INTRODUCTION
In recent decades, Central and South America have experienced a series of emergence events, including but not limited to the following: (a) spillover of endemic arthropod-borne viruses (arboviruses) from wildlife reservoirs into humans; (b) exchange of emerging arboviruses; (c) re-emergence of arboviruses that were previously controlled by vaccination and/or vector control; (d) introduction and spread of novel arboviruses, and (e) spread of viruses to other regions. Furthermore, there is great concern that Zika virus (ZIKV), an arbovirus introduced in the western hemisphere as early as 2013, may establish an autochthonous sylvatic transmission cycle. More broadly, the enormous vertebrate and invertebrate biodiversity of the region harbors a broad range of arboviruses, in which the risk of emergence and spread among humans is unknown. A world experiencing economic globalization, rapid population mobility facilitated by jet travel, uncontrolled urban expansion and slummification, deforestation, land use repurposing, breakdown of public health structures in countries experiencing societal, economic, and political unrest, and global climate change all exacerbate the risk of arbovirus emergence in the neotropics.
The Coordinating Research on Emerging Arboviral Threats Encompassing the Neotropics (CREATE-NEO) provides a network of surveillance sites across Central and South America that are geographically broad, encompass hotspots of biodiversity and different degrees of urbanization, and are well-integrated with appropriate modeling techniques that anticipate, mitigate, and counter the threat posed by emerging and resurging arboviruses. Therefore, collected surveillance and epidemiologic data and generated predictive models will be leveraged to forewarn public health agencies at the local, regional, and global levels of endemic arboviruses that could pose a particularly high risk of spillover, emergence, and/or international spread. The CREATE-NEO network will also be ready to detect the introduction of novel arboviruses from other continents and swiftly respond. The overarching goal of CREATE-NEO is to build local capacity to predict, detect, and respond to emerging arboviruses at the point of origin, thereby maximizing the potential to avert full-blown emergence and large-scale epidemics across the region and beyond.
CREATE-NEO OVERVIEW
Central and South America have borne the brunt of emerging arboviruses [1]. Emergence in this context has taken multiple forms, including the spillover of enzootic arboviruses from wildlife reservoirs into humans (e.g., Oropouche {OROV} [2] and Rocio {ROCV} [3], among others), exchange of emerging arboviruses within the region (e.g., Mayaro {MAYV} [4,5], Venezuelan equine encephalitis {VEEV} [6]), re-emergence of previously controlled arboviruses (e.g., yellow fever {YFV} and dengue {DENV} [1,7–11]), and introduction and spread of novel arboviruses (e.g., West Nile virus {WNV} [12,13], chikungunya {CHIKV} [14,15], and Zika {ZIKV} [16,17]). Furthermore, Central and South America have been exporting arboviruses to other regions (e.g., Madariaga {MADV} [18]) at increasing rates as global mobility has accelerated.
Of all the recent arbovirus introductions to Central and South America, the introduction of ZIKV as early as 2013 and the consequent surge in congenital Zika syndrome constituted a true calamity for the region, particularly Brazil [19]. There is considerable concern that ZIKV may spill back into an enzootic transmission cycle among non-human primates (NHPs) in the Americas, as YFV did centuries ago [20]. Establishment of a sylvatic ZIKV cycle would complicate control of virus transmission and render ZIKV eradication from the Americas unattainable. Although it received somewhat less attention, CHIKV was also introduced to the Americas in 2013 and exploded into a massive outbreak that created a staggering burden of clinical disease [21], sometimes resulting in prolonged disability [22]. Within the last decade, WNV was also introduced to Brazil, which now faces an imminent WNV outbreak [12,13]. All three viruses are now considered endemic in the neotropics.
Over the same time period, arboviruses that had previously been controlled via vaccination (YFV) or vector control (DENV) resurged dramatically [1,7–11]. Between 2016 and 2018, Brazil experienced an outbreak of yellow fever that was shocking in magnitude (the largest since the eradication of urban yellow fever in the region) and location (on the east coast of the country), which was perilously close to the mega-cities of Sao Paolo and Rio de Janeiro [7,23,24]. The thousands of human cases of yellow fever in this outbreak are thought to have been derived entirely from sylvatic transmission from non-human primate reservoirs. Moreover, 2019 had the highest number of DENV cases in the Americas in recorded history [25]. With the exception of a well-established vaccine for YFV and a controversial vaccine for DENV [26], there are currently no licensed vaccines to protect against infection with any of these viruses, and there are no licensed antivirals to alleviate disease.
Central and South America have also recently experienced an uptick in the spillover of endemic arboviruses from wildlife reservoirs into humans. This geographic region encompasses much of the world’s diversity of vertebrate hosts and arthropod vectors [27,28], and as a result, many of the world’s zoonotic viruses (Fig 1). Within this context, MAYV poses a specific threat [29,30]. MAYV is maintained in a sylvatic cycle involving non-human primates and Haemagogus mosquitoes, but readily spills over into humans, with a clinical presentation similar to and easily mistaken for chikungunya [31]. Importantly, the range of hosts and vectors responsible for maintenance of MAYV has not been completely characterized [32]. Phylogenetic analyses have revealed an exchange of MAYV viruses between Amazonia and the Caribbean, attributable to some combination of human movement between the regions and bird migration [5,33]. Similarly, MADV, which belongs to the eastern equine encephalitis virus complex, was initially exported from Brazil to other South and Central American countries [34–37], raising fears that MADV might move further into the Caribbean or North America. Outbreaks of Oropouche fever, attributable to spillover of midge transmitted OROV, have recently occurred in Brazil and Peru [2]. This virus is known to be maintained in a sylvatic reservoir, but the key host species in this cycle are not completely known and the risk of full-blown OROV emergence into human circulation is not fully understood.
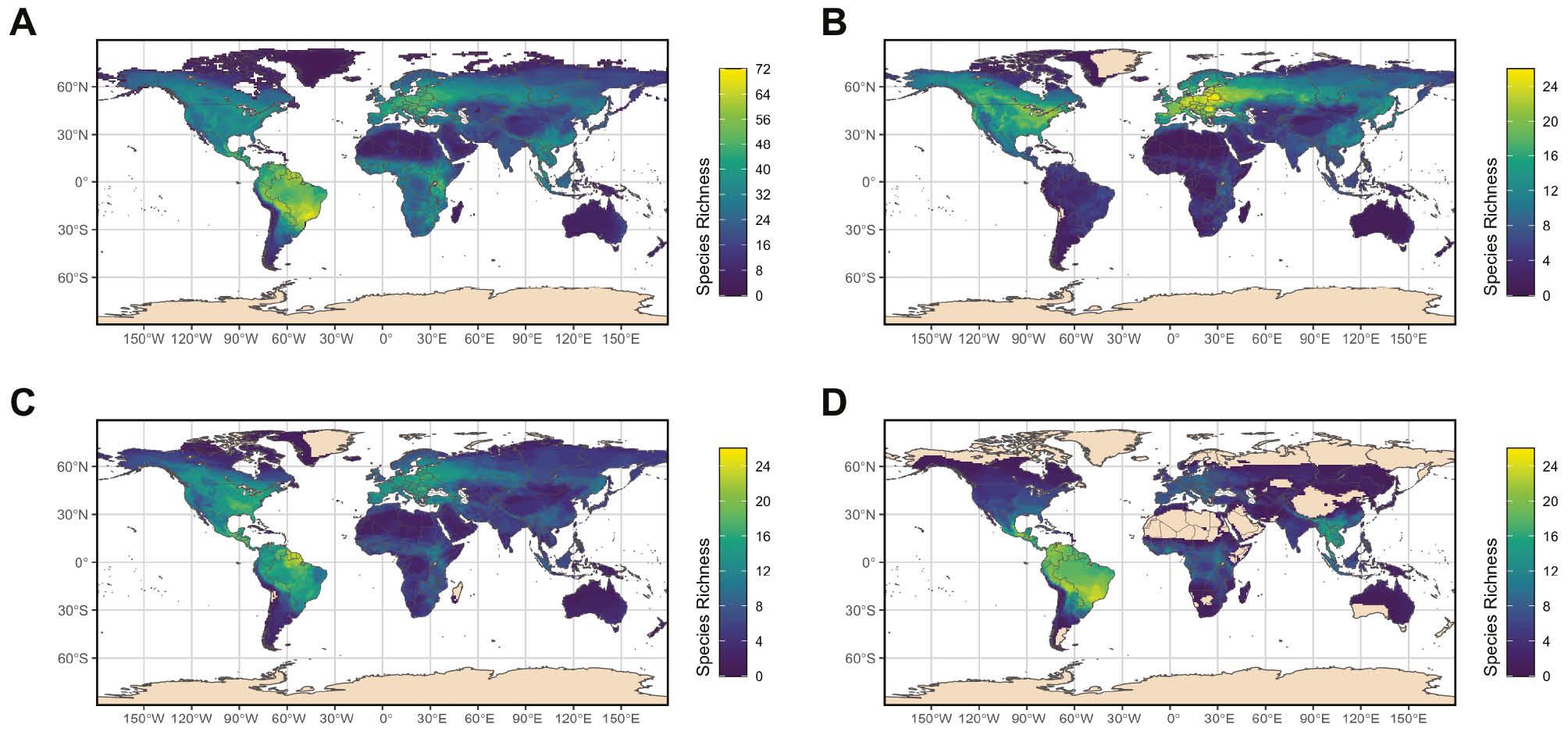
Richness of zoonotic viruses A) across all mammal species currently associated with zoonoses, B) all mammal species associated with tick-borne diseases, C) all mammal species associated with mosquito-borne diseases, and D) all bats associated with zoonoses.
While the viruses discussed above have already demonstrated a capacity for spillover, many additional viruses have been detected in wildlife or arthropod vectors in Central and South America; however, the risk of spillover and spread is presently unknown [38–40].
Unfortunately, the arbovirus spillover and spread rates in Central and South America are likely to accelerate in the future. Deforestation, already extensive in Central and South America, is expanding, primarily for commercial agriculture [41]. Forest conversion enhances contact between humans, and sylvatic vectors and viruses at land cover edges [42], thus favoring competent arbovirus vectors [43] and precipitating arbovirus spillover [44]. Global climate change is also expected to shift vector and arbovirus distributions [45–47], and to facilitate spillover of novel arboviruses. Ongoing urbanization is clearly a boon to key urban vectors, such as Aedes aegypti [48] and Ae. albopictus [46,49], and may also promote spillover of novel arboviruses [50]. Globalization, especially transcontinental travel and relocation of humans, affords both vectors and arboviruses the opportunity to move rapidly into Central and South America from elsewhere, or conversely, from points of origin in Central and South America across the region or across the world [51–53]. For example, as of 22 September 2022, hundreds of thousands of Venezuelans have emigrated to Panama and Brazil [54]. The political, societal, and economic upheaval in Venezuela in recent decades has led to the breakdown of public health structures, which occupy a critical juncture in Central and South America, and exacerbated the resurgence and regional spread of pathogens [55–57], measles [58], malaria [59], as well as the spillover and cryptic circulation of novel pathogens [60].
There is an urgent global need for broad and integrated surveillance networks to better confront the rising threat of emerging and resurging arboviruses [61]. This need was recognized by the National Institutes of Health (NIH), with the issuance of a Funding Opportunity Announcement (FOA [RFA-AI-19-028]) on 4 April 2019, calling for the establishment of a coordinated Centers for Research on Emerging Infectious Diseases (CREID) network in regions around the globe where emerging and re-emerging infectious disease outbreaks are likely to occur. The Coordinating Research on Emerging Arboviral Threats Encompassing the Neotropics (CREATE-NEO), 1 of the 10 funded CREIDs, integrates arbovirus surveillance across the region with summarizing and predictive modeling efforts to better anticipate and counter arbovirus emergence (Fig 2). Since its inception, CREATE-NEO has built local capacity, including comprehensive genomic surveillance, allowing teams to detect, predict, and respond to emerging arboviruses at the point of origin. CREATE-NEO teams have been able to quickly redirect their resources to respond to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), thus allowing CREATE-NEO teams to inform local and regional policy makers and public health authorities [62–64].
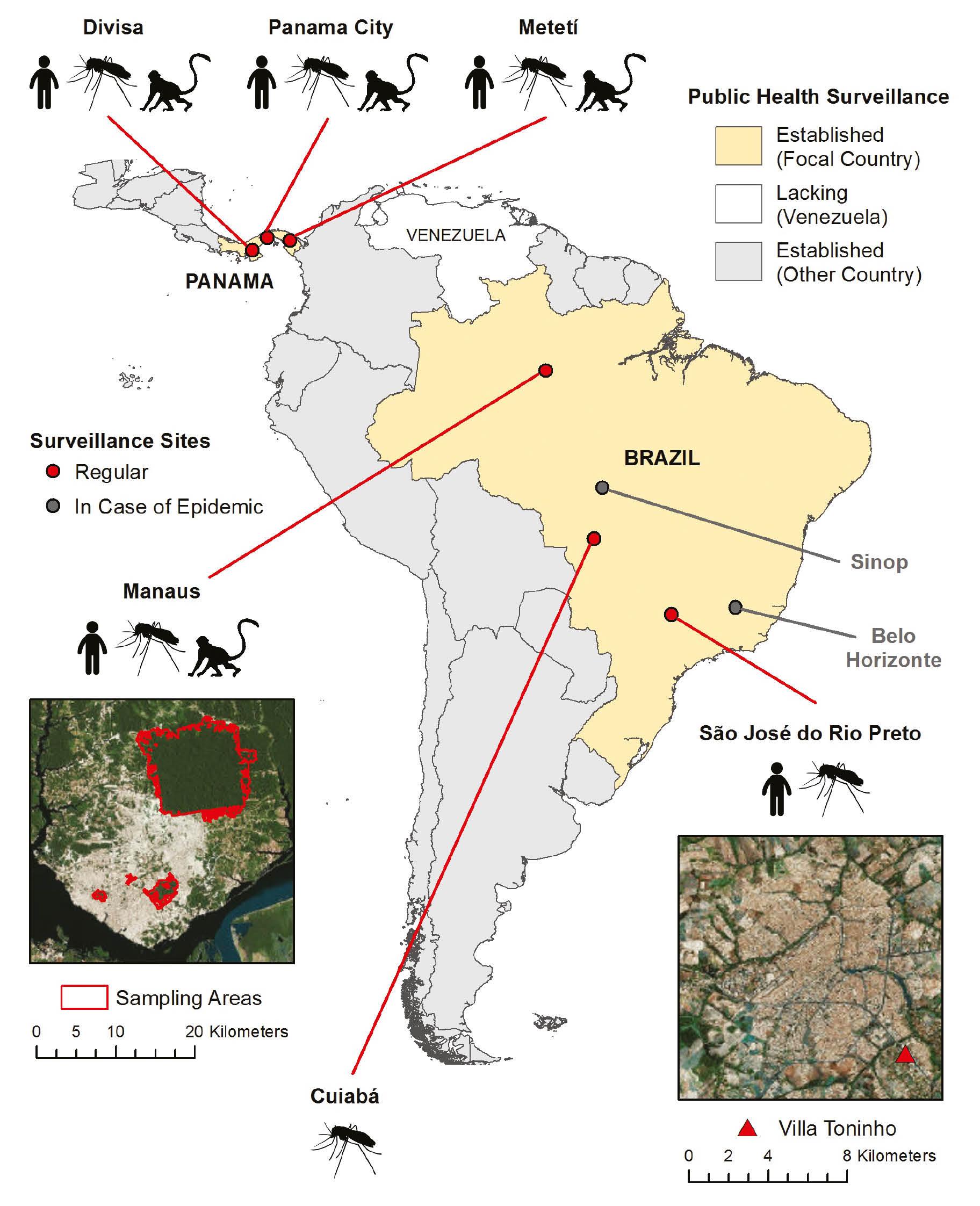
Overview of CREATE-NEO surveillance of humans (human icon), vectors (mosquito icon), and wildlife (monkey icon) in Central and South America.
The major areas of concentration for CREATE-NEO involve the following:
Virus discovery in wildlife and arthropod vectors in Central and South America. Members of CREATE-NEO have discovered, characterized, and annotated many known and new viruses in wildlife and vectors in Central and South America, select examples of which are listed below in Table 1. Additionally, our group was the first to report evidence of a natural ZIKV infection in neotropical, non-human primates in Brazil [77], and a lack of evidence for SARS-CoV-2 spillover in wild, non-human primates across several surveyed locations in Brazil [64].
Development of novel diagnostic assays. Detecting and distinguishing arbovirus infections in human populations among asymptomatic and symptomatic individuals are important CREATE-NEO activities. Without sensitive and specific diagnostics, fever symptoms can go undiagnosed or be misdiagnosed. We have developed affordable, easy-to-use, rapid, paper fluidic tests to detect and distinguish DENV and ZIKV [79] without crossover, thus permitting unambiguous virus identification. Our partners are currently developing nanoparticle-based methods for virus detection and pandemic preparedness [80], initially targeting neglected arboviruses (e.g., Ilheus [ILHV] and ROCV viruses) with enormous potential for emergence and posing a significant impact on public health.
Identification of key vectors and hosts in a regional hotspot for arbovirus spillover and spillback. CREATE-NEO is coupling geographically- and taxonomically-broad surveillance of arthropods and vertebrates with remote sensing and machine learning to predict the distribution of both known and as-yet undiscovered host-vector networks that sustain transmission of key arboviruses. The close integration of empirical sampling with these modeling approaches is a highly innovative aspect of the proposed work [81–85].
Novel insights into arbovirus transmission and cross-protection from clinical studies. CREATE-NEO leverages ongoing human cohorts in geographically-, demographically-, and ecologically-distinct regions in Central and South America that are hyper-endemic for various constellations of arboviruses to gain insight into how pre-existing immunity influences clinical outcomes upon infection. Understanding this interplay between different arboviruses mediated by cross-protection or enhancement is a highly innovative aspect of the proposed work that will influence vaccine design, enhance vaccine efficacy, and predict the risk of future arbovirus epidemics.
Examples of viruses discovered by CREATE-NEO partners in Central and South America.
| Country | Host | Virus name | Virus family | Ref. |
|---|---|---|---|---|
| Colombia | Mosquitoes | Sinu virus | Orthomyxoviridae | [65] |
| Brazil; Trinidad & Tobago | Mosquitoes | Trinity virus | Peribunyaviridae | [66] |
| Brazil; Panama; Ecuador | Mosquitoes, birds | Gamboa serogroup | Peribunyaviridae | [67] |
| Panama | Mosquitoes | PanAr 395145 and PanAr 395144 | Peribunyaviridae | [68] |
| Colombia | Mosquitoes | Chiqui virus | Reoviridae | [69] |
| Panama | mosquitos | Gamboa mosquito virus | Flaviviridae | [70] |
| Panama | Mosquitoes | Mercadeo virus | Flaviviridae | [71] |
| Peru | Mosquitoes | La Tina virus | Flaviviridae | [72] |
| Peru | Mosquitoes | Arboretum, Puerto Almendras | Rhabdoviridae | [73] |
| Colombia | Mosquitoes | Balsa virus | Rhabdoviridae | [39] |
| Panama | Mosquitoes | Rio Chico | Rhabdoviridae | [39] |
| Brazil | Amphibians | Cuiaba virus | Rhabdoviridae | [74] |
| Peru; Brazil | Mosquitoes | Piura, Loreto, and Santana viruses | Negeviridae | [75] |
| Colombia, Panama, Peru, Brazil | Mosquitoes | Wallerfield, Brejeira, and San Bernardo viruses | Negeviridae | [76] |
| Brazil | NHPs | Zika virus | Flaviviridae | [77] |
| Mexico | Mosquitoes | Zika Virus | Flaviviridae | [17] |
| Panama | Equids | Venezuelan equine encephalitis and Eastern equine encephalitis | Togaviridae | [78] |
CONCLUSION
CREATE-NEO is responding to the rising threat of emerging arboviruses in the Americas by linking a group of Brazilian, Panamanian, and US scientists into a network that integrates arbovirus surveillance in vectors, wildlife, and humans across Central and South America. The data generated are analyzed with cutting-edge phylogenetic, statistical, and modeling approaches to better anticipate and counter arbovirus emergence. Close relationships with local, regional, and global public health agencies allow the timely transfer of information on emerging arboviruses to agencies tasked with responding to such events; however, our ultimate goal is to head off emergence events at the source by building local capacity within Panama and Brazil that will expand the already impressive abilities at these sites to collect and assay key samples in situ.



