INTRODUCTION
Taenia solium taeniasis/cysticercosis is an important food-borne infection transmitted between humans and pigs worldwide [1, 2]. Taeniasis generally causes human intestinal diseases, whereas cysticercosis is a serious disease infecting both humans and pigs [3]. Porcine cysticercosis primarily infects pig muscle and brain tissues, thus decreasing pork quality [4]. Human cysticercosis has been reported in a variety of forms, including neurocysticercosis (NCC), ocular cysticercosis (OCC), subcutaneous muscle cysticercosis, oral cavity cysticercosis, and visceral cysticercosis [5–7]. Human NCC primarily causes seizures, high intracranial pressure, and psychiatric disorders, and it continues to be a major disease burden on people in many parts of the world [2, 8–10].
T. solium taeniasis/cysticercosis is a severe cestode infection that is common in humans, and domestic pigs (Sus scrofa domestica) and wild boars (Sus scrofa) co-existing areas, particularly those with warm and mild climates in Latin America, sub-Saharan Africa, Southeast Asia, the Indian subcontinent, and China [2, 3, 11]. In some T. solium endemic areas, cysticercosis remains a major cause of seizures and epilepsy in humans [12–14]. Owing to travel and immigration, NCC poses a health burden even in non-endemic regions, such as the Americas and Europe [1, 15]. T. solium cysticercosis causes NCC in approximately 50 million people worldwide and is responsible for more than 50,000 deaths per year [16]. Studies have shown that the health burden of cysticercosis is greater than noted by public health systems [17, 18].
Humans and pigs both have important roles in the T. solium life cycle (Fig 1). Humans become infected with tapeworms by consuming raw or undercooked pork that contains cysticerci [19]. The larval tapeworms mature into adult worms in the human small intestine approximately 2 months after being consumed [19]. Humans are the only natural definitive hosts of T. solium [3]. The worm’s eggs or the most distal worm segments (proglottids) carrying mature eggs are periodically released/detached and subsequently discharged into the environment with human stool [3, 20]. Via the fecal-oral route, these eggs can infect the same person (auto-infection) or other people through direct contact with tapeworm carriers, or the ingestion of contaminated food or water [19, 21]. Pigs are infected when they consume infectious eggs (gravid proglottids) found in human excrement, food, water, or soil [3]. Humans and pigs can both act as intermediate hosts. After ingestion, embryos are released (oncosphere) and migrate through the intestinal mucosa. Later, the larval stages (cysticercus) commonly infest host body organs such as muscle, brain, eyes, subcutaneous tissues, and viscera via the blood circulatory system [3]. Subsequently, humans become infected by consuming raw or undercooked pork containing cysticerci, and the larvae mature into adult worms in the small intestine in approximately 2 months [19].
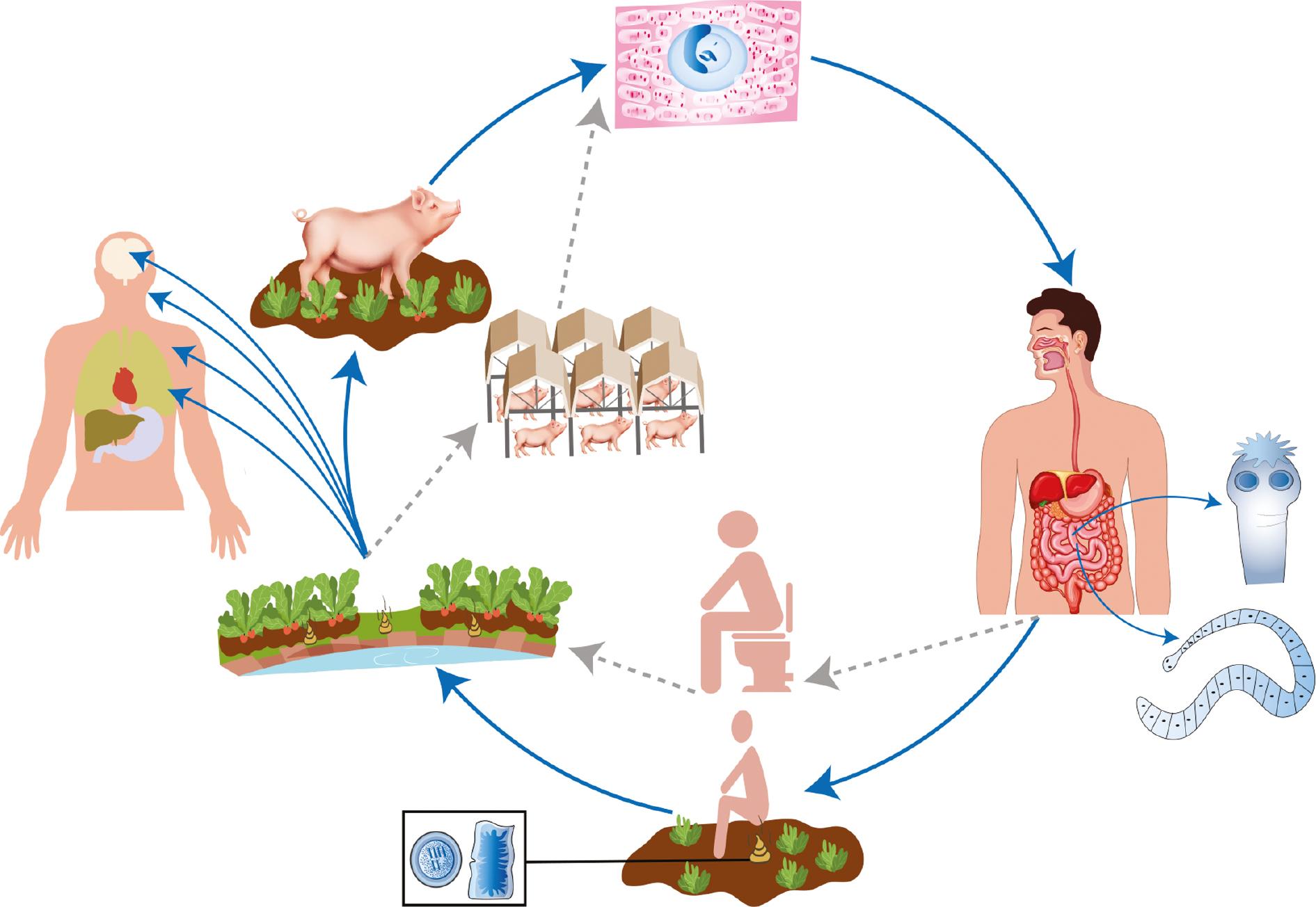
The life cycle and mode of transmission of Taenia solium in humans and pigs.
The blue cycle indicates the typical life cycle of T. solium. After consumption of raw or undercooked pork containing cysticerci, humans become infected, and the tapeworms colonize the gut. The larval tapeworms mature into adult worms in the human small intestine in approximately 2 months. The worm’s eggs or the most distal worm segments (proglottids) carrying mature eggs are periodically released/detached and subsequently discharged into the environment with human stool. Pigs are infected when they consume infectious eggs (gravid proglottids) in human excrement, food, water, or soil. Humans and pigs both serve as intermediate hosts. Embryos are released (oncosphere) after ingestion and migrate through the intestinal mucosa. Later, the larval stages (cysticercus) commonly infest organs in the host, such as the brain, eyes, subcutaneous tissues, and viscera via the blood circulatory system. Subsequently, humans become infected by consuming raw or undercooked pork containing cysticerci, and the larval stage matures into adult worms in the small intestine in approximately 2 months. The gray dashed cycle represents the changed T. solium life cycle. As a result of a structural shift in pig farming from backyard to large-scale intensive practices, as well as the toilet revolution aimed at improving sanitary conditions, the transmission of T. solium between humans and pigs has been significantly attenuated, and the disease’s spread is gradually being halted.
Domestication of wild boars began 9,000 years ago, and domestic pig rearing has been a common practice in Asian countries for more than 2,500 years [22]. Cysticercosis has long been a worldwide zoonosis transmitted between pigs and humans; currently, most cases in China are imported [23]. The prevalence of T. solium taeniasis/cysticercosis in China has significantly decreased in recent decades as a result of ongoing health education and promotion, meat inspection, chemotherapy, and other measures [23–25]. Furthermore, dramatic changes in pig farming patterns and human sanitary conditions have occurred in China’s rural communities over the past few decades [26, 27]. Herein, we review the epidemiology, traditional control measures, and ecological determinants that contributed to the decline in T. solium taeniasis/cysticercosis in China.
We searched PubMed, Web of Science, MEDLINE, ScienceDirect, the China National Knowledge Infrastructure (CNKI), and WANGFANG DATA for publications written in English or Chinese, and containing epidemiology records for humans and pigs, by using the search terms “Taenia solium” AND “China” OR “cysticercosis” AND “China” OR “taeniasis” AND “China” OR “Cysticercus cellulosae” AND “China.” We restricted our search to literature published before April 31, 2021. The titles and abstracts of the literature, followed by the full articles, were screened for inclusion in the epidemiology summary in this article. Reference lists from recent reviews were also used to identify original records. Additional key references were retrieved from the personal publication databases of all coauthors.
EPIDEMIOLOGICAL RECORDS
Three national surveys
China, to date, has undertaken three nationwide surveys on human intestinal parasitic diseases through morphological examination (with the Kato Katz method) (Table 1). In the first national survey (1988–1992), 1,477,742 individuals from 30 provinces (of 31 provinces (autonomous regions and municipalities) in mainland China) were investigated, and 2,449 individuals from 28 provinces were found to have Taenia spp. infections [28]. On the basis of these findings, the overall number of Taenia spp. infections in the country was estimated to be approximately 1.3 million [29]. The second nationwide survey (2001–2004) was conducted in 31 provinces, with a total sample size of 356,629 people; 983 people from 12 provinces (38.7%, 12/31) were determined to have Taenia spp. infection [30]. According to these findings, an estimated 0.55 million people were infected in total [30]. In the third nationwide survey (2014–2015), Taenia spp. was found in 1,752 people from 12 provinces (38.7%, 12/31), among 617,441 people from 31 provinces [31]. An average prevalence of 0.06% was calculated, thus implying that 0.37 million people were infected [31].
National surveys on human taeniasis/cysticercosis in China.
| Surveys | Sampling periods | Number of provinces surveyed/total number of participants | Positive provinces/participants | Estimated prevalence in the whole nation | Estimated infection cases in the whole nation |
|---|---|---|---|---|---|
| First | 1988–1992 | 30/1,477,742 | 28/2,449 | 0.17% | 1.3 million |
| Second | 2001–2004 | 31/356,629 | 12/983 | 0.28% | 0.55 million |
| Third | 2014–2015 | 31/617,441 | 12/1,752 | 0.06% | 0.37 million |
The three national human parasitic disease surveys in China revealed a significant decline in human T. solium taeniasis/cysticercosis. T. solium cysticercosis has historically been prevalent in northeastern, southwestern, and central China. The three national surveys have indicated that the occurrence of taeniasis has decreased considerably in most areas of China. Nevertheless, infection rates have been slightly higher in areas of southwest China with poor socioeconomic conditions, particularly Tibet, Sichuan, and Yunnan [23, 32, 33].
Field prevalence of porcine cysticercosis
Some of the epidemiology of porcine cysticercosis, a human taeniasis/cysticercosis infection, has been documented. Most related studies reported in the Chinese literature have been conducted during slaughter quarantine. A comparison of the prevalence of pig cysticercosis in different years in the same area indicated that the prevalence has significantly decreased (Fig 2). Because humans are the only natural definitive host of T. solium, pigs become infected by consuming human feces as well as contaminated food, water, or dirt [3, 34].

Investigations of porcine cysticercosis by region, and the degree of intensive pig farming and sanitary toilet coverage over the past few decades.
The prevalence of porcine cysticercosis has been declining, according to a comparison of documented cases during slaughter quarantine in different years in the same region. Furthermore, the degree of intensive pig farming (from 2.5% in 1985 to 43.3% in 2015) and sanitary toilet coverage (from 7.5% in 1993 to 81.7% in 2017) in Chinese rural areas increased each year during these periods.
Several field prevalence investigations of porcine cysticercosis have revealed a significant decrease in the disease prevalence in pigs. During pork meat inspection, macroscopic observations and microscopic examinations based on parasite morphology are the primary detection methods for pig cysticercosis. Although, only a few documents were available for porcine cysticercosis in China, it could also reveal a significant decline for infections (Fig 2).
TRADITIONAL CONTROL MEASURES
Health education and promotion
Beyond China, health education and promotion have been shown to be highly beneficial in controlling T. solium taeniasis/cysticercosis in rural communities worldwide [24, 25, 35, 36]. This comprehensive social and political process has included not only improving knowledge and developing life skills for individuals, but also changing societal, environmental, and economic conditions to decrease harmful effects on individuals and communities [24, 37]. To effectively control T. solium taeniasis/cysticercosis in China, health promotion and education measures are directed primarily at the susceptible population and infected individuals [24, 25, 35].
Meat inspection
Pork contaminated with cysticercus is a major source of T. solium taeniasis and cysticercosis in humans [38]. Meat inspection of pigs at slaughter is an important public health measure to prevent T. solium transmission to humans [39, 40]. Meat inspection during pig slaughter has also been implemented in China as a crucial public health <discrhypen>precaution [19].
Chemotherapy
In most human cysticercosis cases, antiparasitic drugs, such as praziquantel, niclosamide, and albendazole, are used to destroy live or degenerating cysticerci [13]. Recently, a combination of pumpkin seeds and areca nut has been explored for taeniasis; praziquantel and albendazole can also be simultaneously administered for cysticercosis, and have shown promising efficacy and low adverse effects [23]. Surgery is required in severe cysticercosis cases, to reduce intracranial pressure and resolve hydrocephalus in NCC, OCC, and other types [41, 42, 43].
ECOLOGICAL DETERMINANTS
Shift in pig farming patterns from backyard to large-scale intensive practices
In general, three types of pig farming patterns are used in China: backyard farms (raising fewer than five pigs per year), specialized household farms (annual pig production of 5 to 500 heads, most commonly 50 to 200 heads), and large-scale intensive pig farms (500 to 50,000 pigs per year, or more) [26]. Smallholders (backyard farms) have produced most of China’s pork for several thousand years. Before 1978, at least 95% of the pork in China was produced on these “backyard farms,” which farmed fewer than five pigs per year along with crops and other domestic animals [44]. In backyard farms, pigs’ feeding troughs, sanitary toilets, and pigsties are usually located adjacently; consequently, pigs easily consume human stool, or contaminated food and water [44].
After China’s Reform and Opening (which began in 1978), the structure of pig farming began to shift toward the large-scale intensive commercial pig husbandry that persists today [24]. The production scale on these farms varies from 500 to 50,000 pigs per year, and is rapidly increasing each year. A single farm may often produce 100,000 pigs in a single year [26]. These farms were responsible for only 2.5% of the overall pork yield in 1985, but their share increased to 21.8% by 2007 and reached 43.3% in 2015 [45]. Human toilets and pigsties are completely separated, and pigs are prevented from consuming human waste or contaminated feed and water (Fig 1).
Specialized household farms are intermediate in scale between backyard farms and large-scale intensive pig farms. The proportions of farming practices significantly differ among areas; for example, in Sichuan Province, specialized household farms account for 25% of the total, whereas large-scale intensive pig farms account for only 5% [26]. Backyard farms produced approximately 27% of all pork produced in China in 2007, whereas the share of smallholders (backyard farms) was significantly higher in some areas, thus demonstrating the imbalanced development of pig farming across geographical regions. For example, during the same time period (2007), backyard farmers produced 70% of the pigs in Sichuan Province, China, which is the historical and current national leader in pork production, whereas Guangdong Province produced only approximately 20% [26].
Overall, pig farming is irreversibly progressing from backyard to large-scale intensive farms in China [26]. As pig farming practices have changed, contact of pigs with human excreta, and contaminated feed and water has decreased. Thus, the transmission of T. solium between humans and pigs has largely attenuated in some large-scale intensive pig farming areas. Consequently, the shifting pig farming patterns have created conditions preventing intermediate host pigs from becoming infected with cysticercus, thus disrupting the T. solium transmission pathway and ultimately halting disease spread [24].
Sanitary toilet revolution aimed at improving sanitary conditions
Poor sanitation and a lack of sanitary toilets have been identified as the main risk factors for the presence of T. solium circulating infections in epidemic areas [46]. Human cysticercosis was once more common in China’s vast rural areas, which had poor sanitation and a lack of sanitary toilets [47, 48].
The initial goal of the sanitary toilet revolution was to promote the use of sanitary toilets in China’s vast rural communities [49]. Currently, the toilet revolution is being expanded to include the overall sanitary system rather than only toilets [50]. In rural China, the number of sanitary toilets has significantly increased over the past few decades, and the toilet revolution has made substantial progress in improving the sanitation infrastructure [51]. Human sanitary toilet coverage increased from 7.5% in 1993 to 81.7% in 2017 [49, 52].
The toilet revolution has been aimed at ensuring the hygienic separation of human fecal matter, thus avoiding human contact and preventing human fecal contamination of the environment [49, 50]. The volume of untreated excrement (which may contain worm eggs or gravid proglottids) released into the environment has significantly decreased, as has the likelihood of pigs consuming human excreta (Fig 1).
During the ongoing toilet retrofitting campaign in rural areas, China has made substantial progress toward a new socialist countryside with significantly improved sanitary conditions in rural communities [52]. These environmental reconstructions have disrupted the life cycle and transmission of T. solium between humans and pigs, thereby preventing disease spread.
COMPREHENSIVE DETERMINANTS
T. solium is transmitted through the fecal-oral route between humans and pigs. The spread of human taeniasis/cysticercosis is promoted by inadequate sanitation, poor hygiene, the use of human excrement (untreated or partially treated) in agriculture, inappropriate food preparation, insufficient knowledge regarding the risk of infection during visits to endemic locations, and ingestion of raw or undercooked pork, particularly in areas where pigs are reared in poor conditions [53–56]. Prevention and control measures for Taenia spp. infections and cysticercosis should include sanitary cleanliness, health education, and treatments consisting of human chemotherapy with better diagnostic techniques for taeniasis, and porcine chemotherapy or vaccination [57].
The past few decades have seen substantial continuing efforts to eliminate human cysticercosis, including health promotion, meat inspection, chemotherapy, and combined comprehensive measures, which are have been the primary control measures for the disease [24]. However, additional factors, such as changes in pig farming practices and the development of modern toilets, have been found to affect human and pig T. solium taeniasis/cysticercosis. These changes can successfully cut off the T. solium transmission route. Although these two factors have often been described in the literature [2, 58, 59], those articles have not adequately covered the fundamental importance of decreasing human cysticercosis by halting disease transmission.
The spread of T. solium taeniasis and cysticercosis is gradually being stopped with the shift from small-scale to large-scale intensive pig farming practices, as well as the revolution in the use of sanitary toilets by humans. However, T. solium taeniasis and cysticercosis remain prevalent in several low-socioeconomic areas of China, particularly in rural communities in Sichuan, Yunnan, and Guangxi, where pigs are reared in substandard management conditions, and raw or undercooked pork are traditionally consumed [32, 44, 53–55]. Therefore, to completely eliminate cysticercosis, health education and promotion, meat inspection, and chemotherapy are necessary.
LIMITATIONS
Although the marked decline in taeniasis and cysticercosis infections in China has been widely acknowledged, limitations exist concerning the epidemiological records for both humans and pigs. Relatively low-sensitivity detection techniques, such as the Kato Katz method, were used during all three nationwide surveys for Taenia eggs. Regarding pig cysticercosis, only several documents were available for the same locations in China. Likewise, both control measures and ecological determinants have been considered to be comprehensive social processes that together lead to a gradual decline in taeniasis and cysticercosis in China. However, scant information was available regarding the individual and combined effect of the factors on the decline of this disease.
CONCLUSIONS
T. solium taeniasis/cysticercosis is an important food-borne zoonotic parasitic disease. Several factors have contributed to the decrease in taeniasis/cysticercosis in humans and pigs. Beyond health education and promotion, meat inspection at slaughterhouses and chemotherapy have contributed to China’s marked decrease in T. solium taeniasis/cysticercosis. Furthermore, the structural shift from backyard to large-scale intensive pig farming, and the toilet revolution’s improvements in sanitation have created conditions preventing the intermediate porcine hosts from acquiring cysticercosis. These two crucial factors should be considered to decrease or restrict the spread of T. solium between humans and pigs.



