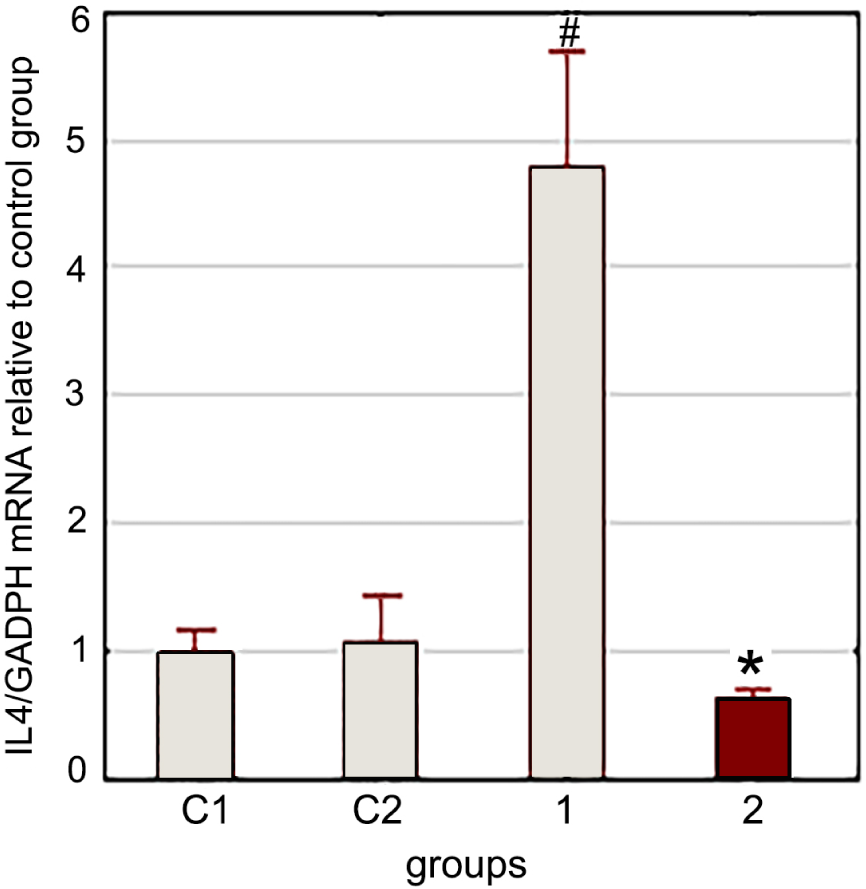
Thus, in this experiment, we have shown that LPS administration does not affect the modulating effect of LF on stress induced Il4 and Tlr4 gene expression in rat splenocytes. This modulating effect of LF on gene expression is independent of its corticostatic activity.
The mechanisms of this immunomodulatory action of LF are not yet clear. This process may involve different mechanisms. It could include central mechanisms, due to the LF ability to accumulate in brain structures like the hippocampus [10], or peripheral ones due to LF interacting with various binding sites on immune cells [11].

