INTRODUCTION
Staphylococcus aureus is a common member of the human microbiota within the upper respiratory tract and on the skin, with approximately 30% of the healthy human population being S. aureus carriers [1,2]. S. aureus may also cause a wide range of severe and life-threatening diseases, including skin and soft tissue infections, implanted medical device-related infections [3,4], and bacteremia, which is associated with a high mortality rate [5].
S. aureus toxigenic strains produce virulence factors, such as toxic shock syndrome toxin-1 (TSST-1), staphylococcal enterotoxins (SEs), and Panton-Valentine leukocidin (PVL) [6]. Heat-stable SEs are regarded as the main cause of staphylococcal food poisoning (SFP) [7,8]. Greater than 20 different SEs and staphylococcal enterotoxin-like (SEls) toxins comprise a superfamily of pyrogenic toxin superantigens (SAgs). The classic enterotoxins (SEA, SEB, SEC1, SECbov, SED, and SEE) have been reported to account for 95% of SFP cases. The clinical characteristics of SFP cases include a short latency (average, 4.4 h), nausea, headaches, violent vomiting, abdominal cramping, and diarrhea [9]. Although SFP is usually a self-limiting illness and seldom causes life-threatening conditions, SFP is associated with significant physical discomfort and a financial burden. Of note, frequent outbreaks of SFP lead to serious public health, food industry, and catering business challenges [10,11]. One hundred twenty-eight foodborne illnesses caused by S. aureus were reported in the US and European Union in 2017, and SEs were responsible for 61 foodborne outbreaks (FBOs), representing 1.5% of all outbreaks reported in 2021 [12,13]. Ninety-four FBOs were attributable to SFP in China between 2003 and 2007, affecting 2223 individual patients and resulting in 1186 hospitalizations [14]; however, the actual incidence of SFP could be much higher because sporadic cases are easily overlooked and not reportable.
In addition, a growing number of multidrug resistant (MDR) strains [15], especially methicillin-resistant Staphylococcus aureus (MRSA), have been reported from food products in recent years [16]. These MDR or MRSA strains could also lead to human infections via various routes, which limits clinical treatment options and poses a serious clinical threat with persistent high morbidity and mortality rates [17].
Ready-to-eat (RTE) food products have become popular among consumers for the convenience they offer; however, retail RTE foods are susceptible to microbial contamination due to handling during packaging, sterilization, transportation, storage, and sales. Therefore, food poisoning readily occurs after contamination by foodborne pathogens. It was reported that 4.3% (1150/27,000) of retail foods were contaminated by S. aureus in China in 2015 [18]. The percentage of contaminated foods was much higher in RTE foods, which aroused a major public health concern [19–21]; however, the epidemic characteristics of MRSA from RTE foods in China have not been established. The aim of this study was to determine the prevalence, antimicrobial resistance, virulence profiles, and molecular characteristic of MRSA affecting RTE foods in China.
MATERIALS AND METHODS
S. aureus isolates
The 276 RTE food-associated S. aureus isolates used in this study were collected from 25 provinces across China in 2018. The RTE food samples were collected from supermarkets, farm product markets, convenience stores, retailers, fast food restaurants, and take-out restaurants, and included a variety of food types (salads, Chinese dim sum, cereal products, sushi, sashimi, ice cream, and sauced meats). All S. aureus isolates were confirmed using API STAPH test strips (bioMerieux, Marcyl’Etoile, France) and polymerase chain reaction (PCR) amplification of the 16S rRNA and nuc genes, as described previously [22]. The S. aureus isolates were then screened for MRSA by amplifying the mecA gene by PCR, according to a previous method [23]. All confirmed isolates were stored in brain heart infusion broth with 20% glycerol (Land Bridge, Beijing, China) at −80 °C.
Antimicrobial susceptibility testing (AST)
AST of all 276 S. aureus isolates were performed using Biofosun® Gram-positive panels (Fosun Diagnostics, Shanghai, China) and the microbroth dilution method [24]. A panel of 13 antimicrobial agents were estimated, including linezolid (LZD [0.25–8 μg/mL]), vancomycin (VAN [0.5–16 μg/mL]), daptomycin (DAP [0.125–8 μg/mL]), gentamicin (GEN [1–32 μg/mL]), trimethoprim-sulfamethoxazole (SXT [0.125/2.4–8/152 μg/mL]), chloramphenicol (CHL [1–64 μg/mL]), ciprofloxacin (CIP [0.125–8 μg/mL]), tetracycline (TET [0.5–16 μg/mL]), clindamycin (CLI [0.125–8 μg/mL]), erythromycin (ERY [0.125–8 μg/mL]), oxacillin (OXA [0.25–16 μg/mL]), cefoxitin (CFX [0.25–16 μg/mL]), and penicillin (PEN [0.06–8 μg/mL]). The S. aureus ATCC™ 29213 strain was used as the quality control for AST.
DNA extraction
All MRSA isolates were cultured on brain heart infusion agar (BHA; Land Bridge) at 37 °C overnight. Genomic DNA of the MRSA isolates was extracted using a bacterial DNA kit (Omega Bio-tek, Norcross, GA, USA) according to the manufacturer’s procedures. DNA was detected using a NanoDrop-2000 spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA). The qualified DNA was stored at −20 °C until analysis was performed.
Detection of virulence factors
In this study 13 enterotoxin genes (sea, seb, sec, sed, see, seg, seh, sei, sej, sell, selk, and selq), as well as the lukS/F-PV and tsst-1 genes in MRSA isolates were investigated by PCR amplification, as previous described [23,25].
Multilocus sequence typing (MLST)
Seven housekeeping genes (arcC, aroE, glp, gmk, pta, tpi, and yqiL) were used in the MLST analysis, as described previously [26]. The alleles and sequence type (ST) were assigned based on the PubMLST database (https://pubmlst.org/). Clonal complexes (CCs) were annotated based on the geoBURST Full MST algorithm using phyloviz software with a primary founder surrounded by single locus variants (SLVs) and known CC type in the MLST database [27].
spa typing
The spa typing for all MRSA isolates was analyzed, as described previously [28]. The spa repeats and types were assigned using BioNumerics software (v7.5; Applied Math, Sint-Martens-Latem, Belgium).
Staphylococcal cassette chromosome mec (SCCmec) typing
MRSA is endowed with resistance to β-lactam antibiotics by obtaining SCCmec elements. The three basic genetic elements (mec and ccr gene complexes, and J region), SCCmec elements were classified into different types with multiplex PCR methods, as described previously [29].
Pulsed-field gel electrophoresis (PFGE)
PFGE was performed as described by Murchan et al. [30] and Salmonella Braenderup H9812 was used as a DNA size standard strain for the PFGE gels [31]. Briefly, the isolates were cultured on BHA (Land Bridge), then the cell suspensions were prepared for PFGE plugs. The genomic DNA of MRSA and H9812 were digested with restriction enzymes (SmaI and XbaI, respectively; New England Biolabs, Ipswich, MA, USA). Electrophoresis was performed through a 1% agarose SeaKem Gold gel in 0.5 × TBE buffer using a CHEF DR III system (Bio-Rad, Hercules, CA, USA) at 14°C for 19 h. The initial and final pulse time were 4.0 s and 40.0 s, respectively. After the gels were stained with GelRed (Biotium, Fremont, CA, USA), images were obtained using a ChemiDoc XRS+ system (Bio-Rad). The PFGE patterns and phylogenetic relationships were analyzed using BioNumerics software (v.7.5).
RESULTS
Isolation of S. aureus and MRSA in RTE foods
A total of 276 S. aureus isolates were cultured from various RTE food items in 25 China provinces (Fig 1A). The food samples included salads, Chinese snacks, cereal products, sushi, sashimi, ice cream, and sauced meats. Of these isolates, 30 strains (10.9% [30/276]) were MRSA.
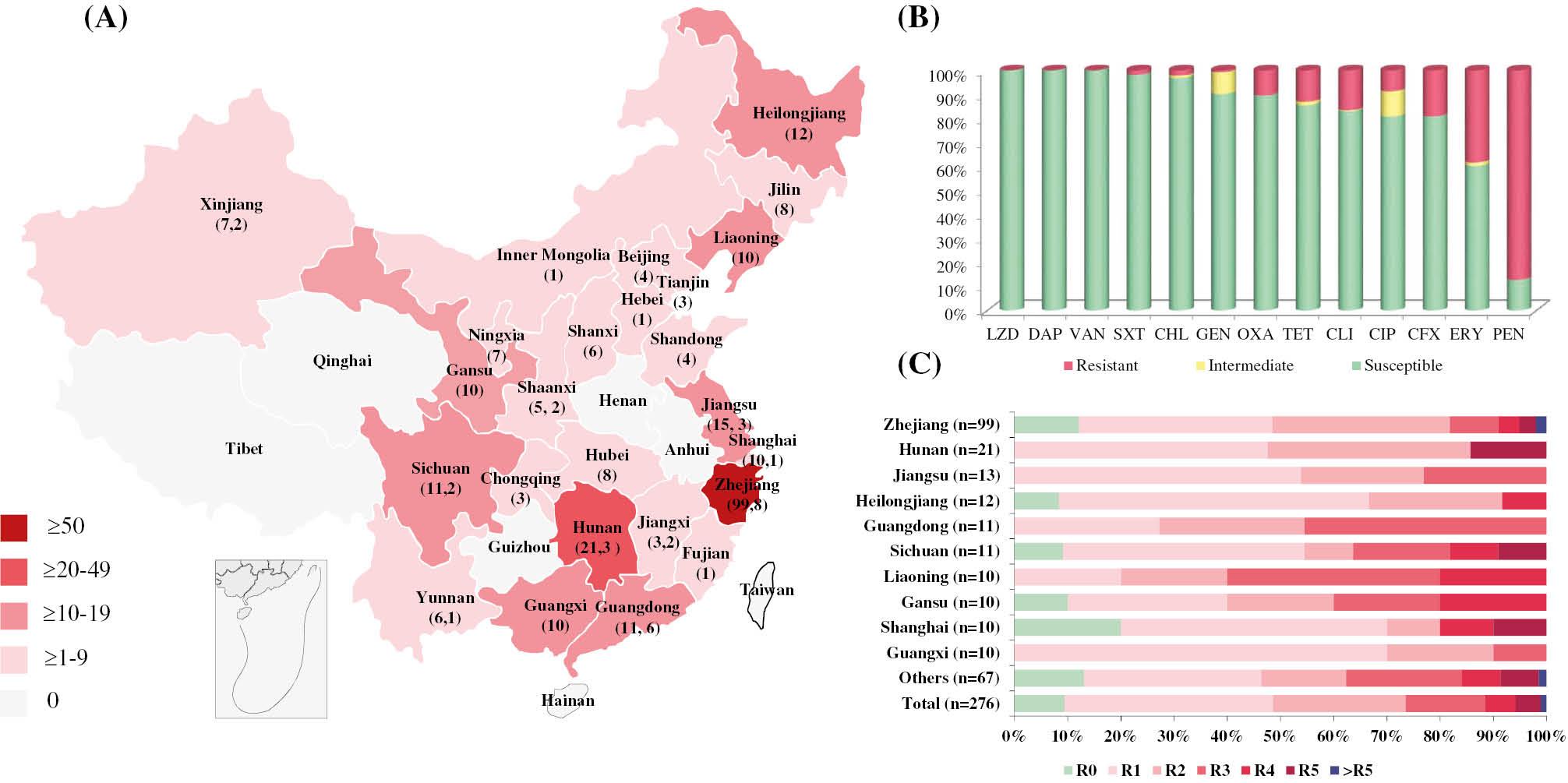
Map of China showing the location of the 25 provinces where the 276 RTE food-associated isolates were collected in 2018. (A) The provinces in gray were not included in this study, while the other 25 provinces where S. aureus was collected from RTE foods are marked with a rose-red gradient. The numbers enclosed in parentheses are the number of S. aureus isolates (before the comma) and MRSA (after the comma). (B) The AST results of S. aureus against 13 antimicrobial agents. (C) Frequency distribution of S. aureus isolated from different provinces in China completely susceptible or resistant to 1 to > 5 antimicrobial classes. N, total number of S. aureus isolates tested for AST in different provinces; R0->R5, resistance to 0 up to 5 antimicrobial classes.
Antimicrobial susceptibility of S. aureus and MRSA isolates
AST was performed for all isolates against 13 antimicrobial agents. Two hundred fifty isolates (90.6% [250/276]) were resistant to at least one antimicrobial, whereas only 26 isolates (9.4% [26/276]) were susceptible or had intermediate susceptibility to all 13 antimicrobials (Fig 1B). Specifically, the greatest resistance was observed for PCN (87.3% [241/276]), followed by ERY (38.4% [106/276]). The resistance rate to CFX, CLI, TET, and OXA ranged from 10.5%-19.2%, while resistance to CIP, CHL, SXT, and GEN was < 10%. Additionally, all isolates were susceptible to drugs of last resort (DAP, LIN, and VAN; Fig 1B).
Among the 276 S. aureus isolates, 73 (26.1% [73/276]) were MDR (resistant to ≥ 3 classes of antimicrobials; Fig 1C). In addition, 16 (5.8% [16/276]) isolates were resistant to 4 classes of antimicrobials, 13 (4.7% [13/276]) were resistant to 5 classes of antimicrobials, and 3 (1.1% [3/276]) were resistant to ≥ 5 classes of antimicrobials. A total of 34 resistance profiles were identified. The three most prevalent resistant profiles in the cohort were PCN (36.6% [101/276]), PCN-ERY (11.6% [32/276]), and PCN-ERY-CLI (5.1% [14/276]). Moreover, isolates resistant to eight classes of antimicrobials had the following resistant profile: PCN-ERY-CLI-CIP-STX-TET-CHL-GEN.
All the MRSA isolates were resistant to PCN, and > 73.3% of the isolates (22/30) were MDR (Table 1). Twelve resistance profiles were identified in the MRSA isolates. The most prevalent resistant profiles were PCN-OXA-CFX (23.3% [7/30]), PCN-OXA-ERY-CLI-TET-CFX (20.0% [6/30]), PCN-OXA-ERY-CFX (16.7% [5/30]), and PCN-ERY-CFX (16.7% [5/30]).
Antimicrobial susceptibility of MRSA from ready-to-eat foods in China.
| Antimicrobial agent | Susceptible | Intermediate | Resistant | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Daptomycin | 30 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Linezolid | 30 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Trimethoprim-sulfamethoxazole | 30 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Vancomycin | 30 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Ciprofloxacin | 29 | 96.7% | 1 | 3.3% | 0 | 0.0% |
| Gentamicin | 28 | 93.3% | 2 | 6.7% | 0 | 0.0% |
| Chloramphenicol | 27 | 90.0% | 0 | 0.0% | 3 | 10.0% |
| Tetracycline | 20 | 66.7% | 2 | 6.7% | 8 | 26.7% |
| Clindamycin | 19 | 63.3% | 0 | 0.0% | 11 | 36.7% |
| Erythromycin | 8 | 26.7% | 0 | 0.0% | 22 | 73.3% |
| Oxacillin | 7 | 23.3% | 0 | 0.0% | 23 | 76.7% |
| Cefoxitin | 0 | 0.0% | 0 | 0.0% | 30 | 100.0% |
| Penicillin | 0 | 0.0% | 0 | 0.0% | 30 | 100.0% |
Detection of virulence factors among MRSA isolates
We searched for the presence of 15 virulence genes in the 30 MRSA isolates; 9 virulence genes were detected in 18 isolates (60% [18/30]), including sea (3.3% [1/30]), seb (36.7% [11/30]), sec (23.3% [7/30]), sed (3.3% [1/30]), seh (16.7% [5/30]), selk (50% [15/30]), sell (23.3% [7/30]), selq (50% [15/30]), and tsst-1 (16.7% [5/30]; Fig 2). Most of the MRSA isolates (53.3% [16/30]) harbored multiple virulence genes (≥ 3 virulence genes). With respect to classic SE genes, the seb gene was the most common. For the newly-identified SE and SEl genes, the selk and selq genes had 50%-positive isolates. Four virulence gene patterns were identified among 18 isolates; seb-selk-selq (33.3% [10/30]) was the most prevalent profile, followed by sec-she-selk-sell-selq-tsst-1 (16.7% [5/30]), sec-sell (6.7% [2/30]), and sea-seb-sed (3.3% [1/30]).
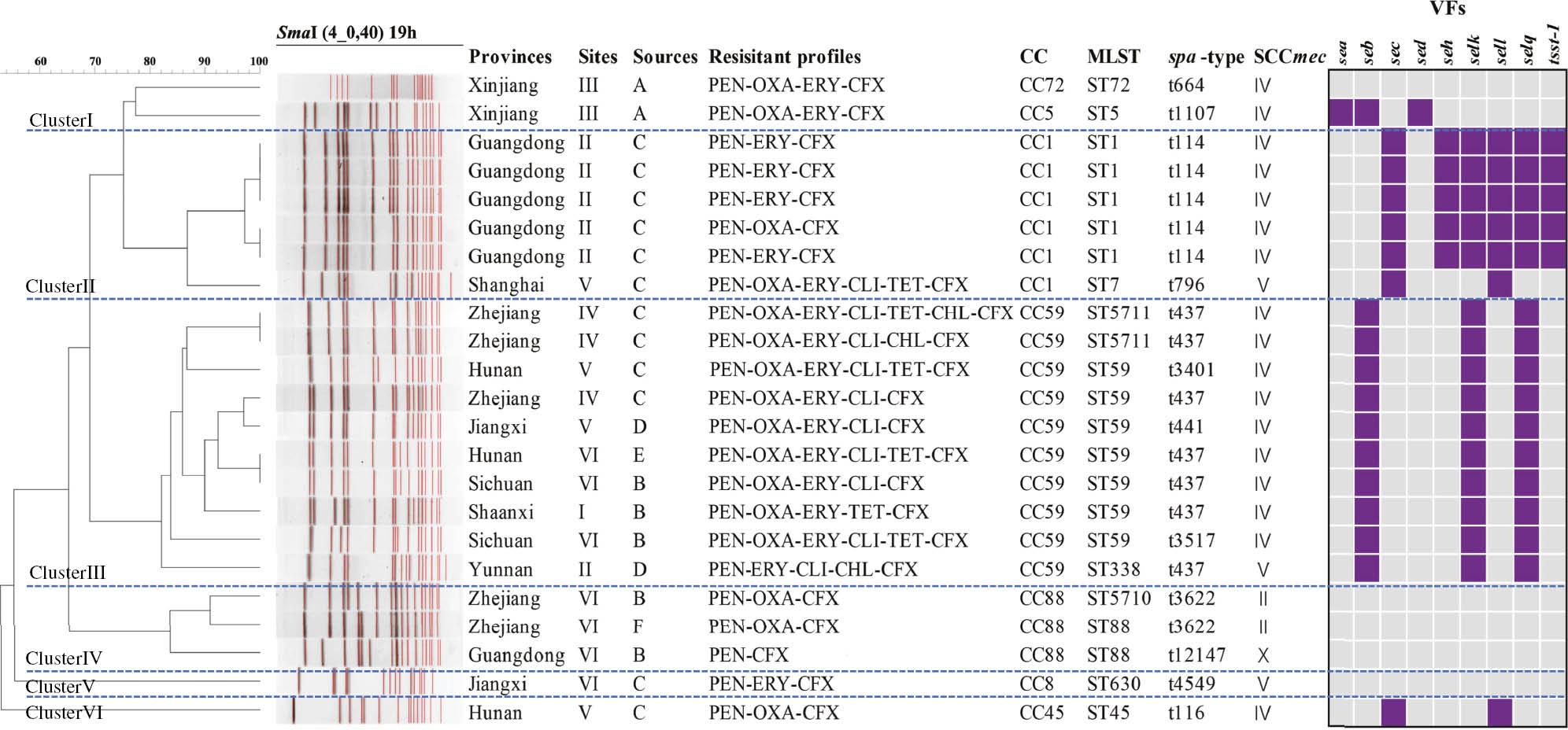
Resistant profiles, virulence factors, and genetic relationships of 23 MRSA isolates established by SmaI PFGE analysis. CCs = clone complexes; Sample sites (Sites): I = convenience store, II = farm product market, III = fast food restaurant, IV = retail outlet, V = supermarket, VI = take-out restaurant; food sources (Sources): A = cereal product, B = Chinese salad, C = Chinese dim sum, D = ice cream, E = sauced meats, F = sushi; virulence factors (VFs): purple = present, grey = absent.
MLST
The presence of different MRSA lineages was investigated by combining MLST, spa typing, and SCCmec typing. A total of 13 STs were identified based on MLST analysis (Figs 2 and 3). ST59 and ST398 were the most prevalent STs (both 23.3% [7/30]), followed by ST1 (16.7% [5/30]). All 13 STs belonged to 8 CCs, including CC1, CC5, CC8, CC72, CC88, CC59, CC45, and CC398. The most common CC was CC59 (33.3% [10/30]), followed by CC398 (23.3% [7/30]), CC1 (20% [6/30]), and CC88 (10% [3/30]).
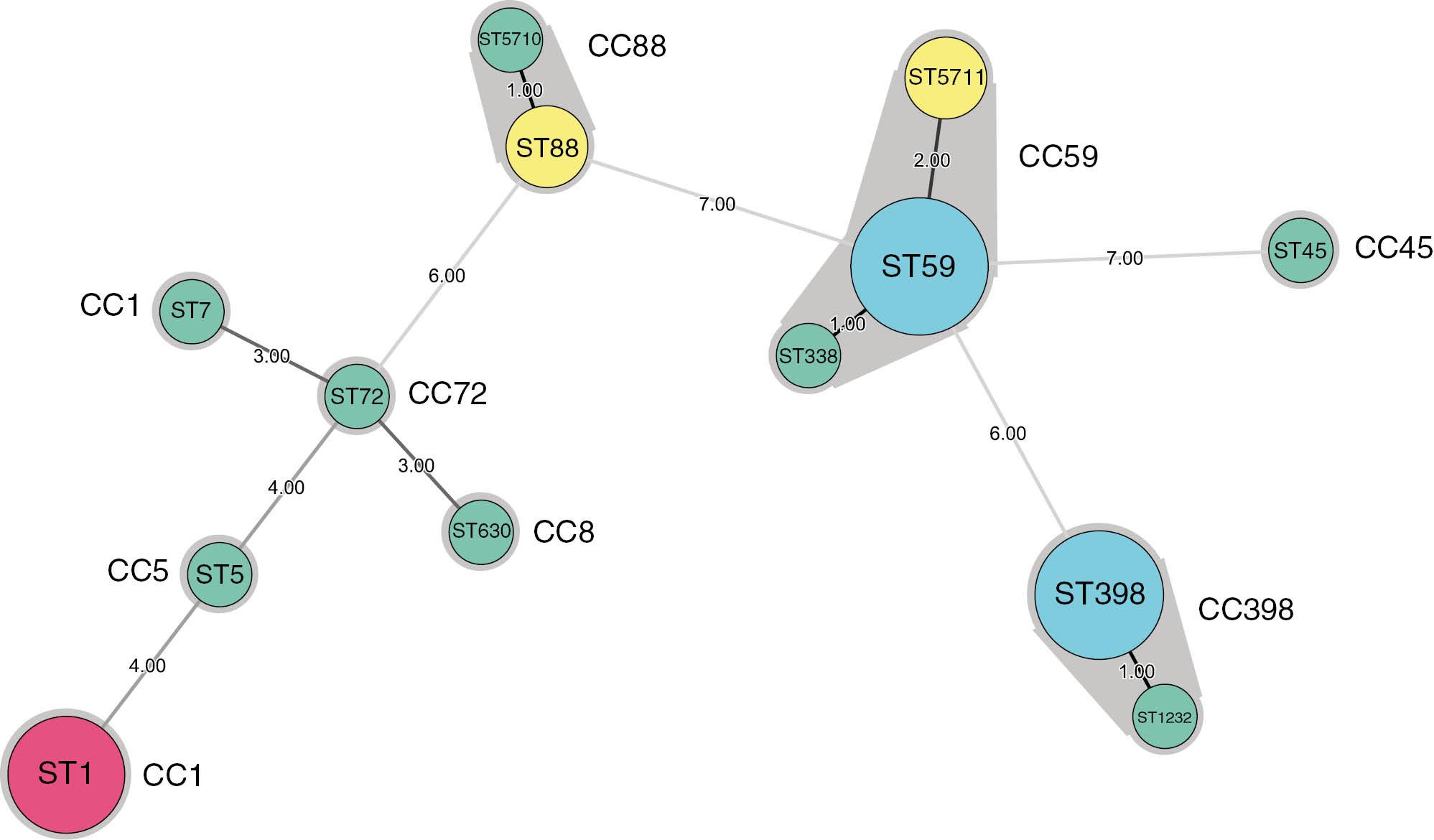
Minimum spanning tree of the 30 MRSA isolates by MLST. The size and color of the circle indicate the number of isolates for each ST type. The grey halos surrounding the ST types denote that the ST types belonged to the same complex. The numbers on the straight line indicate the distance of the relationship between neighboring ST types.
spa typing
Thirteen spa types were identified among the 30 MRSA isolates (Fig 2); t437 and t011 were the most prevalent 23.3% (7/30), followed by t114 (16.7% [5/30]) and t3622 (6.7% [2/30]). The following spa types were identified in one isolate each: t116; t441; t664; t796; t1107; t3401; t3517; t4549; and t12147. The three most prevalent spa types (t437, t011, and t114) were associated with CC59, CC398, and CC1, respectively.
SCCmec typing
Four SCCmec types were identified among the 30 MRSA isolates, including type II, IV, V, and X SCCmec elements (Fig 2); SCCmecIV was the most prevalent 60.0% (18/30), followed by SCCmecV (30.0% [9/30]), SCCmecII (6.7% [2/30]), and SCCmecX (3.3% [1/30]). Combined analysis of CCs, and spa and SCCmec types revealed the presence of 17 different MRSA lineages, of which the most common lineages were CC59-t437-SCCmecIV/V (23.3% [7/30]), CC398-t011-SCCmecV (20.0% [6/30]), and CC1-t114-SCCmecIV (16.7% [5/30]).
PFGE
PFGE was performed on the 30 MRSA isolates, among which 7 isolates were not digested by SmaI and were identified as ST398 (not shown in Fig 2). The remaining 23 MRSA isolates were characterized by 18 PFGE patterns, which were further grouped into 6 clusters based on > 76% genetic similarity (Fig 2). The dominant PFGE cluster was cluster III, including 8 PFGE patterns from 10 isolates all associated with CC59; the dominant MRSA lineage was CC59-t437-SCCmecIV/V. Interestingly, the majority of MDR isolates were within this cluster and all isolates had the same virulence gene profile (seb-selk-selq). Cluster II included six isolates belonging to CC1, among which five were CC1-t114-SCCmecIV; all of the isolates collected were from the same province (Guangdong) and all had the same virulence gene profile (sec-she-selk-sell-selq-tsst-1). Three CC88 MRSA were clustered in PFGE cluster IV. No aforementioned enterotoxin genes were identified in the seven ST398 isolates.
DISCUSSION
In recent years the consumption of RTE foods in China has markedly increased due to its convenience. The global RTE food market sales were > 194 billion dollars in 2016 and is expected to increase by > 13% between 2018 and 2023 (unpublished data). RTE food sales are growing in China; 500 million people consumed RTE food as meals in 2020. Retail RTE foods are intended for direct human consumption without the need for cooking or other processing; thus, foodborne pathogens are not eliminated or reduced. Once contaminated by foodborne pathogens, the possibility of food poisoning is increased [32,33]. Specifically, S. aureus and MRSA have been frequently reported to cause foodborne outbreaks (FBOs) and are detected in RTE foods [10,11,16–18,34–36]. Therefore, the presence of S. aureus and MRSA strains in RTE food represents a potential threat to public health. The presence of S. aureus and MRSA strains, the enterotoxin gene repertoires, and molecular characteristics in RTE foods across China were determined in the current study. Our findings will be helpful in assessing the genetic diversity of MRSA and providing the genetic basis for evolutionary and epidemiologic studies of MRSA among various RTE foods.
We showed that 90.6% of S. aureus isolates were resistant to at least one antimicrobial agent, which was slightly lower than reported for RTE foods in the Sichuan (96.3%) [33] or Shaanxi provinces of China (98.4%) [37]. Because our samples were from 25 provinces across China, our research is more representative of the entire country than the studies that focused on individual provinces. Similarly, we demonstrated a lower MDR prevalence (26.1%) than previous reports from Shaanxi, China (58.6%) [37] and Himachal Pradesh, India (81.3%) [3]. The greatest resistance was to PCN (87.3%), which is in agreement with the findings iin Sichuan province (90.7%) [18] and our previous study (83.7%) [33].
Our results showed that 10.9% (30/276) of S. aureus isolates were MRSA; the prevalence was similar to previously reported in China (10.1%) and Brazil (8.8%) [32,38], but lower than previously reported in Bangladesh (23.1%) and higher than reported in Japan (2.9%) [39,40]. The relatively low prevalence of MRSA in Japan might be due to that the fact that the samples were mainly marine wild raw fish.
S. aureus toxigenic strains can produce a wide variety of virulence factors; the SEs are regarded as the main cause of SFP. In the current study 9 virulence genes, including sea, seb, sec, sed, seh, selk, sell, selq, and tsst-1, were found in 60% (18/30) of the MRSA isolates and 53.3% (16/30) of MRSA isolates contained > 3 virulence genes. Notably, the SEB coding gene, seb (36.7% [11/30]), was the most prevalent gene among the classic SE genes, followed by sec, sea, and sed. This result partially contrasts with previous reports in which sea was the the most common cause of SFP worldwide, causing one of the largest SFP outbreaks involving 14,780 individuals in Japan in 2000 [10]. Based on high prevalence of seb among the MRSA isolates in the current study, it is important to understand the role of this toxin in causing more severe poisoning [41]. Consistent with previous reports, we showed that the seb gene was always detected together with selk and selq genes [42]. The coexistence of multiple virulence genes may reflect a potential link of genetic locations.
ST59, ST398, and ST1 were the three most prevalent ST types among the MRSA isolates in the current study. This finding is agreement with previous studies in which ST59 was the most common ST among community-associated MRSA (CA-MRSA) in China and the predominant clone accounting for 47.7% (51/107) of MRSA isolates in retail foods in China [43,44]. ST59 MRSA were also recovered from humans and pigs in a backyard farm in rural China, indicating a possible bidirectional mode of transmission [45]. ST59 is the most common cause of hospital-acquired CA-MRSA infections, especially in pediatric patients [46,47]. In general, ST59 and ST338 (a single locus variant of ST59) have been shown to be the most common STs associated with community-acquired pneumonia in children in China [48]. Moreover, ST59, ST5711, and ST338 belong to CC59 (33.3% [10/30]), which is the prevalent clone associated with healthy carriers and patients with community-acquired infections in Asia [43]. ST59 was also shown to be the most prevalent clone in food, suggesting the possible high risk of bidirectional transmission between humans and food. In the current study the CC59 isolates were collected from seven provinces, which confirmed the universality of the distribution.
The second most ST is ST398 (23.3% [7/30]), which was the first detected and most widespread livestock-associated MRSA (LA-MRSA) clone in Europe and North America, and has identified colonizing healthy humans or causing clinical infections [49–52]. The ST1 isolates (16.7% [5/30]) are routinely described as CA-MRSA, but have also been collected from animals and has been associated with SFP in Korea and China [53,54]. ST5711 and ST5710 have not been reported, while other STs identified in this study have been reported to cause clinical infections.
The most frequently observed spa types were t436 and t011. This finding is consistent with previous reports that t437 is the most common type in retail foods in China and the most dominant community-associated clone from Asia across Europe [55–57]. Moreover, t011 has been frequently isolated from livestock, especially from pigs [58].
SCCmecIV and SCCmecV were the top two prevalent types in our cohort, accounting for 90% of the total. Only two isolates typed as SCCmecII belonged to CC88, which is usually reported as hospital-associated MRSA (HA-MRSA). These results are in agreement with previous findings that reported CA-MRSA commonly carries SCCmec types IV and V, while HA-MRSA generally carries SCCmec types I, II, and III [59–61].
The seven MRSA isolates of ST398 all belonged to the same lineage (ST398-t011-V), which is a typical LA-MRSA genotype, frequently found as the predominant type of MRSA isolates from livestock, particularly from pigs and the entire pork production chain [58,62]. Interestingly, we found four ST398-t011-V isolates in non-animal foods (mostly cereal products), suggesting potential cross-contamination of these isolates through food processing. The remaining 23 MRSA isolates were distinguished into 18 PFGE patterns and grouped into 6 clusters (I-VI). The clusters were closely relevant to MLST, spa types, SCCmec types, and virulence gene patterns. All five CC1-t114-SCCmecIV MRSA in PFGE cluster II were identified in Chinese dim sum in Guangdong province. According to a previous report, an MRSA CC1-t114-SCCmecIV clone was isolated from SFP outbreaks in China [63]; however, there are few or no reports about this genotype in other countries up to the present. The dominant MRSA lineage was CC59-t437-SCCmecIV/V in cluster III, which was reported as the major clone among CA-MRSA in hospitals and the predominant genotype in bacteremia in China, and also identified in food poisoning outbreaks [46,47]. In addition, the CC59-t437-SCCmecIV/V clone was found in various food types, suggesting a possible diverse dissemination routes of these isolates. We found that the resistance and virulence genes in most cases exhibited a clonal distribution, and isolate CC59 tended to be resistant to more antimicrobial agents. Moreover, the virulence gene profiles among MRSA isolates had a significant correlation with the genotype lineages, which is different from what was reported in a recent study conducted in Italy [64]. The CC1-t114-SCCmecIV clone was shown to possess the most virulence genes (sec-she-selk-sell-selq-tsst-1), followed by CC59-SCCmecIV/V (seb-selk-selq), whereas no virulence genes were detected in CC398 and CC88 MRSA. Our understanding of the antimicrobial susceptibility and virulence genes among specific MRSA clones is vital for the development of therapeutics to target infections and measures to eliminate these clones in RTE-foods.
Finally, there were some limitations in our study. Specifically, we could not detect all virulence genes and distinguish strains among the same CCs or STs using PCR. This limitation might benefit from a whole genome sequencing (WGS) approach that could potentially provide additional insights about our data. Specifically, one recent study involving 673 food- and human-associated S. aureus in China based on WGS exhibited a very powerful capacity [65]. In addition to 15 virulence genes detected in the current study, 80 other virulence genes were identified by WGS. Furthermore, the phylogenetic tree could be reconstructed based on 1585 core genes, thus promising much better resolution than the current study.
CONCLUSIONS
This study provided a detailed investigation of MDR S. aureus and characterization of MRSA isolates recovered from RTE foods in different geographic regions of China. The prevalence of MDR and MRSA isolates was high, with greater than one-half of MRSA harboring multiple virulence genes. Therefore, the clinic pathogenicity and transmission risk cannot be ignored. The genotype diversity of the MRSA isolates was related to antimicrobial resistance and virulence genes. The MRSA isolates with similar genotype generally had similar antimicrobial resistance patterns and virulence gene profiles. These results suggest that monitoring the genotypes of MRSA in RTE foods would be instrumental in tracing the source of contamination and assessment of antimicrobial resistance and risk of SFP. Therefore, such work will be helpful in assisting the government, food industries, and other stakeholders to improve food safety measures and control the transmission route of this bacterium.

