1. INTRODUCTION
The placenta, a temporary organ formed during pregnancy, is responsible for fetal nutrition, respiration and excretion, and plays crucial roles in healthy fetal development [1]. Placental abnormalities are important causes of maternal hemorrhage during childbirth and postpartum hemorrhage. Placenta accreta spectrum disorders (PASDs) are critical complications in the second and third trimesters of pregnancy, and involve abnormal invasion of placental trophoblast cells into the myometrium. According to the depth of invasion, PASDs are divided into placenta accreta, placenta increta and placenta percreta, among which placenta increta and placenta percreta are collectively referred to as invasive placenta [2]. Invasive placenta is more likely than non-invasive placenta to cause hemorrhage during delivery or even emergency hysterectomy during surgery. However, owing to the lack of typical clinical symptoms and specific laboratory indicators, approximately one-half to two-thirds of puerperae are not clearly diagnosed before delivery. At present, prenatal diagnosis is based primarily on the history of relevant risk factors (such as maternal age and maternal history) and imaging examination findings [3]. Ultrasound remains used for first-line examination, but it has several limitations; notably, when the placenta is located in the posterior wall of the uterus, assessing the relationships among the placenta and the myometrium and its adjacent structures is difficult [4]. MRI has advantages of enabling multi-sequence, multi-directional imaging and providing high soft tissue contrast resolution, thus addressing the above shortcomings. In this study, a predictive model was established through logistic regression analysis based on clinical and MRI features, and a nomogram was constructed to intuitively predict the risk of invasive placenta, to provide an objective imaging basis for the formulation of individualized disposition plans aimed at ensuring good pregnancy outcomes.
2. MATERIALS AND METHODS
2.1 Patient selection
The data for 173 pregnant women with clinical suspicion of PASDs who underwent MRI and ultrasound in our hospital between December 2013 and June 2022 were collected. The inclusion criteria were as follows: (1) complete clinical and imaging data; (2) ultrasound confirmed or suspected PASDs, and MR examination performed within 1 week; and (3) delivery in our hospital, with a definite clinical diagnosis. The exclusion criteria were as follows: (1) incomplete clinical or imaging data; (2) delivery outside our hospital and no definite clinical diagnosis; and (3) poor MR image quality for diagnosis. A total of 162 pregnant women were ultimately included in this study; their average age was 31.88±4.34 years (range, 22–43 years). The mean number of gestational weeks at MR examination was 32.40±6.24 (range, 13–39). All pregnancy cases were in the second and third trimesters, including 27 in the second trimester and 135 in the third trimester. Three cases involved twin pregnancies, all with one placenta. In vitro fertilization and embryo transfer were performed in five pregnant women.
2.2 MR examination
The MR studies were performed on a 1.5 T MR scanner (GE 360 fiber optic, GE Medical Systems, Milwaukee, WI, USA). The scanning sequence and parameters were as follows. Sagittal FSE-T2WI: TR/TE 6666 ms/85 ms, FOV 360 mm × 360 mm, NEX 4; sagittal SSFSE-T2WI: TR/TE 891 ms/68 ms, FOV 360 mm × 360 mm, NEX 1; sagittal FIESTA-T2WI: TR/TE 4.1 ms/1.79 ms, FOV 360 mm × 360 mm, NEX 1; axial FSE-T1WI: TR/TE 589 ms/12.69 ms, FOV280 mm × 280 mm, NEX 2; axial FSE-T2WI: TR/TE 6666 ms/102 ms, FOV 280 mm × 280 mm, NEX 2; and coronal FSE-T2WI: TR/TE 3682 ms/68 ms, FOV 420 mm × 420 mm, NEX 2, layer thickness 5 mm, layer spacing 5 mm, eight-channel abdominal phased array surface coil.
Before the examination, pregnant women should ensure a filled bladder (approximately 300 ml volume) to help reveal the relationship between the lower uterine segment and the bladder [5]. Scans should be performed on patients in the supine position. The scan should range from the uterine fundus to the cervix, including both pelvic walls in coronal view.
2.3 Imaging analysis
Two radiologists, with at least 10 years’ experience in obstetric/fetal imaging and more than 17 years’ radiology experience, assessed MR image quality and reviewed the MRI signs to reach a consensus.
According to the 2020 Society of Abdominal Radiology (SAR) and European Society of Urogenital Radiology (ESUR) consensus on MR imaging for PASDs [5], and the 2020 American College of Radiology (ACR) guidelines for PASDs imaging examination [6], the MRI observation signs include the following ( Figures 1 and 2 ):
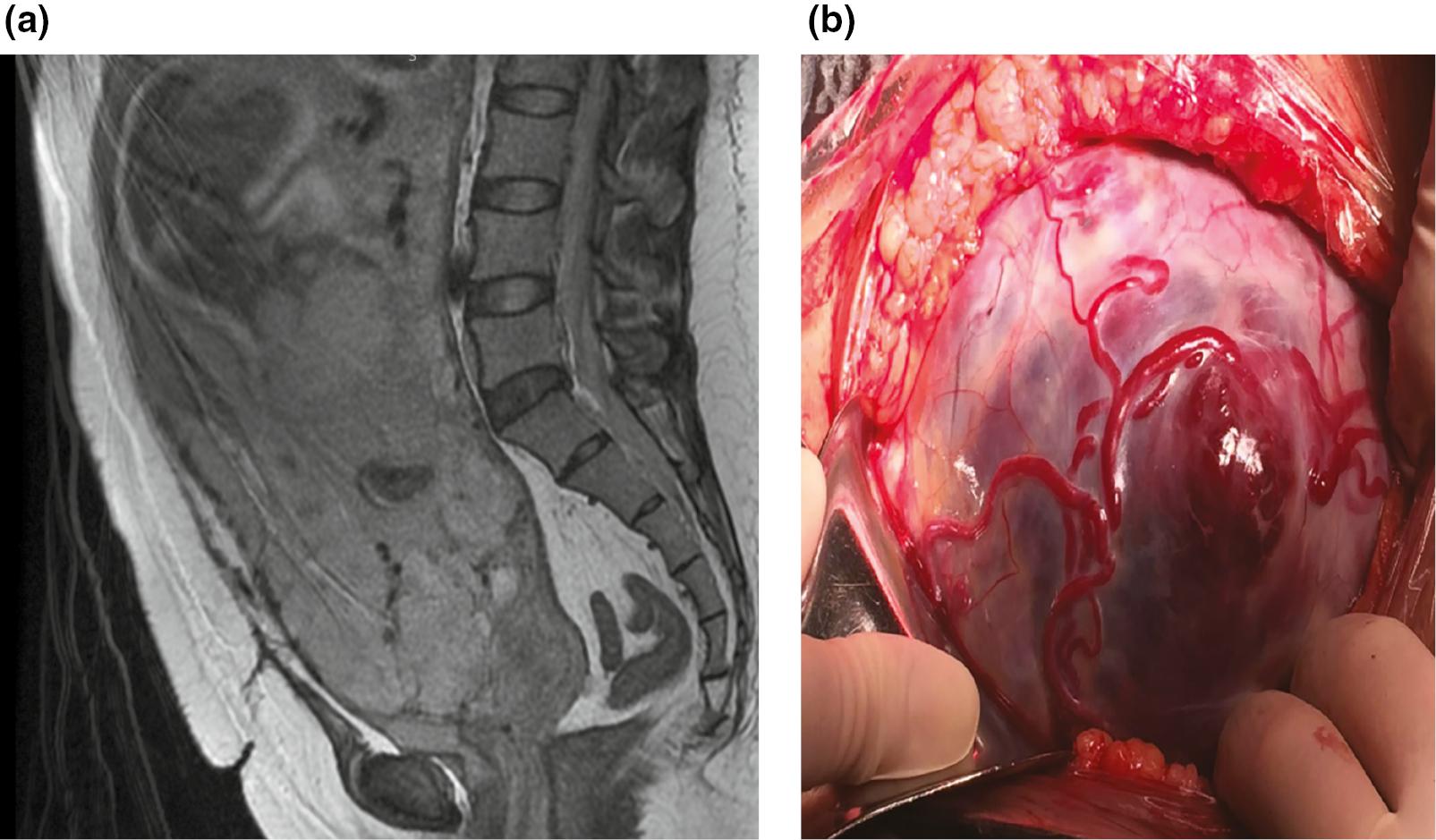
A case of complete placenta previa and placenta percreta in a 32-year-old pregnant woman with one prior cesarean delivery and one abortion.
MRI was performed at 36 weeks’ gestational age. (a) Sagittal T2WI, showing the bulging lower uterine segment. The normal myometrium disappeared and was replaced by enlarged and tortuous vessels, with local bulging of the attached placenta. The upper part of the cervical canal was compressed backward, the normal signal of the anterior cervical lip disappeared, and local placental tissue attachment was observed. The demarcation between the lower uterine segment and the bladder was unclear. (b) Intraoperative findings indicated that the lower segment of the uterus was bulging, with abundant blood supply on the surface and multiple angry vessels, leaving only a thin serosa layer. The placenta completely covered the lower segment of the uterus and the internal cervical os, and partially protruded outward. The lower uterine segment and the bladder were extensively densely adhered. Massive bleeding occurred during the operation, and the placenta could not be completely stripped, so subtotal hysterectomy was performed.
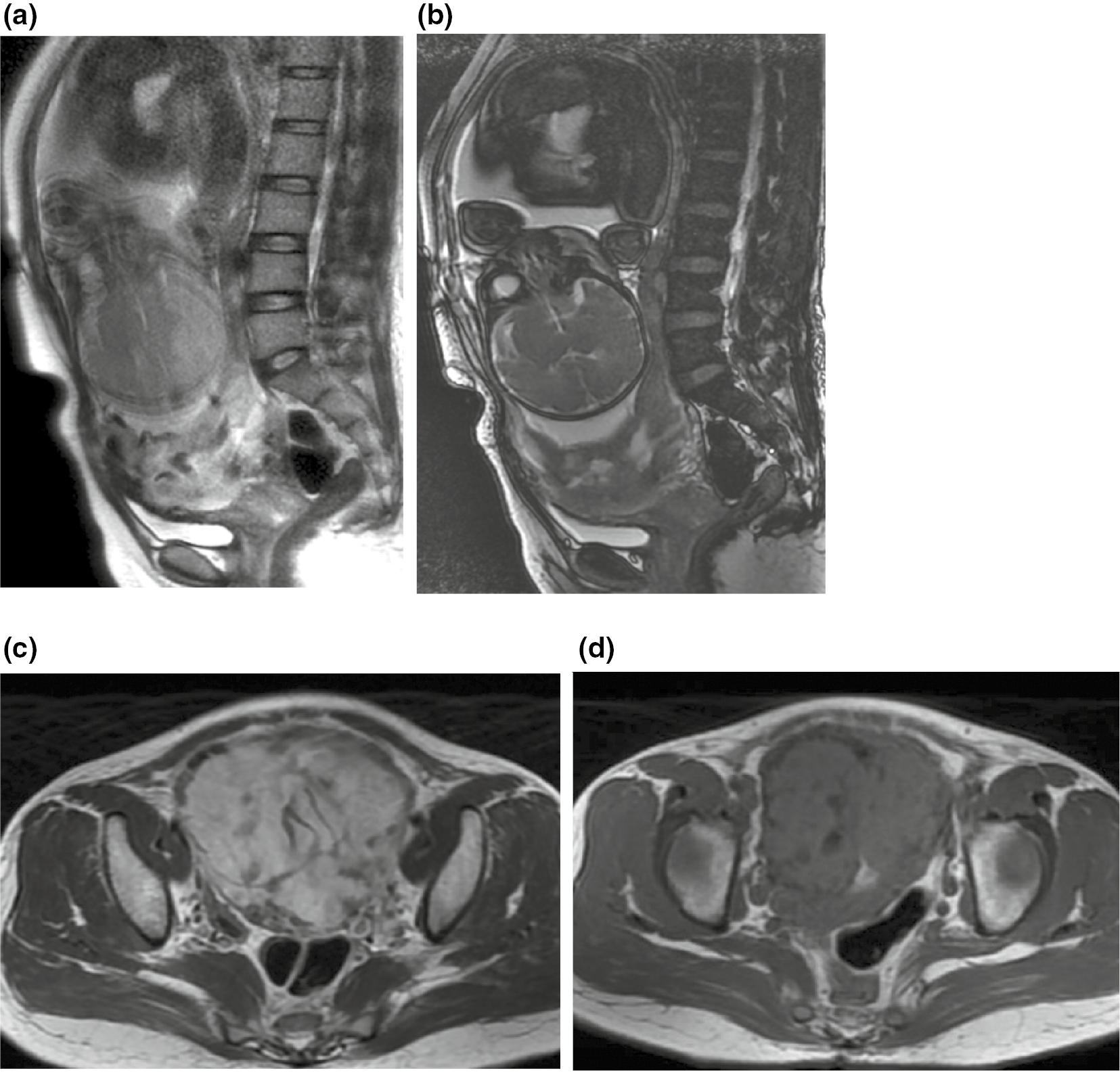
A case of complete placenta previa and placenta percreta in a 36-year-old pregnant woman with one prior cesarean delivery and two abortions.
MRI was performed at 39 weeks’ gestational age. (a-d) FSE T2WI, T1WI and FIESTA, showing uneven thickness of the placenta and marked heterogeneity. FSE T2WI shows tortuous short-striped low signal shadows in the placenta, and FIESTA shows thickened and tortuous high-intensity vascular shadows. Sagittal T2WI and FIESTA show local bulging of the lower uterine segment, loss of the low-signal line at the placental-uterine interface, and tortuous thickening of blood vessels in the right anterior muscle layer of the uterus. The placenta completely covered the internal os of the cervix and appeared to be partially protruding into the endocervix, with indistinct demarcation from the posterior lip of the cervix. Intraoperative findings indicated that the placenta was densely adhered to the anterior wall of the lower uterine segment and the posterior lip of the cervix, and only the serosal layer was seen on the right anterior wall of the lower uterine segment.
Placental/uterine bulge: the placenta partially protrudes, thus resulting in loss of the normal “pear shape” of the uterus, and the presence of enlargement of the lower segment.
Myometrial interruption: the myometrium loses its continuity and is interrupted at the site where the placental tissue protrudes.
Loss of the placental-myometrial interface: focal defects of the hypointense uteroplacental interface are observed, with indistinctness of myometrial delineation.
Placenta location: whether the placenta covers internal cervical os is determined.
Marked placental heterogeneity: the signal of the placenta is clearly uneven.
T2-dark intraplacental bands: intraplacental band-shaped or nodular areas with low signal intensity are present on both FSE-T2WI and FIESTA-T2WI.
Intraplacental abnormal vascularization: enlarged and tortuous vessels represented by flow void signals, with a diameter > 6 mm within the placenta, show low signal on FSE-T2WI and high signal on FIESTA-T2WI.
Abnormal vascularization of the placental bed: the placental bed constitutes the decidua and adjacent myometrium underlying the placenta. The normal myometrium disappears and is replaced by enlarged and tortuous vessels.
Cervical invasion: dilation of the internal os of the cervix, shortening of the cervix, abnormal signal of the cervical wall, and protrusion of the placenta into the cervical canal are observed.
Bladder invasion: “tenting” of the bladder, irregular or interrupted bladder wall, and numerous tortuous signal voids traversing the space from the uterus to the bladder are observed.
2.4 Standard of reference
According to intraoperative findings and/or pathological diagnosis, 162 puerperae were divided into an invasive placenta group and a non-invasive placenta group. The placenta increta and placenta percreta were included in the invasive placenta group, whereas placenta accreta and normal placenta were included in the non-invasive placenta group. The reference standards were as follows [2]: placenta accreta: the placenta was closely adhered to the uterine wall, which was difficult to peel off manually but could be completely peeled off, or the pathology showed placental villi invading the myometrium surface; placenta increta: placental tissue was implanted in the myometrium and could not be completely removed manually, or placental villi were found in the myometrium by pathology; placenta percreta: the placental tissue invaded the entire layer of the myometrium, and entered or even penetrated the serosa layer and reached adjacent organs, or pathology revealed placental villi in the uterine serosa layer or adjacent tissues.
2.5 Statistical analysis
All statistical analyses were performed in Stata software (version 14.1, IC, StataCorp). The enumeration data and proportions are expressed as numbers of cases (n) and percentages (%). Measurement data are expressed as mean ± standard deviation (x–±s). Fisher’s exact test was used for univariate analysis, and the stepwise regression method was used for multivariate logistic regression analysis to identify independent risk factors for invasive placenta, establish a predictive model, and construct the nomogram. The model validation used the cross-validation method. A total of 122 cases were randomly selected as a derivation cohort, and the other 40 cases were used as a validation cohort to verify the model. Finally, the predictive efficiency of the model was evaluated with receiver operating characteristic (ROC) curve analysis. P values <0.05 were considered statistically significant.
3. RESULTS
3.1 Intraoperative and pathological diagnosis
According to intraoperative findings and pathological results, among the 162 puerperae, 91 were diagnosed with invasive placenta (47 with placenta increta and 44 with placenta percreta), and 71 were diagnosed with non-invasive placenta (25 with placenta accreta and 46 with normal placenta; Figure 3 ).
3.2 Univariate analysis of clinical and MRI characteristics
Among the 162 cases, only 7 cases had normal placental position, and the remaining 155 cases (95.7%, 155/162) were all placenta previa. because large deviations in the numbers of cases were observed, the indicator of placenta position was not included in the univariate analysis.
The results of univariate analysis of clinical and MRI characteristics between invasive placenta and non-invasive placenta are summarized in Table 1 . Ten indicators—prior caesarean delivery, loss of the placental-myometrial interface, myometrial interruption, placental/uterine bulge, marked placental heterogeneity, T2-dark intraplacental bands, abnormal vascularization of the placental bed, intraplacental abnormal vascularization, cervical invasion and bladder invasion—showed statistically significant differences between groups (P<0.05).
Univariate analysis results of clinical and MRI characteristics.
| Influencing factors | Invasive placenta | P-value | |
|---|---|---|---|
| No (n = 71) | Yes (n = 91) | ||
| Clinical indicators | |||
| Age (years) | 0.49 | ||
| < 35 | 53 (75%) | 63 (69%) | |
| ≥ 35 | 18 (25%) | 28 (31%) | |
| Gestational weeks | 0.53 | ||
| ≤ 27 | 10 (14%) | 17 (19%) | |
| ≥ 28 | 61 (86%) | 74 (81%) | |
| Prior caesarean delivery | <0.01 a | ||
| No | 23 (32%) | 7 (8%) | |
| Yes | 48 (68%) | 84 (92%) | |
| Prior abortion | 0.75 | ||
| No | 31 (44%) | 43 (47%) | |
| Yes | 40 (56%) | 48 (53%) | |
| MRI indicators | |||
| Myometrial interruption | <0.01 a | ||
| No | 67 (94%) | 35 (38%) | |
| Yes | 4 (6%) | 56 (62%) | |
| Abnormal vascularization of the placental bed | <0.01 a | ||
| No | 49 (69%) | 15 (16%) | |
| Yes | 22 (31%) | 76 (84%) | |
| Placental/uterine bulge | <0.01 a | ||
| No | 64 (90%) | 23 (25%) | |
| Yes | 7 (10%) | 68 (75%) | |
| Loss of the placental-myometrial interface | <0.01 a | ||
| No | 60 (85%) | 28 (31%) | |
| Yes | 11 (15%) | 63 (69%) | |
| Cervical invasion | <0.01 a | ||
| No | 64 (90%) | 61 (67%) | |
| Yes | 7 (10%) | 30 (33%) | |
| Bladder invasion | <0.01 a | ||
| No | 68 (96%) | 72 (79%) | |
| Yes | 3 (4%) | 19 (21%) | |
| Marked placental heterogeneity | <0.01 a | ||
| No | 65 (92%) | 37 (41%) | |
| Yes | 6 (8%) | 54 (59%) | |
| Intraplacental abnormal vascularization | <0.01 a | ||
| No | 67 (94%) | 45 (49%) | |
| Yes | 4 (6%) | 46 (51%) | |
| T2-dark intraplacental bands | <0.01 a | ||
| No | 69 (97%) | 67 (74%) | |
| Yes | 2 (3%) | 24 (26%) | |
aStatistically significant at P<0.05.
3.3 Multivariate logistic regression analysis and model construction
The above ten indicators were included in the multivariate logistic regression analysis. Marked placental heterogeneity (X1), loss of the placental-myometrial interface (X2), placental/uterine bulge (X3) and abnormal vascularization of the placental bed (X4) were found to be independent risk factors for invasive placenta (P<0.05; Table 2 ). The four factors were included in the logistic regression equation to construct a risk prediction model, logit(P)=−2.351+1.661X1+2.008X2+ 2.064X3+1.449X4, and a nomogram was constructed ( Figure 4 ).
Multivariate logistic regression analysis results.
| Influencing factors | Partial regression coefficient | 95% CI | P-value |
|---|---|---|---|
| Prior caesarean delivery | 1.398 | −0.374 to 3.169 | 0.122 |
| Myometrial interruption | −0.444 | −2.469 to 1.58 | 0.667 |
| Abnormal vascularization of placental bed | 1.906 | 0.479 to 3.332 | 0.009 a |
| Placental/uterine bulge | 1.960 | 0.534 to 3.385 | 0.007 a |
| Loss of the placental-myometrial interface | 2.769 | 1.09 to 4.448 | 0.001 a |
| Cervical invasion | 1.249 | −0.587 to 3.084 | 0.182 |
| Bladder invasion | −1.791 | −4.131 to 0.549 | 0.134 |
| Marked placental heterogeneity | 2.315 | 0.504 to 4.126 | 0.012 a |
| Intraplacental abnormal vascularization | −1.144 | −3.149 to 0.861 | 0.264 |
| T2-dark intraplacental bands | 2.101 | −0.51 to 4.712 | 0.115 |
aStatistically significant at P<0.05.
3.4 Performance of the predictive model
The ROC curve for the derivation cohort indicated an AUC of 0.925; the diagnostic coincidence rate was 87.7%, the sensitivity was 87.5%, and the specificity was 88.0% ( Figure 5, Table 3 ). The ROC curve for the validation cohort indicated an AUC of 0.974; the diagnostic coincidence rate was 90.0%, the sensitivity was 94.7%, and the specificity was 85.7% ( Figure 6, Table 3 ). These results demonstrated that the predictive model performed well in identifying the risk of invasive placenta.
4. DISCUSSION
In recent years, the incidence of PASDs has increased nearly tenfold [7]. Advanced maternal age, placenta previa, and history of uterine surgery (including cesarean delivery, curettage and assisted reproductive technology) are all risk factors for invasive placenta, among which placenta previa and prior cesarean delivery are the two most important factors [3, 7]. Invasive placenta is associated with difficulty in peeling off the placenta during delivery. As many as 40–60% of peripartum hysterectomies are due to invasive placenta [8, 9]. MR avoids the limitations of ultrasound in diagnosing placental location in the posterior wall of the uterus and in the presence of maternal obesity, and provides a useful supplement to ultrasound. In this study, independent risk factors for invasive placental implantation were screened from 13 clinical and MRI features in the following order: placenta/uterine bulge, loss of the placental-myometrial interface, marked placental heterogeneity and abnormal vascularization of the placental bed. The predictive model constructed on the basis of the above four factors had good risk assessment performance for invasive placental implantation and therefore may help decrease the occurrence of adverse pregnancy outcomes.
4.1 Univariate analysis of clinical and MRI features
Among the four clinical features included in this study, no statistically significant difference in age was found between the invasive placenta group and the non-invasive placenta group (P>0.05). This finding is inconsistent with the literature [7, 10], and may be associated with the high prevalence of maternal age below 35 years in this group (127/162, 78.40%).
Because pregnant women with PASDs usually undergo cesarean delivery at 34–35 weeks after fetal lung maturity [11], most patients in this study were in late pregnancy (135/162, 83.3%), with an average number of 32.40±6.24 gestational weeks. No significant difference in the number of gestational weeks was observed between groups (P>0.05). The difference in abortion history between groups did not statistically significantly differ (P>0.05), in agreement with literature reports [4, 10]. A significant difference in the history of cesarean delivery was found between groups (P<0.05). Previous cesarean delivery may lead to decidua loss at the uterine scar, where the placental villi are prone to implant in the uterine muscle [12].
Nine MRI characteristic indicators showed statistically significant differences between groups (P<0.05). Except for cervical invasion, which has been reported in few literature reports [13, 14], the other indicators have been reported in several literature reports [6, 7, 12].
4.2 Risk prediction model for invasive placenta
In 2016, Ueno et al. [15] first established a predictive model for invasive placenta by using the Likert scale 5-point method to score six MRI signs. Knight et al. [16] have predicted PASDs according to combined clinical, ultrasound and MRI scores. Both methods have good predictive effects. In this study, four independent risk factors selected by logistic regression analysis were used to construct a risk prediction model for invasive placenta. The AUC of the derivation set and the validation set was 0.925 and 0.974, respectively, thus indicating that both models had highly accurate diagnostic performance. The nomogram provided a visualization of the model. By assessing the scores for each risk factor, the risk of invasive placenta can be evaluated, thus providing an intuitive reference for the selection of clinical surgical plans.
4.3 MRI signs and the risk of invasive placenta
When the placenta invades the myometrium, it may partially bulge outward, thus causing partial protrusion of the uterine wall into the pelvic cavity, bulging of the lower uterine segment, and loss of the normal pear-shaped uterine structure. Jha et al. have suggested that placental bulge, combined with other signs, can indicate 100% of cases of myometrial invasion [17]. Kilcoyne et al. have suggested that local uterine bulge is a reliable sign for identifying invasive placenta. Because local uterine bulge is affected by placenta bulge [18], in this study, referring to the joint consensus of SAR and ESUR [6], we combined the two indicators into placental/uterine bulge. This sign was the most significantly associated with invasive placenta.
Loss of the placental-myometrial interface on the nomogram had the second highest association with the risk of invasive implanta, after placenta/uterine bulge. It is a direct sign of invasive placenta. Normally, a hypointense line is present between the uteroplacental interface on T2WI. When the placental villi invade the myometrium, focal loss of the uteroplacental interface can be observed. Bour et al. have suggested that the focal loss of the placental-myometrial interface is the most accurate diagnostic feature for invasive placenta [10]. Similarly, the results of this study revealed a significant association between this loss and invasive placenta. In addition, the joint consensus of SAR and ESUR have proposed that diagnostic sensitivity is increased when this loss co-exists with myometrium disruption [6]. However, the normal myometrium becomes thinner as pregnancy progresses [19], and thinning of the myometrium in the third trimester hinders determination of whether interruption or just physiological thinning is present. Because most pregnant women in this study were in the third trimester, the sign of myometrial disruption did not show significance after multivariate regression analysis.
Marked placental heterogeneity on the nomogram, as compared with the above two signs, was associated with a relatively lower risk of invasive implanta. Placental heterogeneity may be due to intraplacental hemorrhage, infarction or fibrin deposition [20]. A significant difference in the uneven signal generated by placental degeneration in late pregnancy compared with normal pregnancy is observed. Placental heterogeneity is also affected by T2-dark bands and abnormal vascularization. Intraplacental abnormal vascularization manifests as enlarged and tortuous vessels, represented by flow void signals within the placenta. T2-dark bands can be seen in the mature placenta, and are hypointense on both FSE and FIESTA sequences. These bands can be distinguished from intraplacental vessels on FIESTA. Because of the bright-blood effect of FIESTA, the intraplacental vessels show a high signal in this sequence. Alamo et al. have suggested that T2-dark bands are a distinctive feature of invasive placenta [19]. However, in this study, the dark bands appeared in a small number of cases (16.0%, 26/162), possibly because some pregnant women were not scanned with a FIESTA sequence to identify enlarged vessels in the placenta.
Abnormal vascularization of the placental bed involves the invasion of placental villi into the myometrium, thus stimulating abnormal enlarged uteroplacental circulation vessels or the formation of arteriovenous fistulas [21, 22]. This condition manifests as enlarged and tortuous vessels in the placental-uterine interface or myometrium, and is closely associated with intraoperative massive bleeding [20], and can help clinicians select an appropriate surgical area and reduce the risk of massive intraoperative bleeding. The results of this study indicated that this sign is also an independent risk factor for invasive placenta.
Although the joint consensus issued by SAR and ESUR does not include the sign of cervical invasion, the paracervical vascular hyperplasia caused by invasive placenta often causes hemorrhage, and the shortening of the cervical canal makes placenta percreta operations more difficult [13, 14]. Therefore, this study included signs of cervical invasion. Herein, cervical invasion did not show significance after multivariate regression analysis, possibly because of the small number of cases with cervical invasion. However, among the 16 cases of hysterectomy in this study, 9 cases showed cervical invasion (56.3%, 9/16), thus indicating that cervical invasion strongly affects prognosis and also plays a crucial role in the selection of surgical options.
Invasive placenta usually occurs in the lower segment of the uterus, particularly at the cesarean delivery scar. Therefore, the placenta often invades the bladder, thus resulting in the presence of many abnormal blood vessels at the uterine-bladder interface. If the abnormal blood vessels are not properly treated during the surgery hemorrhage will still occur even after hysterectomy [21–23]. The ACR guidelines recommend that the bladder be moderately filled before MR examination to enable better identification and assessment of the lower uterine segment with the cesarean scar area [12]. However, in this study, because some pregnant women underwent emergency MR examination, the bladder filling was poor, and the bladder-uterine interface showed insufficient clarity, thereby affecting the accurate assessment of bladder invasion.
4.4 Limitations
This study has several limitations. First, it was a single-center retrospective study with a small sample size, thus making selection bias difficult to avoid. Second, MR enhancement has been reported to improve the diagnostic accuracy of invasive placenta [12, 24]. Although the ACR guidelines suggest that enhanced scans can be used in pregnant women who terminate pregnancy immediately [12], doubts persist regarding the safety of gadolinium contrast agents on the fetus. Consequently, no enhanced scans were performed in this study, thus potentially biasing the results.
4.5 Outlook
In recent years, radiomics has rapidly developed and has played an important role in identifying various diseases and predicting therapeutic effects. Romeo et al. have used texture features combined with machine learning to evaluate whether pregnant women with placenta previa have invasive placenta [25]. Sun et al. have used texture analysis to quantify heterogeneity within the placenta and have established a radiomics model based on MRI texture to predict placental implantation [26]. The results of this study and prior research indicate that combined radiomics can improve the diagnostic accuracy of placental implantation. This method has good prospects for predicting PASDs before delivery. Use of radiomics may decrease bias and increase the accuracy of the results.






