INTRODUCTION
The COVID-19 pandemic, which began in Wuhan province of China in December 2019, was caused by a novel coronavirus named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. The disease caused by this coronavirus was named COVID-19. Besides triggering the respiratory symptoms, SARS-CoV-2 suppresses the patient’s immune system resulting in a wide range of secondary bacterial and fungal infections. These secondary infections are usually associated with pre-existing conditions, such as diabetes mellitus, bronchial asthma, chronic obstructive pulmonary disease (COPD), and other lung diseases, or develop as a hospital-acquired infection due to corticosteroid treatment of severe COVID-19 [2]. Secondary fungal infections are also very frequent in patients with hematological and other malignancies and in patients undergoing chemotherapy [3]. Although the number of patients with invasive secondary fungal infection is relatively low, the mortality in these patients is extremely high compared to the patients with COVID-19-associated secondary bacterial infections. The incidence of fungal infections caused by pathogens with decreased susceptibility to the currently available antifungals is increasing [4] which makes these infections a severe threat to healthcare worldwide.
We studied common invasive fungal infections, etiological agents that cause these infections, and comorbidities associated with COVID-19 at the District Medical College Hospital. The clinical manifestations, demographics, treatment, and disease outcomes in patients with COVID-19-associated fungal infections were also analyzed. The presented data on the association of invasive fungal infection with COVID-19 are limited, and further in-depth studies are needed to determine the incidence and outcomes of such infections as well as to establish patient management protocols.
MATERIALS AND METHODS
Study description
An observational cross-sectional study was performed at the Government Medical College and District Hospital in Palakkad, India. The study was executed following the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. The duration of the study was six months from March to August 2021. All documentation has been obtained with permission from Government Medical College and District Hospital in Palakkad, India.
Patients
A total of 50 patients (40 to 74 years old; median 56 years old) were included in the study. Case histories and laboratory records of COVID-19 patients and post-COVID-19 patients with symptoms including facial swelling, headache, and signs of sinusitis or upper air obstruction were analyzed. Data including age, gender, clinical and laboratory data, comorbidities, and duration of hospital stay were registered. A continuous sampling method was used. All measures were taken to keep the confidentiality related to patients’ identity while compiling the data.
Sample analysis
Samples received in the microbiology laboratory were divided into three parts. The first part was subjected to a KOH (potassium hydroxide) mount examination under 10x and 40x magnification, while the remaining two parts were cultured on Sabouraud’s dextrose agar and kept at 25°C and 37°C, respectively. Colonies on the culture media were identified by a tease mount test using lactophenol cotton blue (LPCB). The slide culture technique was used to study the undisturbed morphology of the fungus. The final identification of the fungus species was based on the morphological characteristics as described in the Larone’s Medically Important Fungi: A Guide to Identification [5].
Biopsy samples received for histopathology examination (HPE) were fixed in 10% formalin and processed in tissue processor Histokinette (Leica TP 1020), stained with haematoxylin and eosin stain (H&E), and observed using Labomed Lx 500 microscope at 40x magnification (Labomed, USA).
For statistical analysis, the data were described as either continuous or categorical variables. The categorical variables were presented as frequency and percentages.
RESULTS
We received a total of 50 samples from COVID-19-positive patients with suspected fungal infections of the respiratory tract. Among these samples, 14 (28%) were positive for fungal pathogens (KOH mount, HPE, or culture). From these 14 samples, seven were positive for Mucorales species and another seven − for Aspergillus species.
Patients diagnosed with mucormycosis were 54.7±9.0 years old on average. The male/female ratio was 3:4. None of the patients was vaccinated with a vaccine against COVID-19. The average interval between COVID-19-positive diagnosis and the onset of mucormycosis symptoms reached 10.1 days (from 4 to 20 days). Out of the seven patients with mucormycosis, six (85.7%) had diabetes (Table 1). One of them had diabetic ketoacidosis, another one was on ayurvedic treatment, and the rest had uncontrolled diabetes. One patient with no prior history of diabetes was detected to have abnormal blood glucose levels during the hospital stay, most probably due to extensive corticosteroid therapy. Finally, three out of seven (42.9%) patients were treated with corticosteroids. Only one patient had received remdesivir. None of the patients had a history of admission to the Intensive Care Unit (ICU) for COVID-19 treatment. Four patients had rhino-orbital mucormycosis, three had sinonasal mucormycosis, and all of them had ptosis.
| Patient No | Age/Sex | Facial pain | Facial numbness | Facial abscess | Headache | Tears from eyes | Ptosis | Decreased vision | Restricted ocular movements | Epistaxis |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | 40 / M | Yes | Yes | Yes | Yes | Yes | Yes | |||
| 2. | 45 / F | Yes | Yes | Yes | Yes | |||||
| 3. | 55 / M | Yes | Yes | |||||||
| 4. | 65 / M | Yes | ||||||||
| 5. | 58 / F | Yes | ||||||||
| 6. | 60 / F | Yes | Yes | Yes | Yes | |||||
| 7. | 60 / F | Yes | Yes | Yes | Yes | Yes |
The etiological agent – Rhizopus oryzae (Rhizopus spp.) – was identified in five out of seven (71.4%) samples. One sample demonstrated sporangium along with aseptate hyphae.
Samples obtained from endoscopic sinus surgery were subjected to a KOH mount test and culture analysis. HPE was done in four of these samples. The KOH mount test and HPE of the samples showed aseptate fungal hyphae branching at right angles. In one sample, we noted sporangium along with aseptate hyphae (Fig. 1).
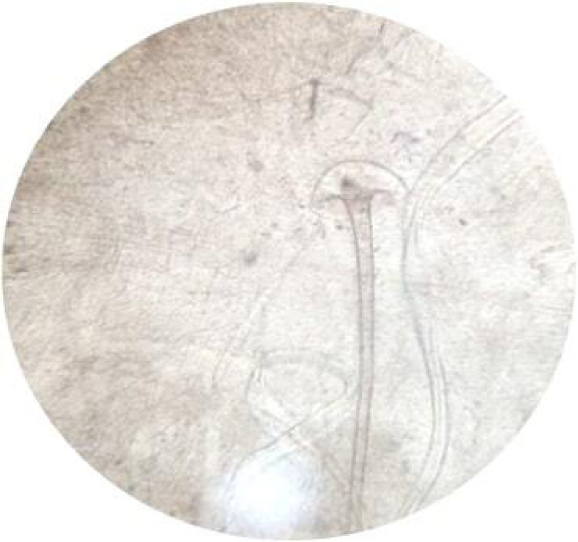
While studying samples obtained from seven patients with mucormycosis, Rhizopus spp. was isolated from five samples, and no fungal pathogens were detected in the other two samples. All these patients were treated with Liposomal Amphotericin B (Bharat Serums, India). Six patients died, and one 55-year-old male patient with sinonasal mucormycosis recovered. The mortality rate was 85.7%. The clinical data of these patients are summarized in Table 2.
| Patient No | Age/Sex | Comorbidities | Corticosteroid therapy | KOH mount | Culture | HPE | Treatment | Clinical form of mucormycosis | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 40/M | Diabetic on OHA | Dexona | Aseptate hyphae | Rhizopus spp. | Broad aseptate hyphae branching at right angles | Liposomal amphotericin B | Rhino orbital mucormycosis | Deceased |
| 2 | 45/F | Diabetic, CKD | NA | Aseptate hyphae | Culture-negative | Not performed | Liposomal amphotericin B | Rhino orbital mucormycosis | Deceased |
| 3 | 55/M | Diabetic on Ayurveda treatment | NA | Aseptate hyphae | Rhizopus spp. | Broad aseptate hyphae branching at right angles | Liposomal amphotericin B | Sinonasal mucormycosis | Recovered |
| 4 | 65/M | Diabetic, DKA | Dexona | Aseptate hyphae | Rhizopus spp. | Broad aseptate hyphae branching at right angles | Liposomal amphotericin B | Sinonasal mucormycosis | Deceased |
| 5 | 58/F | CAD | Methylprednisolone | Aseptate hyphae | Rhizopus spp. | Broad aseptate hyphae branching at right angles | Liposomal amphotericin B | Sinonasal mucormycosis | Deceased |
| 6 | 60/F | Diabetic, left MCA stroke | No | Aseptate hyphae | Rhizopus spp. | Not performed | Liposomal amphotericin B | Rhino orbital mucormycosis | Deceased |
| 7 | 60/F | Uncontrolled diabetes, hypertension | NA | Aseptate hyphae | Culture-negative | Not performed | Liposomal amphotericin B | Rhino orbital mucormycosis | Deceased |
OHA – oral hypoglycemic agents, CKD – chronic kidney disease, MCA – middle cerebral artery, CAD – coronary artery disease, DKA – diabetic ketoacidosis, NA – not applicable, F– female, M– male.
Among patients diagnosed with aspergillosis, none was vaccinated against COVID-19. The mean age of patients was 58.0±7.2 years. The male/female ratio was 4:3. The average interval between COVID-19-positive diagnosis and the onset of symptoms was four days (from 1 to 8 days). All seven patients had diabetes mellitus, five of them had hypertension, and one patient had pulmonary aspergillosis and a history of pulmonary tuberculosis. Five patients had received corticosteroid therapy for COVID-19 treatment: four patients were given remdesivir and one was treated with ivermectin. Cough, breathlessness, sinusitis-like headache, facial pain, and nasal congestion were observed as the common symptoms (Table 3). Some of these patients had other symptoms including chest pain and hemoptysis. None of the patients had a history of prior admission to ICU for COVID-19 treatment. Samples obtained from endoscopic sinus surgery were subjected to the KOH mount test and culture analysis. The KOH mount test and HPE of six samples showed septate fungal hyphae branching at acute angles. In one sample, both the KOH mount test and H&E staining of the biopsy specimen identified conidia along with septate hyphae (Fig. 2).
| Patient No | Age/Sex | Comorbidities | Corticosteroid therapy | KOH mount | Culture | HPE | Treatment | Clinical form of aspergillosis | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 55/M | Diabetic, CAD, hypertensive, old pulmonary tuberculosis | Dexona | Septate hyphae and conidia of Aspergillus | Aspergillus flavus | Thin septate hyphae branching at acute angles | Liposomal amphotericin B | Pulmonary aspergillosis and sphenoid sinusitis | Deceased |
| 2 | 56/M | Diabetic | No | Septate hyphae | Aspergillus flavus | Thin septate hyphae branching at acute angles | Liposomal amphotericin B | Fungal maxillary sinusitis | Recovered |
| 3 | 57/M | Diabetic | Dexona, Methylprednisolone | Septate hyphae | Aspergillus niger | Thin septate hyphae branching at acute angles | Liposomal amphotericin B | Fungal maxillary sinusitis | Deceased |
| 4 | 56/F | Diabetic, hypertensive | Methylprednisolone | Septate hyphae | Aspergillus fumigatus | Not performed | Voriconazole | Fungal maxillary sinusitis | Deceased |
| 5 | 55/F | Diabetic, hypertensive | Dexona | Septate hyphae | Aspergillus niger | Thin septate hyphae branching at acute angles | Liposomal amphotericin B | Fungal maxillary sinusitis | Deceased |
| 6 | 53/F | Diabetic, hypertensive | Dexona | Septate hyphae | Aspergillus niger | Thin septate hyphae branching at acute angles | Voriconazole | Fungal maxillary sinusitis | Recovered |
| 7 | 74/M | Diabetic, CAD hypertensive | No | Septate hyphae | Culturenegative | Thin septate hyphae branching at acute angles | Voriconazole | Fungal maxillary sinusitis | Recovered |
F – female, M – male.
Six out of the seven samples were fungus-positive according to the culture analysis, of which three were found to have Aspergillus niger, two – Aspergillus flavus, and one – Aspergillus fumigatus. Three of the seven patients were treated with amphotericin B and the rest were given voriconazole. Of these seven patients, three recovered. The final mortality rate was 57.1%.
DISCUSSION
We identified two types of deep fungal infections – mucormycosis and aspergillosis – in patients diagnosed with COVID-19. Mucormycosis is caused by the fungi that belong to the order Mucorales, which includes Rhizopus, Mucor, Rhizomucor, Cunninghamella, Lichtheimia, and Apophysomyces spp. [6]. The prevalence of mucormycosis in humans ranges from 0.005 to 1.7 per million worldwide, while in India it ranges from 0.14 to 1000 [7]. In addition, India ranks second in the world in terms of the total number of patients with diabetes mellitus, which is considered a risk factor for mucormycosis [8]. Lionakis et al. [9] showed that a cumulative dose of prednisolone above 600 mg per day or a total dose of 2-7 g of methylprednisolone taken over one month predisposes immunocompromised individuals to mucormycosis. The unregulated use of corticosteroids for the treatment of COVID-19 contributed to an increased number of cases of mucormycosis during the second COVID-19 wave in India [10]. The probability of mucormycosis in individuals with diabetes treated with corticosteroids is even higher. Over the period from May 5 to July 12, 2021, 41,512 cases of mucormycosis with 3,554 lethal outcomes were reported in India [11]. Most of those cases occurred during SARS-CoV-2 outbreaks in India.
Three theories have been suggested to explain COVID-19-associated mucormycosis. The first one suggests that lymphopenia associated with COVID-19 enables opportunistic fungal pathogens to gain entry [11, 12]. Another observation is that increased levels of proinflammatory markers in patients with severe disease help fungal pathogens to proliferate in the human body. The third theory suggests that pronounced damage of pulmonary tissues by COVID-19 makes them conducive to invasive fungal pathogens, especially airborne ones [12].
Six clinical forms of mucormycosis defined by the location in the body are described in the literature including rhino-cerebral, pulmonary, cutaneous, gastrointestinal, disseminated, and uncommon presentations [13]. The most common site of invasive mucormycosis is the sinuses (39%), followed by the lungs (24%) and skin (19%). In a study from a tertiary care center in South India, the rhino-orbito-cerebral mucormycosis was described as the most common type of this disease [14].
Direct microscopy and KOH mount microscopy are invaluable tools for the rapid diagnosis of mucormycosis [15]. Histopathology is important for determining angioinvasion and distinguishing the presence of fungi from culture contamination [16]. In our study of seven patients diagnosed with mucormycosis, 57% had rhinoorbital mucormycosis and 43% had sinonasal mucormycosis. Rhizopus oryzae (Rhizopus spp.) was identified as the etiological agent. This is the most common fungus isolated from clinical specimens of patients with mucormycosis, followed by Lichtheimia corymbifera and Mucor racemosus [17]. The cases where sporangium along with aseptate hyphae were found in the same sample using the KOH mount test are described in the literature [18].
All patients with mucormycosis that were included in our study were treated according to the Recommendations from the European Conference on Infections in Leukemia (ECIL-6) (2016) and ESCMID/ECMM guidelines, which recommend a lipid formulation of amphotericin B (5 mg/kg/day, 10 mg/kg/day for infection of the central nervous system) as the first-line therapy for mucormycosis [15]. A recently developed medication isavuconazole also shows good activity against Mucorales [19]. The successful treatment of mucormycosis depends mainly on surgical debridement of the infected tissues. The mortality rate shown for patients with mucormycosis in our study (85.7%) corresponds to the literature data (46-96%). The mortality rate depends upon the patient’s comorbidities and can reach 90% in case of intracranial involvement [20].
The second infection – aspergillosis – was diagnosed in seven out of 50 patients tested in our study and was caused by Aspergillus niger, Aspergillus flavus, or Aspergillus fumigatus. Aspergillus spp. is one of the most common etiological agents associated with fungal rhinosinusitis; this disease is rare, although it is associated with high mortality [21]. Aspergillus spp. are ubiquitous, and their spores can easily enter the body with airborne droplets. The maxillary sinus is commonly affected by invasive fungal sinusitis. We also noted the involvement of the maxillary sinuses in six of our patients; one patient had aspergillosis of the lungs and sphenoid sinus. The most common Aspergillus species causing invasive infection is A. fumigatus [21]. The main predisposing factors for aspergillosis include uncontrolled diabetes mellitus and human immunodeficiency virus infection, as well as the misuse of antibiotics, immunosuppressants, and corticosteroids [22]. Immune dysregulation associated with acute respiratory distress syndrome (ARDS) and its treatment with tocilizumab or corticosteroids are also factors contributing to infection. Pulmonary aspergillosis is common in patients with a history of tuberculosis due to extensive structural lung changes [23]. All the patients (7/7) diagnosed with invasive aspergillosis in our study had diabetes. Five of them had received corticosteroids for COVID-19 treatment. One patient had pulmonary aspergillosis along with sphenoid sinus involvement.
Microscopy is the easiest way to diagnose aspergillosis, although its specificity is low. Aspergillus species rarely spore in vivo, so the hyphae may represent any of the filamentous fungi. Therefore, the sensitivity of this method varies from 0% to 90% [24]. PCR is the most sensitive method in detecting fungal rhinosinusitis, followed by culture analysis, potassium hydroxide microscopy, and histopathology [25]. Fruiting bodies/conidia are rare in tissue samples, but there are reports of such unusual findings in aspergillosis [26]. In our study, one patient with pulmonary aspergillosis had conidia along with septate hyphae in a direct nasal endoscopy (DNE) specimen. HPE also showed a similar result for this patient. However, diagnosis should not be based on results of a single method. A biopsy can confirm tissue invasion by the fungus [25]. In our study, HPE biopsy analysis and culture analysis confirmed aspergillosis in six out of seven patients, although the latter method is less sensitive for diagnosing aspergillosis [27]. The mortality rate also varies depending on the co-morbidities of the patient. Lin et al. [28] showed that disseminated infection by Aspergillus resulted in 88% mortality rate. The mortality rate in our study was 57.1%. Surgery is an important element of treatment since it helps to obtain tissue samples for diagnosis and to sanitize necrotic tissues [29]. Isavuconazole and voriconazole are considered as the first-line treatment medications, whereas there is a moderate support for the use of liposomal amphotericin B [30]. Posaconazole is another drug that has been used in the treatment of invasive aspergillosis [30]. Only three patients in our study were treated with voriconazole and four received liposomal amphotericin B.
The small sample size is the main limitation of our study, which makes it difficult to generalize the conclusions.
CONCLUSION
Invasive fungal infections are fatal, especially when seen in patients suffering from severe COVID-19 infections. An early diagnosis and recognition of such infections as well as their prompt treatment are required to reduce the mortality and morbidity in such patients. To achieve a prompt diagnosis of invasive fungal infections, we recommend using the KOH mount examination since both fungal culture analysis and histopathological examination of the endoscopic biopsy samples are more time-consuming, which leads to increased patients’ mortality. In conclusion, our data suggest that the risk of invasive fungal infections is increased in COVID-19 patients and their recovery rates are low. Therefore, further research on the association between invasive fungal infections and COVID-19 is needed, especially during the pandemic.


