Introduction
Malnutrition is a frequently observed complication affecting 37%–56% of individuals with heart failure (HF) [1]. Beyond impeded nutrient intake and absorption due to diminished left ventricular ejection fraction (LVEF), malnutrition can impair immune function, promote muscle wasting, disrupt myocardial energy metabolism, and enhance inflammatory responses, all of which contribute to HF progression [2–4]. In a meta-analysis using multiple malnutrition screening tools to investigate nutritional status and its effects on all-cause mortality in 12,537 patients with HF, malnourished patients had nearly twice the risk of non-malnourished patients with HF (HR 2.15; 95% CI 1.89–2.45) [1]. This finding highlights the urgent need for integrating nutritional considerations into patients’ comprehensive care plans to improve outcomes. No consensus exists regarding the optimal nutritional assessment tool for patients with HF [5, 6]; however, the Controlling Nutritional Status (CONUT) index and Geriatric Nutritional Risk Index (GNRI) are frequently used, validated tools that have been studied in HF [7–10].
In managing patients with HF and LVEF <50%, guideline-directed medical therapy (GDMT), including β-blockers, renin-angiotensin system inhibitors (RASIs), aldosterone antagonists (MRAs), and sodium-glucose cotransporter 2 inhibitors (SGLT2i), is critical for enhancing survival rates and alleviating symptoms [11, 12]. In-hospital patients, under close surveillance, have an optimal window for initiation and up-titration of medications, a practice endorsed by both American and European guidelines [13]. Numerous experts have proposed algorithms for managing these treatments, often involving the combination of at least three types of medications and aiming for high GDMT doses as close as possible to the target doses [14–16]. However, concerns exist regarding SGLT2i inclusion in treatment regimens for individuals with low body weight or cachexia [17, 18]. At discharge, prescription of GDMT medications is crucial for maintaining patients’ long-term pharmacological regimens based on the regimens established during hospitalization [19, 20].
Nevertheless, GDMT initiation and dose up-titration are hindered by many factors, such as physician inertia and patient intolerance [21, 22]. In particular, physicians might hesitate to fully implement GDMT in malnourished patients – who tend to be older than non-malnourished patients, and to have multiple health complications – despite the demonstrated benefits of GDMT in improving outcomes for these patients [23].
In this study, we sought to investigate whether malnutrition, as assessed with CONUT and GNRI scores, might be associated with under-utilization of GDMT at discharge among hospitalized patients with HF and LVEF <50%. Our aim was to identify the crucial gaps that must be addressed to support GDMT application.
Methods
Setting
This association study was conducted at Beijing Anzhen Hospital, affiliated with Capital Medical University, a teaching hospital specializing in cardiology. The study protocol was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University (approval number 2024171X). The requirement for informed consent was waived because of the study’s retrospective design. All data handling and analysis were performed in accordance with relevant privacy regulations to ensure patient confidentiality.
Study Population
Patients with a mean age of 58.45 ± 14.49 years, who had been hospitalized with a diagnosis of HF in the Heart Failure Center at Beijing Anzhen Hospital, Capital Medical University, between January 1, 2018, and April 30, 2023, were enrolled. Patients who did not meet the inclusion criteria or lacked relevant data were excluded from the analysis (Figure 1).
Data Collection
This study used data from the electronic medical record system, which comprehensively documented patients’ information. Demographic parameters included age, sex, and body mass index (computed as weight in kg divided by the height in m squared). Laboratory assessments encompassed total lymphocyte count, total cholesterol, estimated glomerular filtration rate (eGFR), B-type natriuretic peptide (BNP), N-terminal pro-B-type natriuretic peptide (NT-proBNP), and serum potassium and albumin levels. Medical histories included conditions such as hypertension, diabetes mellitus (DM), coronary artery disease (CAD), atrial fibrillation (AF), and chronic obstructive pulmonary disease (COPD). Additionally, clinical manifestations and vital signs, including LVEF, New York Heart Association Functional Classification (NYHA), and blood pressure at admission were documented. The prescription of GDMT at discharge, comprising β-blockers, RASIs, MRAs, and SGLT2i, and the respective drug doses were also recorded.
Malnutrition Assessment Tools
The CONUT and GNRI indices were used to assess participants’ nutritional status. The CONUT score included serum albumin levels, total cholesterol levels, and total lymphocyte count, and the total score ranged from 0 to 12 [24]. A score of 0–1 was considered to indicate normal nutrition, whereas scores of 2–4, 5–8, and 9–12 reflected mild, moderate, and severe malnutrition, respectively. We categorized scores of 0–1 as normal nutrition and scores ≥2 as malnutrition. The GNRI was calculated with the formula 14.89 × serum albumin (g/dL) + 41.7 × (body weight [kg]/ideal body weight [kg]) [25]. We calculated ideal body weight with the formula: 22 × square of height in meters [26]. A score >98 was considered normal, whereas scores of 92–98, 82–91, and <82 were considered to reflect mild, moderate, and severe malnutrition, respectively. We categorized scores >98 as normal nutrition and scores ≤98 as malnutrition.
Outcome of Interest
The GDMT regimens at discharge were categorized according to therapy type and doses. The triple-therapy regimen was defined as β-blockers, RASIs, and MRAs at any doses. The dose-optimized triple-therapy regimen was defined as β-blockers and RASIs at ≥50% of the target doses, along with the inclusion of MRAs. The dose-optimized double-therapy regimen was specified as any two of the three therapies at optimal doses (β-blockers and RASIs at ≥50% of the target doses, with the use of MRAs). The definitions for target doses were derived from the Chinese Guidelines for Diagnosis and Treatment of Heart Failure 2024 [17]. Because most patients receiving MRAs took 20 mg spironolactone, which did not require up-titration, we did not define the MRA dosage for dose-optimized therapy. Furthermore, SGLT2i were not included in the regimens, because they were not widely used until the year 2021.
Statistical Analysis
Data are presented as numbers (and percentages) for categorical variables and as medians (and first and third quartiles) for continuous variables. Continuous variables were analyzed with the Mann-Whitney U test, whereas categorical variables were compared with Pearson’s chi-squared test.
A multivariable logistic regression model was used to assess the association between GDMT use and nutritional status. Model 1 was adjusted for age (≥60 years median), sex, CAD, DM, AF, CKD, and COPD history. Model 2 was additionally adjusted for LVEF and SBP <100 mmHg. Model 3 was further adjusted for NYHA ≥ III. Statistical significance was defined as a two-sided P < 0.05. All statistical analyses were performed in R software (version 4.2.3).
Results
Among 1312 patients assessed with CONUT, with a mean age of 58.60 ± 14.50 years, 534 (40.7%) were well-nourished, and 778 (59.3%) exhibited varying degrees of malnutrition. The GNRI assessment included 1141 patients with a mean age of 57.86 ± 14.45 years, 962 (84.3%) of whom had normal nutrition and 179 (15.7%) of whom were identified as malnourished. The participants categorized as malnourished were older, had a higher NYHA classification, a higher proportion of eGFR <30 mL/min per 1·73 m2, and higher levels of BNP and NT-proBNP. Moreover, the malnourished patients had greater medical history burdens, including CAD, DM, CKD, AF, and COPD, than the patients with normal nutrition status (Table 1). Nutritional status distribution, assessed with the CONUT and GNRI scores, is shown in Table 2.
Characteristics of Patients, Stratified by Nutrition Status (Assessed with GNRI and CONUT Scores).
| Variables | CONUT score | GNRI score | ||||
|---|---|---|---|---|---|---|
| Normal nutrition (n = 534) | Malnutrition (n = 778) | P value | Normal nutrition (n = 962) | Malnutrition (n = 179) | P value | |
| Demographics | ||||||
| Age, years | 56.0 (43.0, 64.0) | 63.0 (55.0, 71.0) | <0.001 | 58.0 (48.0, 67.0) | 67.0(56.0, 75.0) | <0.001 |
| Male | 400 (74.9) | 583 (74.9) | 0.990 | 739 (76.8) | 125 (69.8) | 0.045 |
| Clinical manifestations and vitals | ||||||
| SBP, mmHg | 119.0 (106.0, 130.0) | 117.0 (104.0, 130.0) | 0.330 | 119.0 (107.0, 130.0) | 115.0 (100.0, 130.0) | 0.007 |
| DBP, mmHg | 73.0 (66.0, 80.0) | 70.0 (64.0, 80.0) | 0.001 | 72.0 (64.0, 80.0) | 70.0 (62.0, 78.5) | 0.007 |
| LVEF, % | 30.0 (25.0, 35.0) | 31.0 (25.0, 38.0) | 0.015 | 30.0 (25.0, 37.0) | 30.0 (25.0, 38.0) | 0.735 |
| New York Heart Association class | <0.001 | <0.001 | ||||
| II | 155 (33.5) | 171 (25.2) | 274 (32.3) | 24 (16.1) | ||
| III | 241 (52.2) | 347 (51.0) | 427 (50.4) | 86 (57.7) | ||
| IV | 66 (14.3) | 162 (23.8) | 146 (17.2) | 39 (26.2) | ||
| Laboratory measures | ||||||
| eGFR, mL/min per 1·73 m2 | ||||||
| <30 | 12 (2.3) | 76 (9.9) | <0.001 | 41 (4.3) | 25 (14.0) | <0.001 |
| Serum potassium, mmol/L | 4.2 (4.0, 4.5) | 4.15 (3.9, 4.4) | 0.003 | 4.2 (3.9, 4.5) | 4.2 (3.9, 4.5) | 0.471 |
| BNP, pg/mL | 369.0 (154.0, 900.0) | 653.0 (282.0, 1535.0) | <0.001 | 444.0 (190.7, 929.2) | 1042.0 (410.0, 2394.0) | <0.001 |
| NT-proBNP, pg/mL | 1235.0 (638.5, 2736.2) | 2858.0 (1032.2, 6072.7) | <0.001 | 1492.5 (628.0, 3602.7) | 3900.0 (1696.0, 8239.0) | <0.001 |
| Medical history | ||||||
| HTN | 272 (50.9) | 425 (54.6) | 0.188 | 519 (53.9) | 78 (43.6) | 0.011 |
| CAD | 215 (40.3) | 432 (55.5) | <0.001 | 452 (47.0) | 93 (52.0) | 0.222 |
| DM | 165 (30.9) | 293 (37.7) | 0.012 | 319 (33.2) | 63 (35.2) | 0.596 |
| CKD | 18 (3.4) | 61 (7.8) | <0.001 | 53 (5.5) | 12 (6.7) | 0.527 |
| AF | 136 (25.5) | 250 (32.1) | 0.009 | 274 (28.5) | 57 (31.8) | 0.363 |
| COPD | 25 (2.60) | 13 (7.26) | 0.001 | 14 (2.62) | 36 (4.63) | 0.062 |
Data are reported as n (%) or median (first quartile, third quartile). AF, Atrial Fibrillation; BNP, B-type Natriuretic Peptide; CAD, Coronary Artery Disease; CKD, Chronic Kidney Disease; CONUT, Controlling Nutritional Status; COPD, Chronic Obstructive Pulmonary Disease; DBP, Diastolic Blood Pressure; DM, Diabetes Mellitus; eGFR, Estimated Glomerular Filtration Rate; GNRI, Geriatric Nutritional Risk Index; HTN, Hypertension; LVEF, Left Ventricular Ejection Fraction; NT-proBNP, N-terminal pro-B-type Natriuretic Peptide; NYHA, New York Heart Association Functional Classification; SBP, Systolic Blood Pressure.
Nutritional Status, Calculated with CONUT and GNRI Scores.
| CONUT | |||||
|---|---|---|---|---|---|
| Normal nutrition (n) | Malnutrition (n) | Unknown (n) | Total (n) | ||
| GNRI | Normal nutrition (n) | 443 | 509 | 10 | 962 |
| Malnutrition (n) | 37 | 138 | 4 | 179 | |
| Unknown (n) | 54 | 131 | 71 | 256 | |
| Total (n) | 534 | 778 | 85 | 1397 | |
CONUT, Controlling Nutritional Status; GNRI, Geriatric Nutritional Risk Index.
Association between Malnutrition and Triple-Therapy Regimen Administration
Compared with patients with normal nutrition, patients with malnutrition, as indicated by the CONUT score, were less likely to be prescribed β-blockers (94.0% vs. 86.8%), RASIs (82.5% vs. 73.6%), and MRAs (62.3% vs. 47.9%) (all P < 0.05). The triple-therapy regimen was administered to 47.9% of malnourished patients and 62.3% of patients with normal nutrition, with an odds ratio (OR) of 0.56 (95% CI 0.44–0.70; P < 0.001). After adjustment for confounding variables, this association remained significant, with an OR of 0.70 (95% CI 0.54–0.91; P = 0.007). Similar patterns were observed after patient stratification by GNRI score (41.3% vs. 57.2%; OR 0.53; 95% CI 0.38–0.73; P < 0.001). This relationship remained significant after adjustment for possible confounders, with an OR of 0.63 (95% CI 0.43–0.92; P = 0.015) (Figures 2 and 3).
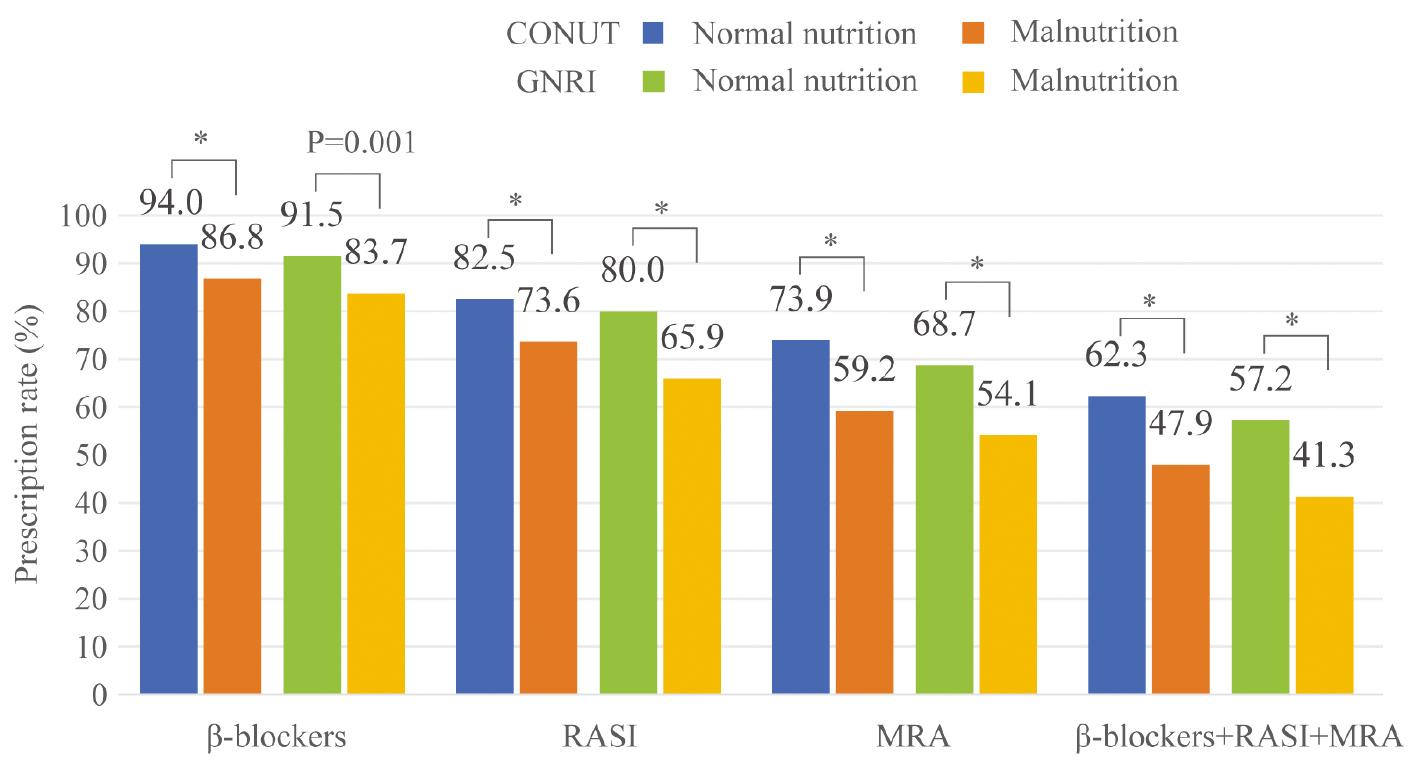
Prescription of Guideline-Directed Medical Therapy in Patients with Heart Failure with Varying Nutritional Status.
*P < 0.001; RASI, Renin-Angiotensin System Inhibitors; MRA, Aldosterone Antagonists; CONUT, Controlling Nutritional Status; GNRI, Geriatric Nutritional Risk Index.
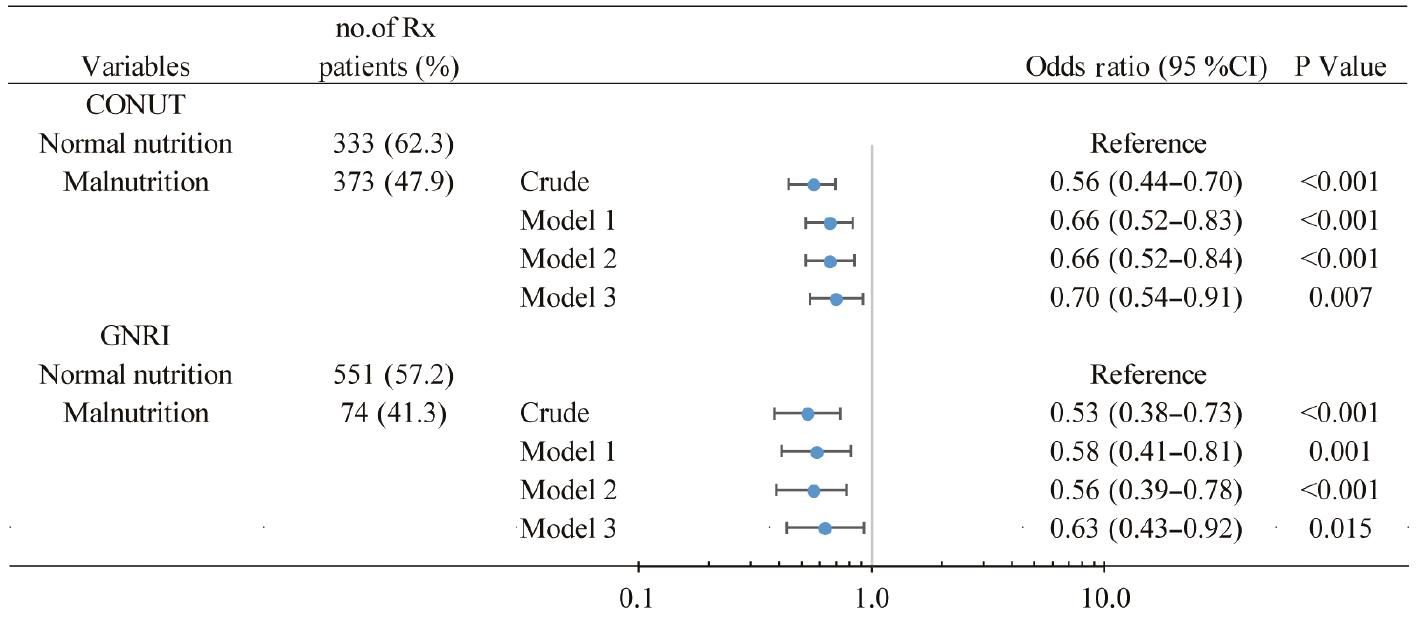
Association between Malnutrition and Administration of a Triple-Therapy Regimen.
Model 1: adjusted for age (≥60 years median), sex, coronary artery disease, diabetes mellitus, atrial fibrillation, chronic kidney disease, and chronic obstructive pulmonary disease history. Model 2: adjusted for variables in model 1 plus left ventricular ejection fraction and systolic blood pressure <100 mmHg. Model 3: adjusted for variables in model 2 plus New York Heart Association class ≥III. CONUT, Controlling Nutritional Status; GNRI, Geriatric Nutritional Risk Index; Rx, prescription.
Association between Malnutrition and Administration of a Dose-Optimized Triple-Therapy Regimen
Malnourished patients, identified according to CONUT scores, had a significantly lower likelihood of receiving the dose-optimized triple-therapy regimen (4.8% vs. 9.7%; OR 0.46; 95% CI 0.30–0.72; P < 0.001) than patients with normal nutrition. This trend was consistent after confounders were accounted for (OR 0.59; 95% CI 0.36–0.97; P = 0.039). Similarly, the GNRI score showed a lower prescription rate for malnourished individuals (2.2% vs. 8.2%; OR 0.26; 95% CI 0.09–0.71; P = 0.009), and an OR of 0.35 (95% CI 0.11–1.17; P = 0.088) after potential confounders were controlled for (Figure 4).
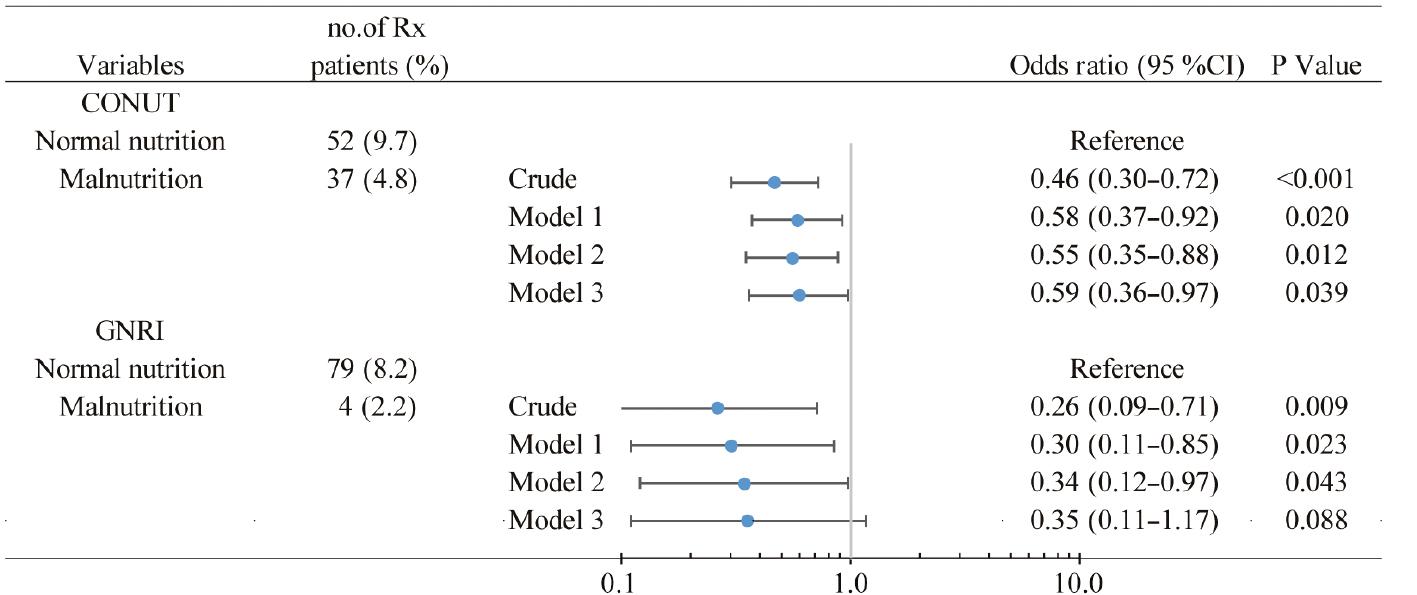
Association between Malnutrition and Administration of a Dose-Optimized Triple-Therapy Regimen.
Model 1: adjusted for age (≥60 years median), sex, coronary artery disease, diabetes mellitus, atrial fibrillation, chronic kidney disease, and chronic obstructive pulmonary disease history. Model 2: adjusted for variables in model 1 plus left ventricular ejection fraction and systolic blood pressure <100 mmHg. Model 3: adjusted for variables in model 2 plus New York Heart Association class ≥III. CONUT, Controlling Nutritional Status; GNRI, Geriatric Nutritional Risk Index; Rx, prescription.
Association between Malnutrition and Administration of a Dose-Optimized Double-Therapy Regimen
Patients with malnutrition, as identified according to CONUT scores, were less likely to be prescribed a dose-optimized double-therapy regimen than patients with normal nutritional status (24.3% vs. 37.8%; OR 0.53; 95% CI 0.42–0.67; P < 0.001). This relationship remained significant after adjustment for confounding factors (OR 0.65; 95% CI 0.50–0.86; P = 0.002). The GNRI score showed a similar trend, with a lower prescription rate for malnourished than non-malnourished individuals (17.3% vs. 33.5%; OR 0.42; 95% CI 0.28–0.63; P < 0.001); this finding was also robust to adjustment (OR 0.56; 95% CI 0.35–0.89; P = 0.015) (Figure 5).
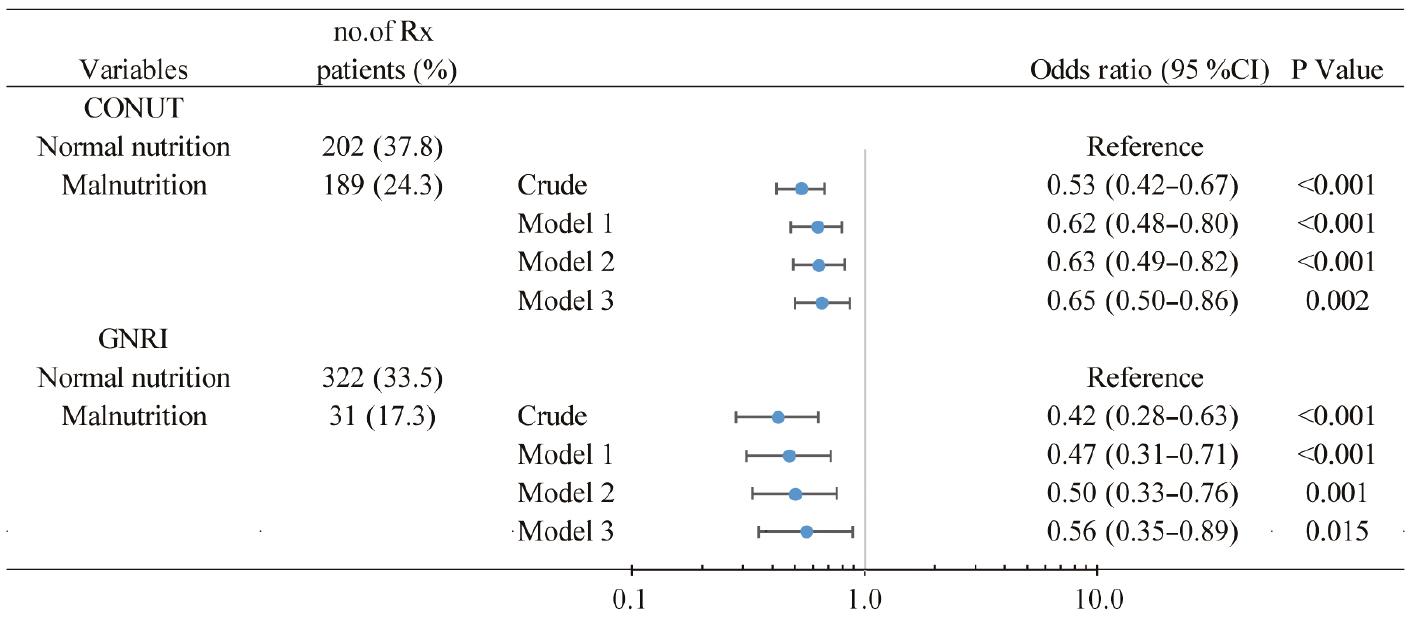
Association between Malnutrition and Administration of a Dose-Optimized Double-Therapy Regimen.
Model 1: adjusted for age (≥60 years median), sex, coronary artery disease, diabetes mellitus, atrial fibrillation, chronic kidney disease, and chronic obstructive pulmonary disease history. Model 2: adjusted for variables in model 1 plus left ventricular ejection fraction and systolic pressure <100 mmHg. Model 3: adjusted for variables in model 2 plus New York Heart Association class ≥III. CONUT, Controlling Nutritional Status; GNRI, Geriatric Nutritional Risk Index; Rx, prescription.
Discussion
Among 1397 hospitalized patients with HF and LVEF <50%, we observed a significant association between malnutrition and lower prescription rates of the triple-therapy regimen, dose-optimized triple-therapy regimen, and dose-optimized double-therapy regimen than observed among patients with normal nutritional status. This association retained significance after adjustment for confounding factors.
No consensus exists regarding the optimal nutritional assessment tool for patients with HF [5, 6]. Body composition analysis methods, such as dual-energy X-ray absorptiometry and magnetic resonance imaging, may be inaccurate in the presence of excess body water or impractical for routine use. Single biomarkers, including albumin and prealbumin, can be influenced by comorbidities [27]. In contrast, multidimensional assessment tools, such as the CONUT index and the GNRI, assess multiple domains of nutritional status, are easy to measure, and have been studied in HF, thus showing promise for clinical use [7–10]. The present study, using both CONUT and GNRI scores, revealed a notable discrepancy in the malnutrition prevalence rates among patients with HF (60% according to the CONUT score and 16% according to the GNRI score). This finding underscores the complexity inherent in accurate assessment of nutritional status in patients with HF. The GNRI’s reliance on body weight might lead to overestimation of nutritional status, particularly in patients with fluid retention – a frequent comorbidity in HF. In contrast, the GNRI might not fully capture malnutrition in overweight patients, thus posing a risk of underdiagnosis [9]. In contrast, the CONUT score, which is based on serum parameters reflecting protein, lipid metabolism, and immune function, might not accurately represent the nutritional status of patients with HF who are prescribed lipid-lowering drugs [28]. Our results are supported by a recent review indicating a broad spectrum of malnutrition prevalence – ranging from 16% to 62% in clinically stable patients and reaching as high as 75%–90% in patients with advanced and decompensated HF [10]. This variability underscores the effects of disease stage and the choice of the nutritional assessment tool on the reported rates. Because of the lack of consensus for choosing nutrition assessment tools and routines for patients with HF, the inconsistencies in the results obtained with different tools warrant further research, to provide insight and guidance for selection of the most appropriate nutrition assessment tool [5, 29–31].
The insufficient GDMT in patients with HF and malnutrition is potentially explained by the following: malnutrition might lead to changes in body function and metabolism, in the presence of various diseases, thus resulting in varying patient responses or tolerances to certain medications [32–35]. In addition, malnutrition might increase the risk of adverse reactions or complications to medication in patients with HF. We observed that individuals with rather than without malnutrition tended to be older, and to have more comorbidities and poorer renal function. Therefore, physicians might be more cautious in prescribing to patients with malnutrition, such as by decreasing the medication dosage or selecting the medication type to avoid adverse events [36, 37]. In addition, malnourished patients often exhibit symptoms such as weight loss, sarcopenia, and fatigue [38], which can overlap with those of HF, thereby leading to potential misdiagnosis or oversight, and affecting the timely implementation of GDMT. Finally, malnutrition leads to overall poor health status in patients [38], and consequently might affect their compliance with medication treatment and result in lower actual GDMT use.
Traditionally, HF treatment has focused primarily on pharmacological management, whereas nutritional status is often overlooked. Our study identifies notable gaps in the application of GDMT for malnourished patients. We believe that this study has three important clinical implications for improving the quality of care for patients with HF. First, malnutrition is prevalent among hospitalized patients with HF and should be identified through screening, given its well-established association with adverse outcomes and poor prognosis [1, 9], partially as a result of inadequate therapy. Second, malnutrition is a major barrier to GDMT provision. Whether incorporating multidisciplinary teams – including cardiologists, dietitians, nurses, and other healthcare professionals – might enhance GDMT use in malnourished patients with HF remains unclear. Finally, whether interventions aimed at optimizing GDMT would improve prognosis for patients with HF and malnutrition is uncertain.
Strengths and Limitations
This study investigated the association between malnutrition and the prescription patterns for GDMT regimens, considering both therapy types and doses, among hospitalized patients with HF and an LVEF below 50% at discharge. The study’s innovative use of both the CONUT and GNRI scores to assess malnutrition provides a nuanced understanding of nutritional status in patients with HF. Furthermore, the comprehensive analysis of both therapy types and doses in relation to malnutrition is a notable strength of this research, which provides detailed insights into the effects of nutritional status on GDMT prescription patterns. The focus on hospitalized patients with HF and an LVEF below 50% is a notable study strength, because this vulnerable population is often overlooked in nutritional studies. Our research advances existing knowledge in this field by potentially enhancing the medical community’s understanding of the importance of nutritional factors in HF management.
Nevertheless, the limitations of the retrospective observational design might have introduced bias and confounding from unmeasured or unidentified variables, even after adjustment for relevant clinical characteristics. Moreover, this study was conducted at a single center, and our research team collectively shared similar experiences in GDMT administration. To some extent, this aspect might have mitigated potential biases associated with variations in GDMT prescription strategies across healthcare providers. In addition, HF attributed to specific etiologies, such as myocardial amyloidosis and Fabry’s disease, was not definitively excluded; however, given that this group of affected individuals is relatively small, it would be expected to have had negligible effects on the results.



