Introduction
Bone fractures are the most common human trauma. Fracture repair is a regeneration process, which summarizes many events of skeletal development [1]. Under healthy conditions, bones have a unique ability to recover that does not lead to the formation of scarring. But, a complex or severely damaged fracture (i.e., cracks above critical size) may not heal, resulting in a nonunion fracture [2]. Comorbidities such as diabetes [3], hereditary factors [4] and an unhealthy lifestyle [5] (e.g., alcohol abuse or smoking) are associated with a heightened risk of delayed healing and nonunion of fractures. Besides, improper treatment may lead to nonunion of fractures.
The treatment strategy of bone defects is mainly directed toward autogenous bone grafts, which constitute the clinical “gold standard” for the treatment of bone defects due to their osteoconductivity and osteoinduction [6]. Although autotransplantation is restricted by the volume of bone which is obtained from the iliac crest and afterwards transplanted to the defect, it does not cause an immune response [7]. Moreover, complications also exist, such as local hematoma and bone graft reconstruction. Using the same or an xenogeneic or an allogeneic bone source is a way of overcoming the limitation of the donor site and autograft supply. Xenografts, mainly from bovine origins, have conductive/osteoconductive ability [8]. Alternatively, allogeneic bone obtained from cadaveric sources has the advantages of being osteoinductive and osteoconductive, and can be obtained in various shapes and sizes through a regional tissue bank [9]. But studies stemming from clinical trials have identified that allograft treatments usually produce an increased risk of transmission of infectious disease, immunogenicity, and host rejection [10, 11].
In recent years, there have been increasing applications of growth factors (GFs), including bone repair. GFs have a great advantage in bone repair, as they do not cause many side effects compared with bone grafts. The pathology of bone healing is complex, involving several vital GFs [12]. In this section, we review a strategy for promoting bone healing with biomolecules based on the recent research publications databases. The BFs-based strategies can be divided into four subcategories: (i) use of inflammatory factors, (ii) use of angiogenic factors; (iii) use of osteogenic factors, and (iv) mixtures of these GFs. Meanwhile, the clinical challenges for these BFs-based therapies are carefully explored. Finally, the current proposed strategies for delivery of BFs are reviewed from different delivery vehicles, such as rigid bone grafts, microspheres, nanoparticles and gels.
The self-healing process after bone fracture
The sequence of hematoma formation, inflammation, callus formation, and bone remodeling are the processes a fractured bone undergoes as it regenerates [13]. These processes partially overlap in time, with revascularization of the injured site and activation of local and systemic defenses (Figure 1). The first period of secondary fracture repair is hematoma formation, followed by acute inflammation. After a bone fracture, the plasma coagulation cascade is activated first, and then the peripheral blood vessels and bone marrow rupture to form a hematoma. Platelets are dispersed to the extravascular site, forming a fibrin network in the extravascular environment as the first temporary matrix for the inflammatory cells to flow into. Massive immunocytes including neutrophils, lymphocytes, and macrophages are attached to the fibrin network at the fracture site by inflammatory molecules derived from the damaged tissues to elicit acute inflammation. Neutrophils, the first inflammatory cells, reach to the fracture area within the first 24 h. For example, neutrophils secrete chemotactic mediators, such as interleukin-6 (IL-6) or C-C motif chemokine ligand 2 (CCL2), to recruit the next wave of inflammatory cell infiltration to the fracture site, named monocyte/macrophage [14]. Subsequently, the mononuclear cells in osteoclasts absorb the necrotic bone fragments and the necrotic end of the fracture, and macrophages remove the temporary fibrin matrix and necrotic cells through phagocytosis. Classically-activated M1 macrophages and alternatively activated M2 macrophages are two major phenotypes of macrophages. They are important immune cells and play an important role in the activation and decomposition of inflammation. M1 macrophages secrete tumor necrosis factor-α (TNF-α), IL-1β, IL-6, CCL2 and other inflammatory and chemotactic mediators. These mediators can activate the recruitment of fibroblasts, mesenchymal stem cells (MSCs) and bone progenitor cells from their local niches [15]. While M2-typed macrophages produce transforming growth factor-β (TGF-β) family members, especially TGF-β1, -β2, -β3, and vascular endothelial growth factor (VEGF), bone morphogenetic proteins, fibroblast growth factor-2 (FGF-2), and bone morphogenetic protein-platelet-derived growth factor (BMP-PDGF), which are the key mediators of fracture healing [16, 17].
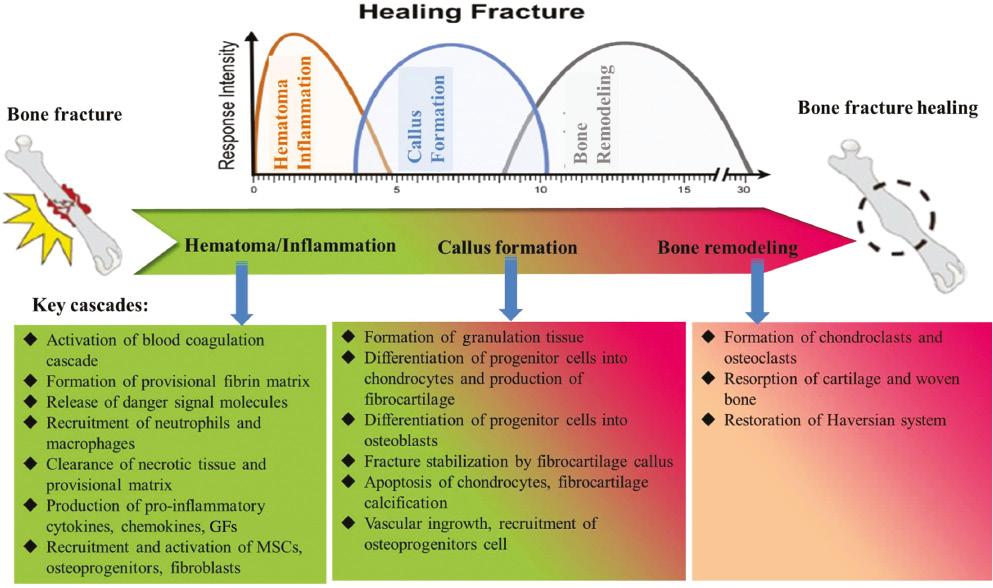
The process of fracture healing and the main cascades during each stage: the process of bone defects self-repairing includes hematoma formation, inflammation, callus formation, and bone reconstruction. These processes partially overlap in time.
After the inflammation has subsided, the bone marrow mesenchymal progenitor cells gather around the bone defect and form granulation tissue, which is mainly differentiated into chondrocytes [13]. Chondrocytes produce a cartilage matrix, form a soft callus, and finally extend into the fracture gap to connect the end of fractured bone for several weeks. Cartilaginous tissue in the callus provides the mechanical stability of the bone fracture, and is then gradually replaced by immature bone tissue (this process is commonly known as endochondral bone formation) [18]. If the fracture is small, the fragments are stable, then the formation of endochondral bone is minimal, and the formation of intramembrane bone occurs [14].
With the formation of a soft callus, new bone formation takes place in an area with a good local blood supply and mechanical stability through the intramembrane pathway. Direct differentiation of bone marrow MSCs and periosteal osteoblasts into osteoblasts occurs. Finally, the external surface of the fibrocartilaginous callus is covered with woven bone. At the same time, chondrocytes undergo hypertrophy and apoptosis in the soft callus, and secrete a calcium medium that stimulates vascular ingrowth, and finally leaves the calcified cartilage extracellular matrix (ECM). This stage of fracture repair is often called hard callus formation. In general, the transformation from soft tissue to hard tissue occurs over several time periods after the primary injury. Finally, osteoclasts remove the woven bone and underlying cartilage matrix, initiating the remodeling process, and finally reconstruct the typical bone tissue structure and Haversian system. It may take several months to complete the remodeling process, but eventually it restores the shape and function of the bone without any scarring.
Classification of the exogenous GFs enhancing bone healing
Fracture repair is a complex and multi-step process, which is composed of different spatiotemporal cytokines. It requires the hybrid of many cell types, including osteocytes, inflammatory cells, mesenchymal progenitor cells, vascular cells, and inflammatory cells. The function of bioactive factors (BFs) is beneficial to the repair of bone tissue. BFs provide signals at the injured site, which allows inflammatory cells, angiogenesis, and progenitor cells to migrate and start the repair process. The BF family involved in the regulation of bone regeneration mainly includes fibroblast growth factors (FGFs), bone morphogenetic protein (BMPs), VEGF, insulin-like growth factor (IGF), and TGF-β. Special consideration has been paid to the use of BMP-7 or BMP-2, which have been included in Food and Drug Administration (FDA)-approved for bone regeneration devices. These BFs were pharmacologically classified into inflammatory factors, angiogenic factors, and osteogenic factors in Figure 2 and their specific roles in repair are overviewed in the following sections. Meanwhile, the combinational therapy of multiple BFs is also outlined in this section.
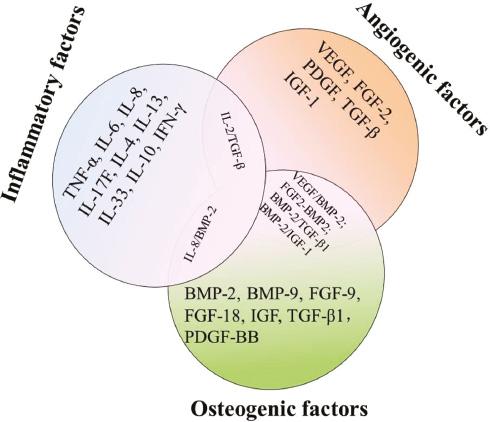
The classification of BFs involved in bone repair and the common combination of multiple BFs. BFs, bioactive factors.
Inflammatory factors
In the initial period of repair, pro-inflammatory cytokines, including TNF-α, IL-6, IL-17F, and IL-8, are released from the injured site, and then rapidly shift to the anti-inflammatory cytokines. These proinflammatory cytokines play various roles in attracting inflammatory cells or bone marrow stromal cells (BMSCs) into the site of the damage. Moreover, these proinflammatory cytokines can regulate the osteogenic differentiation and proliferation of hASC (human adipose stem cells) in vitro cell experiments. For example, TNF-α and IL-6 increase hASC proliferation and alkaline phosphatase (ALP) activity while IL-17F decreases the proliferation of hASC without increasing ALP activity [19]. It was also considered that these proinflammatory and anti-inflammatory cytokines are critical for fracture healing during bone self-healing [14]. However, in the case of diabetes or a nonunion facture, uncontrolled proinflammatory factors such as TNF-α can promote serious bone desorption.
On the one hand, TNF-α can stimulate BMSCs to express a sort of inflammatory-related protein, thus increasing the extreme inflammatory response after a fracture [20]. On the other hand, high TNF-α can inhibit the differentiation of BMSC into osteoblasts because it expresses less osteogenic-related proteins, thus delaying bone formation [21]. The results showed that the local sustained delivery of TNF-α antibody by a glucose-sensitive scaffold could effectively decrease the inflammatory response of diabetic patients and promote the healing of alveolar bones [22].
Bone tissue healing relies on the switching from an initial inflammatory response to a pro-reparative status. Macrophages play a vital role in the activation and resolution of inflammation, which are important as immune cells [23]. Typically-activated M1 macrophages and alternatively-activated M2 macrophages are the two main phenotypes of macrophages. M1 macrophages are activated by interferon-gamma (IFN-γ) and/or lipopolysaccharide (LPS), while M2 are activated by IL-4, IL-10, or IL-13. Macrophages can switch from one phenotype to another according to the surrounding microenvironment, which has strong plasticity. The dynamic change of macrophages is of great significance for tissue repair. IL-4 is a major cytokine responsible for polarizing M1 to M2 phenotype. It is reported that in the subcutaneous model of mice, the anti-inflammatory cytokine IL-4 transformed the macrophage of proinflammatory M1 into the phenotype of pro-healing M2, restored the M2/M1 ratio to the normal level, effectively solved the inflammation, and finally promoted the bone regeneration and differentiation of osteoblasts [24].
Angiogenic factors
Angiogenesis provides the necessary nutritional support for newly formed tissue and a cell source for more tissue repair. Thus, angiogenesis is regarded as a necessary process for bone regeneration, and it is defined as the formation of new blood vessels from pre-existing vascular networks. The main angiogenic factors include VEGF, FGFs, PDGF and TGF β.
VEGF is a vital regulator of angiogenesis in the process of bone formation. It is produced by endothelial cells, osteoblasts, and other cells at the fracture site, and the expression of VEGF reaches a peak at the early stage of a fracture. VEGF is one of the two important factors in promoting the formation of new bone in the process of endochondral and intramembrane osteogenesis.
The FBF family consists of 22 members and has many biological functions, such as bone formation. It can promote the proliferation of endothelial cells and osteoblasts and induces angiogenesis by participating in the FGF pathway. FGF2, FGF18, or FGF9 are considered to be candidates beneficial for bone healing, based on the phenotype of transgenic mice and the level of spatiotemporal expression during fracture healing [25]. FGF2 is an effective angiogenesis-inducing factor, which regulates cell function through the RAS/MAP kinase pathway. In the mouse model, the destruction of the FGF2 gene leads to the decrease of bone formation. This suggests that FGF-2 (or bFGF) plays an important role in wound healing and tissue repair.
PDGF a kind of protein released by platelets and macrophages during tissue damage and fractures. PDGF-BB dimer is the most effective GF with the ability of chemotaxis and mitosis of osteoblasts (the key osteoblasts) [26]. Additionally, PDGF has a synergistic effect with endogenous VEGF, which can stimulate the neovascularization of the defect site, promote the repair of hard and cartilage tissue, and is a kind of angiogenic factor. Recombinant human platelet-derived growth factor (rhPDGF-BB) was approved by the FDA in 1997 as Regranex® for the repairing of diabetic dermal ulceration [27]. Subsequently, in 2015, the FDA approved the use of PDGF-BB/β-tricalcium phosphate (TCP) as a substitute for autograft in ankle and hindfoot fusion surgery.
Osteogenic factors
The progenitor cells can be differentiated into osteoblasts, which can form bone through endochondral and intramembrane ossification, and are then reshaped by osteoclasts to form new intramembrane bone. Therefore, bone repair depends on the recruitment of progenitor cells. Multiple pro-osteogenic BFs include BMPs, TGF-β1, FGF-9, FGF-18, and IGF. Therefore, the use of BFs is a promising method to improve the methods of bone tissue engineering.
In fact, BMP-2 and BMP-7 have been used to treat open tibial fractures, nonunion injuries and spinal fusion, and have been included in the FDA-approved bone regeneration system. BMP-2 is a member of TGF-β superfamily. It has a high osteogenic induction effect on progenitor cells. It has been proven that BMP-2 can induce osteogenesis in both ectopic and in situ in animal models and clinical trials.
Among them, the most well-known bone regeneration pathway is BMP signaling. BMP-2 and BMP-7 are members of BMP family and have good characteristics. In fact, BMP-2 and BMP-7 have been used to treat open tibial fractures, nonunion injuries and have been included in the FDA-approved bone regeneration system. BMP-2, a member of the TGF-β superfamily of proteins, is highly osteoinductive through its effect on progenitor cells, and it has been verified in clinical trials and animal models that BMP-2 can induce bone formation in both ectopic and orthotopic sites.
With the decrease of neovascularization and the recruitment of osteoclasts, the repair of a 1-mm unicortical bone fracture was destroyed. This can be avoided by using the exogenous addition of FGF9 (2 μg) with a collagen sponge instead of FGF2. FGF9 promotes chondrocyte hypertrophy and cartilage angiogenesis [28]. In diabetic mice (dB/DB), 1-mm unicortical fracture healing was significantly delayed, with decreased levels of osteogenic marker expression [29]. The better bone healing in these model mice was achieved by treatment of FGF9 with a collagen sponge. The collagen sponge soaked in FGF9 also had a sufficient bone regeneration effect on the mouse circular calvarial bone defects with a diameter of 2 mm [30]. FGF18 has a positive effect on osteoblasts by promoting the proliferation and differentiation of osteoblasts, but has a negative effect on the formation of cartilage [25, 31]. The experiment also showed that heparin coated acrylic microspheres soaked in FGF18 promoted the differentiation of mouse embryonic osteoblasts by up-regulating the expression of BMP2 in osteoblasts cell lineage cells [32].
It is well known that insulin-like GF-1 stimulates cell growth and differentiation in various tissues, especially through endocrine/paracrine and autocrine mechanisms to regulate the growing skeleton [33]. It is reported that recombinant human IGF-1 produced by a suspension culture of transgenic plant cells can promote the formation of new bone in skull defects [34]. Furthermore, IGF-1 plays an important role in maintaining the proliferation of chondrocytes and osteoblasts and inducing full osteogenesis of cartilage. IGF-1 is also necessary for the action of the parathyroid hormone (PTH) on bone; in the absence of IGF-1 in osteoblasts, PTH has no positive effect on bone formation.
Combinational therapy of multiple GFs
Because there are many signal pathways involved in bone healing, it is a very interesting field to try and improve the therapeutic effect through the transmission of various GFs. The combined application of dual BFs shows their effectiveness in bone repair. Because of the strong osteogenic effect of BMPs on bone regeneration, current multiple BFs therapy are mainly based on a combination of BMPs with angiogenic factors or inflammatory factors. In particular, simultaneous interpreting of ImageJ plural time and osteoinductive effects is considered a promising strategy for enhancing bone formation. BMP-2 is commercially approved on bone injury sites for bone tissue regeneration by the US FDA. Compared with BMP-2, the current research shows that the combination of BFs with different biological activities can promote bone regeneration. For example, many studies have shown the role of GFs such as VEGF-BMP-2 or FGF-2-BMP-2 in promoting bone formation in severe defects. In recent years, the role of the VEGF-BMP-2 scaffold and the FGF-2-BMP-2 scaffold in promoting vascularization and new bone formation has been directly compared in the literature [35]. The results show that there is no significant difference between FGF-2 and VEGF-loaded scaffolds in promoting angiogenesis and new bone formation. However, FGF2 mainly promotes stem cell migration, while VEGF promotes neovascularization in the defect site. Dual delivery of BMP-2 and IGF-1 can also promote the differentiation of osteoblasts in vitro [36].
Additionally, the combination of BMPs with inflammatory factors was also demonstrated to be synergistically active in promoting bone fracture healing. For example, the exogenous chemokines IL-8 and BMP-2 are coordinated in a spatiotemporal transport system based on mesoporous bioactive glass (MBG) to promote the rapid initiation of endogenous repair and subsequent osteogenesis stimulation [37]. The synergistic signal stimulation of BMP-2 and IL-8 induces rapid initiation in the first period of endochondral ossification, which includes effective stem cell recruitment and cartilage/osteoblast balance, which together lead to extensive bone mineralization and regeneration in the process of large-scale defect repair.
Sometimes, it has been very helpful for bone regeneration by combining several inflammatory factors to inhibit immune reaction. Regulatory T cells (Tregs) are a small subset of T cells and an important regulator of the immune system. By inhibiting the activity of other immune cells, they can maintain their autotolerance to self-antigens and reduce the indicants of autoimmune diseases [38]. Therefore, expansion, perfusion, and regulation of Tregs are considered as a treatment for destructive inflammation and autoimmune diseases. TGF-β and IL-2 are known to promote Treg recruitment, differentiation, and proliferation. Rapamycin combined with TGF-β and IL-2 can increase the Treg number in vitro. By using nanofiber sponge microspheres, TGF-β/IL-2, and miR-10a are released, T cells are collected locally and stimulated to differentiate into Tregs, which effectively saves periodontal bone defects [39].
Except for the combination of multiple BFs, therapeutic chemical factors such as vitamin D, NEL-like protein-1 and dexamethasone (DEX), which can promote cell differentiation angiogenesis and bone regeneration, was also combined with BFs to promote bone healing by delivery platforms. These biochemical signals combined BMP-2 to synergistically accelerate bone repair and remodeling, which was carefully reviewed in literature [40].
The confronted challenges to deliver BFs
Compared with bioactive small molecules, BFs have inherent adverse characteristics, and their delivery needs to be controlled very carefully. BFs usually exists in the form of a recombinant protein, which has non-specific biological activity and obvious adverse reactions with poor stability in the physiological environment [41]. In the early stage of GF administration, such as direct injection or systemic local supplement, because of the accelerated degradation of bioactive BFs in vivo, the short half-life under physiological conditions and enzyme deactivation, the utilization rate of BFs is low. For example, the half-life of FGF-2 is very short, only t1/2 of 90 s in a wound zone, which limits its angiogenesis during tissue repair. Similarly, BMP-2, an unstable protein, that can be removed rapidly under conditions with t1/2 of 6.7 min after intravenous injection in non-human primates [42]. Therefore, because BFs have rapid diffusion/degradation, short half-life, and low effective dose in vivo, they are usually injected with super physiological dose repeatedly to hold their concentration at the site of injury. Currently, BMP-2 delivery in the form of a sponge-like gelatin is being topically administrated in clinics [43]. Because of dissolution or rapid hydration of the carrier, the rapid washout of BMP-2 from the target site is unavoidable [44].
BMP-2 is usually administered at an over physiological dose of rhBMP-2 (1.5 mg/mL) to compensate for this loss, but this amplifies the risk of heterotopic ossification, hyperplasia of bone, and other complications, including acute respiratory and neurological complications [45]. In addition, serious consequences that include systemic inflammation and reactive oxygen species (ROS) generation may be associated with high-dose BMP-2. Similarly, in vivo, IL-4 has a short half-life and has a limited therapeutic effect on a variety of inflammatory diseases. Therefore, it is inevitable that cytokines are administered in large doses with systemic side effects. Besides the short half-lives, another issue associated BFs-based therapy is the lack of non-specific targeting toward injured cells, from which severe side effects arise due to their multiple bioactivities. For example, the use of BFs in an over physiological dose raises concerns as it augments the risk of cancer [46]. A remarkable example is the Regranex® gel, which was the first product carrying GFs approved by the FDA for wound healing in diabetes, and its active ingredient is 0.01% recombinant human PDGF-BB. Nevertheless, the FDA warns that when three or more tubes are used, the incidence rate of malignancies increases. For example, VEGF can generate vascular permeability, leading to edema and systemic hypotension, so the released amount and rate of VEGF need to be properly controlled [47]. With the development of vehicles, it is possible to decrease the need for high doses, and the occurrence of side adverse reaction by allowing the control and sustained release of BFs.
The engineering strategies to deliver BFs in the field of bone repair
To improve the bioavailability and stability of proteins, several strategies have been adopted. Therefore, the application of GF delivery in tissue aims to enhance the incidence of recovering, limit excessive osteogenesis, and promote the recovering process. The following section of this review looks at the application of BFs in various forms of modulated biomaterials in bone tissue engineering applications, and summarizes some examples in Table 1 . Firstly, rigid BGSs were used to imprint BFs for large scale bone defect repair. And then the soft sponge-like scaffold carrying BFs was summarized as the filling materials for the cavity of the bone defect. Finally, various injectable vehicles including hydrogel, nanoparticles, and microspheres for the delivery of BFs were also introduced for irregular bone fracture (Table 2) repair. The underlining principle was based on improving the affinity of BFs to the engineering biomaterials. These principles include the controlled separation of recombinant proteins with specific ECM-binding motifs [48], covalently bound immobilized protein [49], surface modification by electrostatic bonding [50], and incorporation of specific BFs binding glycosaminoglycans (GAGs) or peptides, such as heparin sulfate into the scaffold [51].
Different Type of Cells Secretes Cytokines at Different Periods
| Factors | Cells | Cytokines | Period |
|---|---|---|---|
| Inflammatory factors | M1 macrophage | TNF-α, IL-6, CCL2, IL-1β | Initial stages of fracture |
| Angiogenic factors | Endothelial cells and osteoblasts | VEGF, FBFs, TGF-β | During fracture angiogenesis |
| Platelets and macrophages | PDGF | During tissue injury and fracture | |
| Osteogenic factors | M2 macrophage | BMPs, TGF-β1, FGF-9, FGF-18, PDGF | During the healing of bone tissue |
BMPs: bone morphogenetic protein; CCL2: C-C motif chemokine ligand 2; FBFs: fibroblast bioactive factors; FGF: fibroblast growth factor; IL: interleukin; PDGF: platelet derived growth factor; TGF: transforming growth factor; TNF-α: tumor necrosis factor-alpha; VEGF: vascular endothelial growth factor.
The Engineering Strategies to Deliver Single or Dual BFs for Promoting Bone Healing
| GFs | Bone Defect | Technique | Advantage in vitro/in vivo | Refs | |
|---|---|---|---|---|---|
| Inflammatory factors | IL-8 and BMP-2 | Radius diaphysis | Polymer coated MBG scaffoldss | The spatiotemporal delivery system of BMP-2 and IL-8 facilitated stem cell recruitment and the osteoinductivity | [37] |
| IL-4 | Periodontal bone | Heparin-modified gelatin microsphere | Switching M1 macrophage into M2 phenotype | [52] | |
| IL-2 and TGF-β | Alveolar bone | PLLA nanofibrous spongy microspheres | Rescuing periodontal bone loss through enrichment of regulatory T cells | [39] | |
| Angiogenic factors | IGF-1 and TGF-β3 | – | Meniscus-derived decellularized matrix | Chondrogenic differentiation induced by synovial fluid MSCs in vitro | [53] |
| BMP-4 and TGF-β1 | Alveolar bone | Liposomes | Enhancing the restitution process in rat tooth sockets | [54] | |
| TGF-β1 and BMP-2 | Tibia | Visible light-cured glycol chitosan gels | Greater bone formation abilities than that containing a single factor | [55] | |
| PDGF | – | PLGA microsphere incorporated scaffolds | Inducing differentiation of BMSCs to bone-like cells in vitro | [56] | |
| VEGF and BMP-2 | – | Microspheres-incorporated hydrogel | – | [57] | |
| Osteogenic factors | BMP-6 and IGF-1 | Periodontal regeneration | Chitosan/alginate/PLGA hybrid scaffolds | Controlled release and sequential administration of IGF-1 and BMP-6 induced the osteoblastic differentiation and proliferation of cementoblasts | [58] |
| BMP-2-encoding plasmid DNA | Ectopic osteogenesis | nHAC-collagen scaffolds | BMP-2 has a large output over a long period of time | [59] | |
| BMP-2 or FGF-2 encoding plasmid DNA | Long bone defects in a diabetic rabbit model | PEI complexes embedded in collagen scaffolds | PEI − (pBMP-2 + pFGF-2) scaffolds produced better bone regeneration than PEI-pBMP-2 scaffolds alone | [60] | |
| BMP-2 and IGF-1 | Femoral condyle osteochondral defect | Bilayered hydrogel scaffolds | Synergistically enhancing the degree of subchondral bone formation | [61] | |
| BMP-2 and FGF-2 | Calvarium | Study on PLLA yarn reinforced silicon coated nHAC–gelatin composite scaffold | The enhanced vascularization and new bone formation | [35] | |
| BMP-2 | calvarium | Hierarchical nanofibrous PLLA microspheres | A multiple-controlled release manner and enhanced bone regeneration | [62] |
BMPs: bone morphogenetic protein; FGF: fibroblast growth factor; GF: growth factor; IGF: insulin-like growth factor; IL: interleukin; MBG: mesoporous bioactive glass; PEI: polyethylenimine; nHAC: nanohydroxyapatite-collagen; PDGF: platelet derived growth factor; PLGA: poly(lactic-co-glycolic acid); PLLA: poly(l-lactic acid); TNF-α: tumor necrosis factor-alpha; VEGF: vascular endothelial growth factor.
Rigid BGS imprinting BFs
Natural demineralized/decellularized implanting grafts
ECM plays a basic role in coordinating and capturing the activity of GFs in vivo, and is an ideal device for tissue regeneration. ECM is an excellent GF library, because many GFs can bind to specific sites of the cell within ECM, including VEGF-A, BMP-2, PDGF-BB, and FGF. Bone-derived ECM, which originated from human cadaveric or animal allogeneic bone, was the commonly used for BGS due to its good mechanical strength.
The demineralized bone matrix (DBM) contains a small amount of inorganic phosphates, GFs, calcium-based solids, and some micro cell debris, and also retains many natural bone protein components. It is a kind of high acid extracted allogeneic bone derivative [63]. Before the advent of FDA approved synthetic bone transplantation, DBM scaffolds from allogeneic sources accounted for 50% of the total bone grafts. In the broad prospect of bone substitutes, porcine-derived DBM scaffolds also show great effect on human bone regeneration, which has been affirmed by the results of trials published in several studies [64, 65]. Moreover, porcine bone scaffolds were also successfully used as BF storage carriers. For example, porcine bone scaffolds exhibit time-dependent release of GFs, such as VEGF and bFGF, in vitro [66]. After calvarial implantation, the GFs-adsorbed scaffold significantly increased the markers for bone and vascularization and bone tissue formation. However, the large areas of cortical allografts were not well healed and often failed in 5–10 years after transplantation. A cortical allograft containing one or two GFs that allows the first release of GFs, followed by the later and more persistent release of another GF, to a certain extent controls the time transmission of one or two factors. VEGF and BMP are both the key components of bone formation and repair, and Farzana developed these two factors-loaded allogeneic bone transplantation. One of these two proteins was transplanted to the long cortical bone, causing VEGF to be released first, and then followed soon after by the release of BMP-2 [67]. When the allografts loaded with GF were placed in a critical size of rat femoral segment defect, the new bone formation around the allografts loaded with both BMP-2 and VEGF was statistically significant increased significantly in 4–8 weeks compared with that without GF.
As DBM has the ability to induce ectopic bone formation, more and more evidence has also proven that DCM from natural tissue has the ability to adjust to the differentiation of stem cell in tissue engineering applications. Hypertrophic chondrocytes differentiated from stem cells are the key types of cell in endochondral ossification. hASC are clinically-related stem cells that can differentiate into hypertrophic chondrocytes. Tissue engineered grafts were constructed by seeding ASCs into decellularized bone scaffolds followed by static culture in sequence initially for 2 weeks in chondrogenic medium to differentiate ASCs, and then in hypertrophic medium for 3 weeks to mature them into hypertrophic chondrocytes [68].
When these grafts are transplanted into severe femoral defects in athymic rats, ASC-derived hypertrophic chondrocytes in the osteogenic scaffold can promote long-term bone repair. In contrast, only 1/8 of osteoblasts and 3/8 of acellular scaffolds were implanted. Twelve weeks later, hypertrophic chondrocytes repaired 7/8 of the defects. Sometimes, the differentiation of stem cells into chondrocytes needs to be supplemented with some exogenous GFs. For example, DCMs have the ability to induce synovial-derived MSCs (SF MSCs) to differentiate into meniscus fibrochondrocyte (MFC) phenotypes, but it was required for the assembly of TGF-β3 and IGF-1 [53]. TGF-β3 combined with IGF1 can induce the production of DCM cartilage matrix and up-regulate the expression of collagen I, II, and aggrecan. In addition, under the effect of GFs, SF MSCs in DCM scaffolds showed a round morphology.
Synthetic bone scaffold substitutes
The severe deficiency of natural bone transplantation and the lack of opportunities have triggered the development of bone grafts and substitutes [69]. Artificial bone substitutes and biological factors have gradually become mainstream in bone transplantation procedures. Synthetic bone scaffold substitutes can be originated from inorganic materials or organic polymers.
Among these artificial bone substitutes, inorganic scaffolds and recombinant human bone morphological are the most widely used, and the inorganic scaffold material is mainly calcium phosphate [CaP, hydroxyapatite (HAP), ceramics, and CaP cements]. As the previous bone substitutes were only osteoconductive, they were mainly used for the rebuilding of large-area defects. However, rhBMPs are osteoinductive and can promote fracture healing.
Calcium hydroxyapatite is a kind of chemical similar to the mineral phase of calcified tissue, which can form CaP ceramics. Because of its excellent osteoconductivity, the CaP ceramics have been extensively used in clinical research. Several pivotal parameters such as absorptivity and the mechanical properties of CaP ceramics are closely affiliated to the Ca/P ratio, the Ca/P ratio of TCP and HAP, which are extensively used in orthopedics [70]. A promising bone tissue engineering biomaterial is synthetic polymer, which has good biomechanics and biodegradability. In addition, compared with other types of scaffolds, these have better controllability in porosity, physical and chemical structure, and immune adverse reactions [71]. Aliphatic polyesters, such as polylactic acid (PLA), polycaprolactone (PCL), polyglycolic acid (PGA) and their derivatives, are the most studied synthetic polymers in bone tissue regeneration. Other synthetic polymers include polypropylene, polyurethane, polycaprolactone, and polymethylmethacrylate. The characteristics of these polymers are that they can be easily cut into different shapes according to the mechanical requirements of the specific bone treatment. However, there are still some concerns about the absorption time of synthetic polymers, which may change their mechanical strength in vivo. In addition, for some polymers, including PGA, PCL, and PLLA, degradation can lead to the release of acidic compounds, which may pose adverse problems for natural bone health.
Soft sponge-like polyelectrolyte multilayer
A physical method of surface modification of biomaterials is to assemble charged polyelectrolytes into multilayer films. Multilayers can control the adsorption of proteins from the surrounding solution, and can be used as reservoirs to control the release of BFs. When they are used as implanted coatings in biomedical applications, both of them will affect the fate of cells. Moreover, the release of BFs from multilayers can be easily adjusted by controlling surface and intrinsic traits of multilayers, including mechanical properties, wettability thickness, and surface charge. For instance, taking chitosan as heparin, polycation, and chondroitin sulfate as natural polyanion or oxidized polyanion to form polyanion layer by layer [72]. Heparin oxide and chondroitin sulfate oxide can achieve additional stability by forming an imine bond between the amino group of polycation and the aldehyde of oxidized amino polysaccharides (oGAGs). BMP-2 was loaded into the oGAG-based multilayer for sustained release. BMP-2 multilayers can effectively support the proliferation and differentiation of C2C12 myoblasts into osteoblasts in vitro. In another study, oxidized sodium alginate (OSA) and chitosan coated bovine serum albumin nanoparticles (CBSA NPs) were used as film-forming materials and deposited on porous titanium scaffolds in a layer by layer (LbL) manner [73]. An LBL membrane has a micro/nano structure, a nano structure, and is rich in CBSA NPs and OSA functional groups, which improves the surface biocompatibility and biological function of titanium scaffolds. These groups can also provide stable and effective active sites for the immobilization of BMP-2 through physical and chemical interactions without affecting its biological activity. The results of in vitro release experiments showed that BMP-2 could be released for a long time. In addition, to enhance cell adhesion and proliferation, or to induce osteogenic differentiation of BMSCs, some researchers have used the hierarchical structure of the assembly membrane and BMP-2 fixation on the scaffold.
Recently, the combination of a biomimetic calcium phosphate (bCAP) barrier with poly lysine (PLLys) and poly glutamic acid-polyelectrolyte multilayer (PLGlut-PEM) has been found to be one of the minority systems that can transfer two factors; non-overlapping and continuous. PEM coating is composed of many layers, and polyelectrolyte molecules are deposited on the surface using various technologies, including immersion in alternative solutions for different charged molecules. The transportation of PEM coating and biomolecules can be adjusted by changing the temperature, pH value, ionic strength, and the molecule of PEM coating. It is polyelectrolyte multilayers that provide non-denatured reservoirs for GFs. Biomolecules are adsorbed into PEM/bCAP and other layered structures at different molecular intervals to simulate the embedding of biomolecules in ECM during bone formation.
In order to deliver dual factors for controlling the process of bone repair, a new type of poly(lactic-co-glycolic acid) (PLGA) microspheres was designed to release osteogenic growth peptide (OGP) and BMP-2 in a continuous manner [74]. First, OGP was impregnated in a simple solution and then entrapped into PLGA microspheres. Then, [(HA-CS)2-Hep-BMP-2-Hep-(CS-HA)2] multilayer polyelectrolyte coating was assembled on the surface of PLGA microspheres loaded with OGP layer by layer. The sequential release patterns of OGP and BMP-2 from PLGA porous microspheres was confirmed in vitro. On the first 15 days, the release of OGP was very minimal (< 17.1%), but it accelerated significantly thereafter, at least 60.3% of the load of BMP-2 was released, and then only very slow release was observed.
Biodegradable microspheres
Gelatin is extracted from collagen hydrolysate. The structure of amino acids is similar, and gelatin can be formed at a lower concentration. Although changes in net charge can be controlled by hydrolysis conditions, their immunogenicity is lower than that of full-length collagen, because of the removal of aromatic groups. Gelatin microspheres (GMPs) could be simple water-in-oil emulsification. Gelatin has undergone a sol–gel transition near 30 °C, so preparing stable microspheres by crosslinking GS matrix at physiological temperature is required. Various chemicals, including glyceraldehyde, formaldehyde, and glutaraldehyde, are used to crosslink gelatin. Recently, GS with low toxicity was prepared by using genipin as a crosslinking agent [44]. By precisely controlling the concentration and crosslinking time of genipin, the negatively charged GS was obtained, which is helpful to adsorb the positively charged BMP-2 (PI 8.5) into the GMPs through the multi-ion complexation. BMP2 was released from GMPs by degradation of the gelatin matrix mediated by macrophages. The release of GF can be better controlled by a controlled release system synchronized with inflammatory response, so as to improve the prognosis of refractory wounds. But it was difficult to sequester IL-4 in GS by electrostatic interaction, let alone control its release behavior. In this case, a modified gelatin was developed. For example, a novel heparin-modified gelatin was synthesized via an 1-ethyl-3-(3-dimethylamino) propyl carbodiimide/N-hydroxysuccinimide (EDC/NHS) coupling, considering that heparin has a high affinity for IL4 and stabilizes this cytokine, it can prevent its denaturation and degradation, so as to prolong its release time. Heparin modified gelatin porous microspheres were prepared by emulsified phase separation. IL-4 was loaded into microspheres as an injectable scaffold for bone immune regulation [52]. To provide microenvironment of osteoconduction for bone marrow stem cells, heparin modified gelatin nanofiber self-assembled microspheres can be used to simulate the structure of natural bone ECMs.
Polyester-based polymers, such as PLGA, have been widely used to control peptide or protein delivery due to their good biodegradability, non-toxicity, biocompatibility, strong adhesion, and bacteriostatic [75]. In recent years, some BFs such as rhBMP-2 and PDGF-BB with PLGA have been used to prepare nano carriers for efficient protein transfer in vivo [76]. For example, PDGF-BB is re-embedded into PLGA microspheres by typical water-in-oil-in-water (W/O/W) emulsion solvent evaporation to realize the controlled release of PDGF-BB on stem cell differentiation in a three-dimensional (3D) polymer network [56]. However, due to the exposure of proteins bound in polymeric carriers to a range of pressures, such as detergents during formulations, hydrophobic interfaces, vigorous agitation, as well as temperature-elevated, polymer degradation by-products and enhanced moisture during transport, the retention of their activities are still challenged for PLGA-based micro/nanoscale carriers [77]. To overcome these drawbacks, hierarchical nanofibrous microspheres were developed to deliver BMP-2 [62]. Heparin-conjugated gelatin (HG) was synthesized for the first time. It provides a binding domain for BMP-2, stabilizes the GF of BMP-2, prevents its proteolytic enzyme degradation and denaturation, and thus prolongs its release time. Secondly, BMP-2-bound HG nanospheres were encapsulated in PLLA nanofiber microspheres via the water-in-oil-in-oil (W/O/O) double emulsion process (Figure 3A).
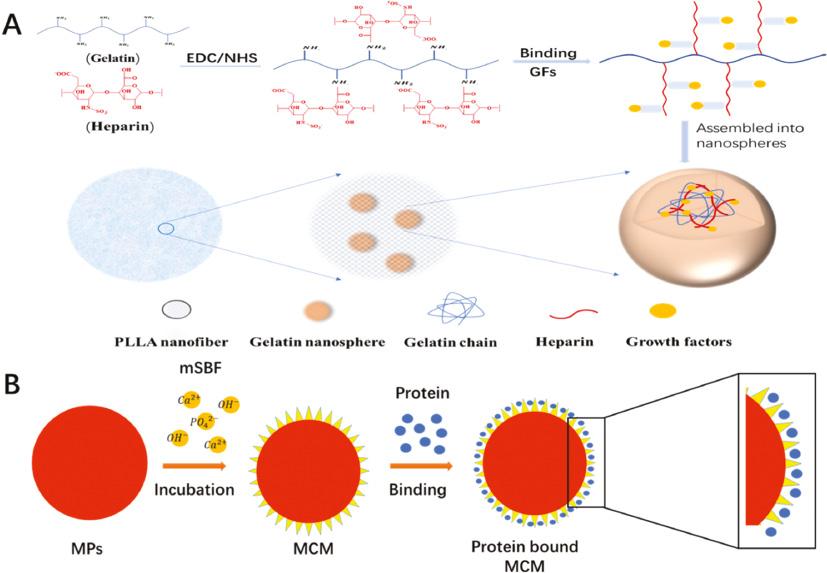
(A) Schematic diagram of hierarchical microspheres: GF is bound to heparin and encapsulated in gelatin nanospheres, which are further immobilized in PLLA microspheres; MCM [62]. (B) bFGF binding on the mineral coating of MCM surface. (Adapted from [78].) bFGF: bone foration growth factor; GF: growth factor; MCM: mineral coated microspheres.
The prepared hierarchical nanofiber microspheres have the feature of superior surface area, low density, and high porosity, which makes them good carriers for cell adhesion and tissue growth. BMP-2 is released by conjugation with heparin, wrapped up with nanospheres and microspheres in various controlled manners. This microsphere is proven to be an superior osteoinductive scaffold for increased bone regeneration of calvarial defect models in vivo. However, glutaraldehyde as a chemical crosslinking agent might compromise the activity of BMP-2 in hierarchical nanofibrous microsphere in this study.
Porous mineral micro/nanoparticles
Recently, a novel biomimetic microsphere was developed to deliver BFs under gentle preparation conditions. It was inspired by the fact that natural bone tissue can preserve biomacromolecules embedded in bone matrix for thousands of years after an individual dies [79]. Nanostructured mineral coatings on microspheres were probed to stabilize GFs for therapeutic delivery. The nanostructured mineral coating was prepared on PLGA microspheres by incubation in modified simulated body fluid (mSBFs), and bFGF was combined with mineral coating by electrostatic interaction of protein side chains and charged surface of mineral coating (Figure 3B) [78]. bFGF released by nano structure mineral coating can keep its bioactivity for several weeks in the process of release, while in the process of PLGA microspheres release, the bioactivity of bFGF can be kept for less than 7 days.
Some porous inorganic materials have been modulated into micro/nanoparticles for delivering BFs. In vitro, the electrolyte-coated HAP particles have been proven to be able to bind GFs electrostatically and have adjustable sustained release characteristics [80]. HAP particles were first coated with electrolyte in mSBF to obtain the mineral-coated microparticles (MCMs). BMP-2 was electrostatically absorbed onto the porous surface of MCMs. Local application of BMP-2-coated MCMs improved bone healing in challenging mouse models of non-union. After 10 weeks, BMP-2-coated MCM-treated mouse femurs appeared to have complete osseous bridges, while in untreated femurs the non-union rate was 100%.
Alternatively, due to its porous channels, mesoporous silica nanoparticles (MSNs) with particle size less than 200 nm, and with high colloidal stability have been extensively studied for the systematic delivery of GFs. As an effective drug carrier, MSNs have attracted attention due to their unique surface structure, large surface functionalization ability and pore volume [81]. Different hydrophilic or hydrophobic drugs can be loaded and released by controlling the microporous structure, surface group modification and pore size of MSNs [82]. The uniform and controllable size MSNs (XL-MSNs) with 30-nm pore size were prepared by embedding IL-4 with organic additives and inorganic nanoparticles [83] (Figure 4). The loading of XL-MSNs to different molecular weight model proteins was significantly higher than that of traditional microporous MSNs. The results showed that IL-4 loaded XL-MSNs successfully triggered the polarization of M2 macrophages in vivo, indicating that the XL-MSNs have the clinical potential to target a variety of cytokines to regulate the immune system.
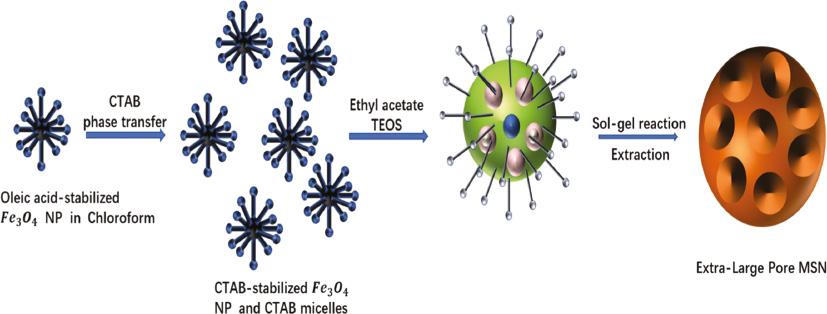
Synthesis of large pore MSNs (XL-MSNs)(83). (Adapted from [83].) MSNs: mesoporous silica nanoparticles.
Injectable hydrogel for healing irregular bone fracture
Hydrogel constructed of a 3D hydrophilic polymer chain has a unique configuration and tunable physicochemical characteristics, which can provide a nutritional environment for endogenous cell growth. In addition, hydrogels can be customized to obtain the desired injection, porosity, degradation rate, and the release curve can facilitate control by changing the crosslinking degree and method. Hydrogels have been extensively studied and applied in the field of bone repair, because hydrogels can be administered in a minimally-invasive way in vivo, and filled with irregular bone cavities. Although hydrogels have many good qualities, they also have limitations, such as, bad tissue adhesion, which may be detrimental to integration with host tissue.
Natural composite-derived hydrogel
Natural polymer material is a component of natural ECM, or is similar to natural ECM, with a low immune response, favorable biocompatibility, and low cytotoxicity, which can advance cell adhesion and new tissue regeneration. Various polymers have been developed as carriers and type I or II collagen and have already draw attention for the preparation of collagen hydrogels [84]. Collagen hydrogel can simulate 3D living microenvironment and fill any shape damage ability, and has been extensively applied in tissue engineering [85]. Type I collagen has good biocompatibility and biodegradability [86], it can induce MSCs to differentiate into chondrocytes without adding biological or chemical materials. And it is easy to form hydrogels through self-assembly [87].
One of the natural ECM components of cartilage is type II collagen which can promote the secretion of cartilage specific ECM, regulate the proliferation of chondrocytes, retain the phenotype of chondrocytes, and induce cartilage regeneration. However, under physiological conditions (pH 7.4 37 °C), type II collagen formed short self-assembled nanofibers [88]. Hydrogels with a particular mechanical strength are difficult to form. But, an injectable type II collagen gel can be prepared by adding a polyethylene glycol cross-linking agent and a II type collagen chemical reaction [89]. Collagen hydrogel matrix was used to fill the nasal dorsal bone defect in rats. After 6 weeks, a thin layer of bone appeared on the surface of the injured part. The recovering area of the nasal cavity in rats without gel was less than 7%, thus it is thought that collagen gel played a positive role in the repair of defects. Some ideal properties of the material are necessary for clinical implant surgery. However, natural polymer hydrogels can only be used in non-load-bearing parts due to their poor mechanical strength.
Glycosaminoglycans, such as heparan sulfate (HS), bind to and promote the activity of chondrogenic GFs, so they have the potential to target tissue regeneration. The binding affinity of HS with several proteins (TGF-β1, BMP-2, PDGF-BB, and VEGF) was confirmed using surface plasmon resonance-based analytics [90]. When these HS-gels were implanted into a large osteochondral defect of the femoral trochlea in New Zealand white rabbits, both subchondral and chondral tissue exhibited an increased hyaline cartilage in comparison with a hydrogel control over a 12-week period.
Chitosan (CS), a deacetylated derivative of chitin, used as a drug carrier, can promote the absorption of hydrophobic macromolecules. CS nanoparticles (CSNPs) can reduce the toxicity and prolong the activity of the drugs by putting the therapeutic agents in close contact with the action site because of their adhesive cation properties.
CS, composed of acetylglucosamine and glucosamine, is similar to glucosamine in structure and exists in cartilage ECM, and is similar to glycosaminoglycan in structure and exists in cartilage ECM [91]. CS and its derivatives are widely used as drug carriers and in tissue engineering due to their good structural properties and biodegradability [92]. Hyaluronic acid (HA) is a huge molecular weight natural polymer made of D-glucuronic acid and N-acetyl-D-glucosamine. It is the main component of cartilage extracellular matrix [93]. Chitosan uronic acid hydrogel has the capability of high-water content, embedding cells and retaining the phenotype of cells. It is used in drug delivery and cartilage tissue engineering in vitro, and is an ideal material for cartilage repair [94]. The preparation method is to use periodate to oxidize sodium hyaluronate, then dialysis to remove periodate and freeze-drying to make hyaluronate dialdehyde. Add 2% CS solution, neutralize with HEPES buffer (pH 7), and stir evenly. CSHA dialdehyde gel was prepared by reaction of CS with a Schiff base [95]. When chondrocytes are combined with appropriate biomaterials, they help protect unaffected cells and accelerate cell proliferation and ECM deposition [96]. In the animal model of severe cartilage defect of New Zealand white rabbits, the severe cartilage defects in the knee joint of rabbits were implanted into the hydrogel by allogenic rabbit chondrocytes. The regenerated cartilage was analyzed 12 weeks after implantation. Morphological scores showed that all animals formed repair tissue within 12 weeks.
The synthesized or modified hydrogel
Polymer hydrogels can easily adjust their properties and functionalization to summarize the molecular cues of ECM. A new type of chitosan glycol (GC) GF sustained-release hydrogel for injection was prepared and its effect on promoting bone healing was studied [97]. Methoxypolyethylene glycol carboxyl (mPEG-COOH) was coupled with GC by condensation to a reaction resulting in mPEG/GM-g-GC. Glycidyl methacrylate (GMA) was used as a crosslinking agent and covalently coupled with the mPEG-g-GC amine group. The mPEG/GM-g-GC chain was crosslinked to form a gas phase gel by visible light irradiation. An injectable hydrogel system based on visible light curable ethylene GC hydrogel was developed for the BMP-2 and/or TGF-β1 of bone formation [55]. The developed hydrogel system showed that the controlled release of GFs lasted for 30 days. The hydrogel load GFs was injected into the tibial defect with a syringe. After 4 weeks of treatment, mineralized bone was formed by GC hydrogel loaded with GF. In addition, the osteogenic capacity of GC hydrogels containing BMP-2 (10 ng) and TGF-β1 (10 ng) in vitro and in vivo was higher than that of hydrogels containing BMP-2 (5 ng) and TGF-β1 (5 ng).
HA, a biomaterial, has lately been used in bone surgery. HA is an important component of ECM and belongs to the glycosaminoglycan family [98]. HA not only protects the integrity of the ECM structure, but also has a bacteriostatic effect and is highly compatible with water [99]. HA induces the production of fibroblasts, pro-inflammatory cytokines, etc., and stimulates the synthesis of vascular endothelial cells. It promotes bone regeneration, proliferation, differentiation, and induction of osteogenesis. It also up-regulates osteocalcin gene expression at an early stage, and interacts with DEX and rh BMP-2 to affect cell proliferation and differentiation [100]. HA can also interact with hyaluronan-mediated motility receptor (RHAMM) and the CD44 receptor, it promotes stem cells to differentiate into chondrocytes through CD44 receptor, and activates various signaling pathways [101]. It can interact with cells and proteins involved in bone metabolism and contribute to bone healing. And has been used as the material of choice of areas such as orthopedics.
HA has remarkable mechanical properties and a high tolerance structure. It is a potential bone regeneration agent and a delivery system for bone healing. The healing effect of HA on bone defects in different parts has been studied in many aspects. Koca et al. [102] compared the effect of HA and/or bone transplantation on bone defect healing, and concluded that the efficacy of using HA is similar to the use of bone grafts. But HA has a gel form after contact with blood, and has a certain viscosity. Thus, when used in combination with a graft material, it can ensure uniform distribution and fixation of graft materials. Thus, the combination of the two can accelerate bone healing more than when used alone. Aslan et al. [103] found that HA combined with bone transplantation could be used to establish a rabbit tibial defect model. After 40 days, bone formation was found in the defect area. HA can be used as a bone healing material or as gel coated GFs in the treatment of bone fracture.
HA is an ECM component, but without crosslinking it was difficult for HA to control the release of BFs because of its poor gelatin properties. To improve its gelatin time and tuning mechanical properties, multiple types of HA-based hydrogels have been developed by crosslinking reactions. For example, hydrazine crosslinked HA hydrogel was prepared by mixing hydrazide-modified HA derivatives and aldehyde-modified HA derivatives. Hydrazine crosslinking shows good stability in physiological conditions, which is attributed to the wide non-locality of the charge on the urea type bond [104]. The results showed that adjusting the primary protonation state of HA carboxylic acid in hydroquinone crosslinked HA hydrogels could adjust the gel time and thus regulate its BMP-2 release curve under physiological pH [50]. The time sweep rheology experiment showed that the gel time of acidic hydrogel was shorter (90 s), and the gel time of neutral hydrogel was 13 min. Moreover, in neutral HA hydrogels, the release of BMP-2 is mainly controlled by Fickian diffusion, while the electrostatic interactions and diffusion between BMP-2 and HA become more important under acidic pH. After subcutaneous injection of 4 mg rhBMP-2 into 200 ml acidic or neutral HA hydrogel, the bone formation of BMP-2 loaded acidic hydrogel was better than neutral hydrogel in rat ectopic model. Besides, ultraviolet (UV) crosslinked HA-based hydrogels were prepared by acrylic acid or methacrylic acid. These hydrogel scaffolds can be easily patterned by using a mask or stamp in the process of a UV gel. However, when UV technology is used to form hydrogels, the side effects that may occur during the formation of the network should be noted. The free radicals produced by UV irradiation can change the molecular structure of BFs. In contrast, the selectivity and cytocompatibility Michael type addition of maleimide and thiol groups (“click chemistry”) was used as a network formation reaction to minimize the reaction between hydrogel materials and collagen or amino groups of BFs [105]. A hydrogel composite consisting of collagen type I, maleimide functionalized heparin, and starPEG peptide conjugates, was constructed. Composite gels were easily functionalized with stromal cell-derived factor-1 (SDF-1), VEGF, and bFGF because of their high affinity to heparin.
Temperature sensitive hydrogels
Thermosensitive hydrogels are attractive materials because they can be used directly as liquid in defective parts and then be gelled in situ at body temperature. A Laponite® (BYK Additives Ltd, Widnes, UK) crosslinked pNIPAM-co-DMAc hydrogel loaded with HAP nanoparticles has been developed [106]. It is not necessary to add chemicals for gelation or clean-up, and hydrogels are rapidly gelatinizated at body temperature. Moreover, we found that no osteogenic GF was needed, and hydrogel could induce osteogenic differentiation of MSCs [107]. Poloxamine-based hydrogel has osteogenic activity and has been used in bone regeneration research. Poloxamine, which is marketed as Tetronics, is a X-shaped copolymer composed of four polyethylene oxide-polypropylene oxide arms the structure of which is formed according to the concentration, pH value, and temperature. The sol-to-gel transition of T dispersions occurred at about 26 ºC, and the viscosity was enhanced as the temperature increased to 37 ºC. A new type of injectable thermo responsive hydrogel was designed for local application to repair calvaria critical bone fractures in octylphenol (OP) female rats [108]. The hydrogel system consists of sodium alginate crosslinked calcium chloride enhanced poloxamine (T-1307).
It has also been claimed that injectable hydrogel can firmly adhere to the cavity of bone tissue after administration. Therefore, in situ photo crosslinking platelets containing plasma composite hydrogel was constructed made of o-nitrobenzyl alcohol (NB) modified HA as a material to promote the repair of cartilage defects [109]. Under light irradiation, HA-NB produces aldehydes, which react with amino groups that are distributed on another component, such as CS, and form hydrogels in situ, and achieve the adhesion and integration of covalent hydrogels. Additionally, platelet rich plasma (PRP)-complexed HNPRP hydrogel could easily complex with PRP, because of the imine crosslinking reaction between amino groups in the components of PRP and the photoinduced in aldehyde groups of HA-NB. HNPRP hydrogels can be conveniently formed to achieve the controlled release of GFs. The most important thing is to enhance the adhesion and integration of PRP hydrogels, thus overcome the shortcomings of clinical application of PRP.
Microspheres-incorporated hydrogel
Because of the rapid diffusion of drugs in microspheres, the phenomenon of explosive release of undesirable drugs reduces the effectiveness, thus limiting the application of delivery vehicles. However, research shows drug-loaded microspheres (MS) can be incorporated into composite hydrogels to effectively solve the burst release of drugs.
BMP-2 and PRGF in PLGA microspheres as bone active substances were incorporated into the thermo-responsive hydrogel for delivery to the site of damage. The drug was released in a fast phase (3 days) during 6 weeks, followed by a slow phase 12 weeks later, histological and histomorphometry analysis showed that BMP-2 had a synergistic effect on hydrogels. But the PRGF did not increase bone repair. GFs could be also loaded inside porous GMPs followed by embedding them within the matrix of oligo-poly-ethylene glycol fumarate (OPF) hydrogel to act as a GF delivery vehicle. These microspheres-incorporated OPF hydrogel composites have been used for single GF delivery such as TGF-β1 [110], and dual GFs delivery such as IGF and BMP-2 [61] to repair cartilage and osteochondral tissue.
Sometimes, the outer hydrogel matrix could also serve as a barrier to prevent phagocytosis of GF-loaded MS by the immune cells. After injecting BFs-containing microspheres in the bone defect zone, macrophages would recognize them, and would finally start the degradation and clearance of the drug delivery system, thus affecting the repair of exogenous BFs. The encapsulation of PLGA microspheres in high-structure collagen hydrogels can prevent them from being detected and maintain the integrity of BFs. Confocal laser microscopy showed that non-embedded microspheres were easily internalized, and when concealed, J774 and bone marrow-derived macrophages (BMDM) could not detect them. Moreover, the release kinetics of reporter protein remained unchanged in the presence of macrophages only when microspheres was concealed.
BFs-based gene delivery strategies
The scaffold was immersed in the solution containing GFs to prepare the combination of scaffold and GF. In this method, the release of GFs depends on the degradation of the scaffold, and the activity of GFs decreases in a time-dependent manner. Therefore, it is hard to release enough GFs during tissue regeneration. In order to achieve the gradual release, the gene transfection techniques were recently focused. The application of viral vector scaffolds has been proved successfully in vitro and vivo. In the critical size bone defect (CSD) model, the bone regeneration behavior of BMP-2 plasmid DNA (pBMP) and BMP-2 was compared [111]. RhBMP-2 (62.5 μg) or pBMP-2 (2.5 μg) were implanted into polylactide coated titanium discs and imbed into the damage area. RhBMP-2 initially induced the formation of ectopic new bone, and then remolded in an unorganized hypo dense mode. In contrast, pBMP-2 results in slow but stable physiological tissue morphology and bone regeneration. Moreover, rhBMP-2 triggers rapid ectopic but inadequate bone formation, while pBMP-2 successfully induces locally physiological bone regeneration.
CaP is the principal component of hard tissues, such as bones and teeth. It has high compatibility with siRNA, DNA, peptides, and proteins. Multi-shell calcium phosphate nanoparticles were exploited to load peptides or genes. For example, DNA-functionalized CaP nanoparticles encoding BMP-2 were prepared and combined with a nanohydroxyapatite-collagen (nHAC) scaffold for the gene transfection of human MSCs. The results demonstrated that the controlled-release of BMP-2 was maintained over 10 days [112]. The CaP bearing scaffold was grafted into the subcutaneous of the back of the rats, and obvious ectopic osteogenesis was observed [59].
Opinion
Fracture is the most common injury in human. Once injured, takes a long time to recover. In many treatment methods, GFs can effectively accelerate bone healing. At present, research in GFs has increased with the growing understanding of its involvement in bone healing, such as using BMP-7 to repair femoral defect in rats [113], signal transduction of BMP, FGF, and TGF promotes the differentiation of periosteal cells, which leads to the formation and stability of cartilage and bone in vivo [114].
Nevertheless, there are still some problems that cannot be solved, such as poor stability of GFs in physiological environment, poor biological activity, obvious adverse reactions [41]. At present, the clinical application of GFs is not satisfactory, and often cannot achieve good therapeutic effect. Therefore, it has become a research hotspot to find suitable carriers to delivery GFs. There are many carriers carrying GFs for treating bone defects, but different carriers have their own advantages and disadvantages. This review briefly refers to rigid BGSs, soft sponge-like polyelectrolyte multilayers, microspheres, nanoparticles, and hydrogel. BGSs have been shown to have a great effect on bone regeneration, which is confirmed by clinical trials published in some studies [64]. However, for large-scale bone transplantation, the therapeutic effect is very limited, and the transplantation fails after 5–10 years. The advantage of synthetic substitutes is that they can be cut into different shapes according to the wounds. But the stability in vivo is still questionable. Thus, the strategy of bone transplantation remains to be developed. Soft sponge like polyelectrolyte multilayer is a kind of multilayer, which is based on surface modification of biomaterials and assembly of charged polyelectrolytes into multilayers. Then, the release of BFs from the multilayer can be regulated by controlling the thickness, mechanical properties, surface charge, pH, and temperature of the multilayer. Although it can regulate the release of BFs by many means, but it is also easily influenced by these factors in clinical application. For microspheres and nanoparticles, their advantages are strong targeting, slow-release, and high-efficiency transmission, which can greatly improve the bioavailability of GFs. The problem of easy degradation of GFs can be effectively solved by preparing different size particles and loading different amounts of GFs. However, there are still some problems in clinical preparation, such as chemical crosslinker, encapsulation efficiency, and particle size [115]. Hydrogel is also a very suitable carrier with a unique structure and adjustable physical and chemical properties [116]. Now many modified hydrogels have better properties, such as adhesion, fluidity, and even temperature sensitivity. It can be completely attached to the defective surface, adhered to it, and wrapped to the whole part. By changing the crosslinking method and degree, the degradation rate and release GFs are easy to control. Especially the thermosensitive hydrogels [117]. At low temperatures, it can be injected into the damaged part easily when it is in a flowing state. Then, under the body temperature, it will quickly become a gel, which will wrap around the injured parts completely and effectively treat the fracture.
In contrast, different carriers have different advantages. Gel as a carrier to bone defects is effective because it not only can effectively transfer GFs, but also has a certain adhesion, which can effectively immobilize tissue in clinical application and prevent bleeding from the injury site. The degradability, fluidity, and adhesion of gels can be controlled by the crosslinking degree and different crosslinking methods, so that they can maintain the sustained release of GFs. And different from microspheres and nanoparticles, the drug delivery method is simple, as it can be injected, and sustained controlled release can greatly reduce the amount of drug delivery to avoid the need for frequent medication, so that the compliance of patients is greatly improved.
Conclusion
Fracture is generally divided into three stages: hematoma formation, callus formation, and bone formation. Multiple GFs are related to fracture healing, and their joint action can stimulate the activity of osteoblasts and regulate local osteogenesis. BFs combine with receptor, activate intracellular signal transduction, activate transcription factors into the nucleus, and induce gene expression. The synergistic effect of cell and cytokine can effectively cure bone injury. BFs play a key role in all stages of bone repairs, so BFs are extensively used in the treatment of bone fracture. However, due to its short half-life, poor protein stability, poor stability, and rapid enzyme degradation and deactivation under physiological conditions, the utilization rate of the biological activity of BFs is low, which limits its application in medical tissue engineering and hinders the transformation to clinical treatment.
To improve the bioavailability and stability of the protein, GF has been applied together with various kinds of biomaterials. For example, rigid BGS imprinted BFs and BFs were contained in various injection carriers, nanoparticles, microspheres and hydrogels. Among them, rigid bone grafts include natural demineralized/acellular implants and artificial bone scaffold substitute. Natural acellular grafts are a good GF library, because many GFs can combine with specific parts of cells, but the natural bone transplantation materials are in serious shortage and there is a lack of supply, which has led to the development of artificial bone scaffold substitutes. Among these artificial bone substitutes, CaP-based inorganic scaffolds and rhBMPs are widely used. In addition, the charged polyelectrolyte can be assembled on the surface of the charged biomaterial layer by layer to shape a multilayer to modify the surface of the biomaterial. The release of BFs from multilayers can be regulated by controlling the thickness, surface charge, mechanical properties, and wettability of multilayers. Microspheres and nanoparticles can also target and deliver many kinds of cytokines to the damaged tissues, and slowly control the release GFs. Injectable hydrogel consists of 3D hydrophilic polymer chains with unique structures and adjustable physical and chemical properties. It is easy to control the porosity or release curve, and degradation rate by changing the crosslinking method and degree, and it can be administered in a minimally-invasive way in vivo, and filled with irregular bone lumen. There are also various modified hydrogels and thermosensitive hydrogel encapsulated BFs for the treatment of bone injuries. Although these engineered biomaterials can successfully improve the bioavailability of BFs, there are still some problems, such as the rapid explosive release of loaded substances, the inadequate release curve of compounds in biomaterials and so on. In addition, postoperative complications, disputes over off label use, and high application costs have also been recorded. Therefore, the effective delivery strategy of BFs has not been fully implemented. With the continuous development of bone engineering, the progress of basic science will be further applied in the clinics.

