| What is the clinical question being addressed? |
| How can acute pulmonary embolism be treated more safely and effectively? |
| What is the main finding? |
| Treating intermediate-risk patients with acute pulmonary embolism with a large-diameter thrombus aspiration and mechanical thrombectomy device may have acceptable clinical benefits. |
Globally, acute pulmonary embolism (PE) is the third leading cause of cardiovascular-related deaths after myocardial infarction and stroke [1]. Several studies have reported increased annual incidence of PE and consequently social medical burden [2]. Determining how to treat acute PE more safely and effectively, while decreasing long-term complications, remains an important issue in medicine.
The aim of this study was to report the results of a single-arm clinical trial in intermediate-risk patients with acute PE treated with a device for large-diameter thrombus aspiration (16 F or greater) and mechanical thrombectomy.
This prospective, single-arm clinical trial was conducted between March 2022 and January 2023 at Xiamen Cardiovascular Hospital, Xiamen University, China. The clinical trial was registered at http://clinicaltrials.gov (Clinical Trial ID: NCT 05443919). A comprehensive description of the study design, data collection methods, inclusion and exclusion criteria, and outcome measure can be found at https://clinicaltrials.gov/study/NCT05443919. PE severity was evaluated in accordance with the scoring criteria outlined in the ESC acute PE guidelines [2], and patients with intermediate-risk acute PE were included in the study.
All 13 patients (average age: 66.85 y) were treated with the device consisting of thrombus aspiration and pulmonary artery retrieval systems (Figure 1). Most patients were women (n = 8, 61.54%). The median PE severity index score at admission was 102 (interquartile range, IQR: 73–106). Ten patients presented with bilateral PE. Table 1 provides a detailed summary of the perioperative adjuvant drugs used, as well as the blood biomarkers and cardiac ultrasound markers observed pre- and post-surgery. Femoral access was chosen for all enrolled cases. The median procedure time was 65 min (IQR: 59–69). The median length of intensive care unit stay was 16 h (IQR: 13–22.32), and the median length of hospital stay was 7 d (IQR: 6–14). No technical complications were observed in any enrolled patients. The average blood loss was 157.69 ± 73.16 mL.
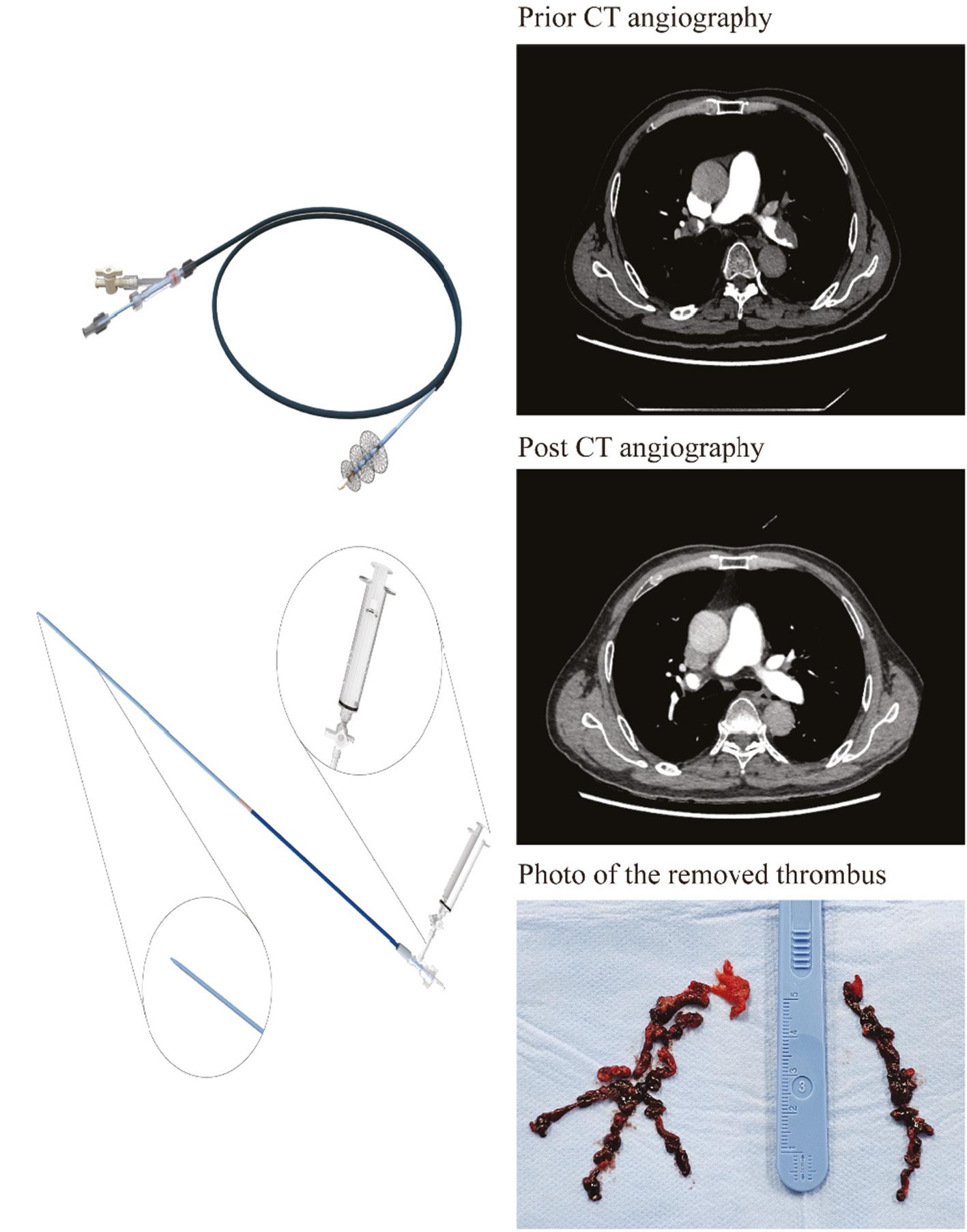
Diagram of the Thrombus Aspiration and Mechanical Thrombectomy Device.
The system consists of thrombus aspiration and retrieval systems. The thrombus aspiration system consists of an aspiration guide catheter, a dilator, and an aspirator. The thrombus retrieval unit consists of three self-expanding nickel-titanium alloy circular mesh disks with differing diameters and a push rod.
Relevant Characteristics of the Enrolled Patients.
| ID | Preoperative anticoagulant drugs | Intraoperative anticoagulant drugs | Postoperative anticoagulant drugs | Follow-up anticoagulant drugs | Preoperative BNP (pg/mL) | Preoperative TNT (ng/L) | Postoperative 48 h BNP (pg/mL) | Postoperative 48 h TNT (ng/L) | Preoperative right ventricular end-diastolic internal diameter (mm) | Right ventricular end-diastolic internal diameter at 14 d postoperatively (mm) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Enoxaparin sodium salt 8000 IU q12 h | Heparin sodium 25,000 IU | Enoxaparin sodium salt 8000 IU q12 h | Rivaroxaban 15 mg bid | 2223 | <40 | 1597 | 74.7 | 50.8 | 41.4 |
| 2 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 25,000 IU | Enoxaparin sodium salt 4000 IU q12 h | Rivaroxaban 15 mg bid | 1233 | 56.3 | 381 | 47.8 | 43.3 | 28.2 |
| 3 | Enoxaparin sodium salt 4000 IU q12 h | Heparin sodium 24,000 IU | Enoxaparin sodium salt 4000 IU q12 h | Rivaroxaban 15 mg bid | 1121 | 27.8 | 316 | 26 | 41.5 | 19.7 |
| 4 | Enoxaparin sodium salt 4000 IU q12 h | Heparin sodium 25,000 IU | Enoxaparin sodium salt 4000 IU q12 h | Rivaroxaban 15 mg bid | 2994 | 66.1 | 1451 | 142 | 44.7 | 38.8 |
| 5 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 25,000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 813.51 | 28.22 | 1077 | 67.5 | 37.2 | 34.4 |
| 6 | Enoxaparin sodium salt 4000 IU q12 h | Heparin sodium 25,000 IU | Enoxaparin sodium salt 4000 IU q12 h | Rivaroxaban 15 mg bid | 5116 | 50.7 | 491 | 26 | 50.7 | 36.4 |
| 7 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 6000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 396 | 8.73 | 335 | 37.3 | 26 | 28.4 |
| 8 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 6000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 227 | 23.1 | 383 | 31.9 | 39.6 | 33.8 |
| 9 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 25,000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 1113 | 24.8 | 247 | 14.4 | 34 | 35.5 |
| 10 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 6000 IU | Enoxaparin sodium salt 4000 IU q12 h | Rivaroxaban 15 mg bid | 4824 | 11.2 | 532 | 15.3 | 41.3 | 25.1 |
| 11 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 7000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 1262.25 | 36.56 | 561 | 37.4 | 43.1 | 26.1 |
| 12 | Enoxaparin sodium salt 6000 IU q12 h | Heparin sodium 6000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 3310 | 33 | 308 | 22.9 | 62 | 35.9 |
| 13 | Low-molecular heparin sodium 4500 IU q12 h | Heparin sodium 25,000 IU | Low-molecular heparin sodium 4500 IU q12 h | Rivaroxaban 15 mg bid | 1182 | 10.2 | 436 | 15.5 | 45.8 | 37.4 |
| ID | Preoperative left ventricular end-diastolic internal diameter (mm) | Left ventricular end-diastolic internal diameter at 14 d postoperatively (mm) | Preoperative RV/LV | RV/LV at 14 d postoperatively | Preoperative LVEF (%) | LVEF (%) at 48 h postoperatively | LVEF (%) at 14 d postoperatively | Preoperative pulmonary artery systolic pressure (mmHg) | Pulmonary artery systolic pressure at 48 h postoperatively (mmHg) | Pulmonary artery systolic pressure at 14 d postoperatively (mmHg) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 32.5 | 46.7 | 1.56 | 0.89 | 75 | 60 | 67 | 60 | 61 | 53 |
| 2 | 33.1 | 41.5 | 1.31 | 0.68 | 50 | 72 | 64 | 71 | 72 | 41 |
| 3 | 40.1 | 34.5 | 1.03 | 0.57 | 60 | 69 | 72 | 68 | 40 | 31 |
| 4 | 32.3 | 30.9 | 1.38 | 1.26 | 65 | 63 | 62 | 41 | 40 | 24 |
| 5 | 23.7 | 35.5 | 1.57 | 0.97 | 58 | 70 | 76 | 52 | 51 | 38 |
| 6 | 37.4 | 52 | 1.36 | 0.7 | 60 | 61 | 66 | 61 | 28 | 32 |
| 7 | 26.6 | 29 | 0.98 | 0.98 | 62 | 74 | 75 | 45 | 43 | 33 |
| 8 | 38.7 | 34.5 | 1.02 | 0.98 | 56 | 75 | 56 | 39 | 33 | 20 |
| 9 | 31.6 | 46.8 | 1.08 | 0.76 | 60 | 70 | 75 | 35 | 32 | 31 |
| 10 | 26 | 31.5 | 1.59 | 0.8 | 75 | 56 | 65 | 85 | 38 | 29 |
| 11 | 31 | 29.7 | 1.39 | 0.88 | 67 | 64 | 68 | 59 | 27 | 27 |
| 12 | 28.3 | 50.9 | 2.19 | 0.71 | 68 | 74 | 73 | 28 | 25 | 31 |
| 13 | 30.5 | 39.6 | 1.5 | 0.94 | 64 | 63 | 63 | 42 | 26 | 20 |
For reasons of space, it has been necessary to split the table over 2 pages. The second set of characteristics for patient IDs 1-13 can be found on the second page of the table.
The primary outcomes included the change in the site-reported right ventricular/left ventricular ratio (RV/LV ratio) from baseline to 14 d after surgery, as well as the rate of major adverse events (MAEs) within 48 h after surgery. The average RV/LV ratio decreased from 1.38 ± 0.33 to 0.86 ± 0.18 (P = 0.0003). After the intervention, the average RV/LV ratio decreased by 0.53. MAEs occurred in one patient, who was evaluated by an additional experienced researcher and was confirmed to meet the study criteria. During the operation, the patient’s condition worsened, and the right ventricular function decompensated. Extracorporeal membrane oxygenation was used as an auxiliary treatment after interventional thrombectomy. The symptoms improved after postoperative treatment, and the patient was healthy at discharge. After evaluation by the hospital review committee, the MAE was found to be unrelated to the experimental device. In this patient, the RV/LV ratio changed from 1.57 at baseline to 0.97 at 14 d after surgery. The pulmonary artery systolic pressure changed from 52 to 38 mmHg, and the ejection fraction (EF%) changed from 58% to 76%.
Other important clinical indicators also showed significant improvements after surgery. The average postoperative pulmonary artery systolic pressure decreased significantly with respect to preoperative values (48 h: 53.42 ± 4.48 mmHg vs. 39.69 ± 3.96 mmHg, P = 0.009; 14 d: 53.42 ± 4.48 mmHg vs. 30.69 ± 2.62 mmHg, P = 0.0006). The average postoperative PaO2 increased significantly above the preoperative value (48 h: 69.32 ± 21.96 mmHg vs. 97.31 ± 32.34 mmHg, P = 0.015). Ultrasound examination of cardiac structural indicators was performed 14 d after surgery. The average left ventricular end-diastolic dimension changed from 31.68 ± 4.92 to 38.70 ± 8.15 mm (P = 0.013), and the average right ventricular end-diastolic dimension changed from 43.08 ± 8.72 to 32.39 ± 6.32 mm (P = 0.001).
Before and after the procedure, patients underwent CT angiography of the pulmonary artery for assessment of thrombus location and extent. A marked decrease in the burden of pulmonary vascular thrombus was observed, as evaluated with the modified Miller score based on CT angiography (pre: 19.00 ± 4.02, post: 7.92 ± 3.68, P = 0.0004).
No device-related heart injury, clinical death, or symptomatic pulmonary recurrence occurred in any patients within 30 d of surgery. Moreover, none of the 13 patients experienced MAEs during the 30-d postoperative follow-up period.
Current guidelines for the diagnosis and treatment of PE recommend anticoagulation for the treatment PE [2]. However, rapid haemodynamic changes and cardiac decompensation have prompted clinicians to focus on early thrombus clearance via systemic thrombolysis, percutaneous catheter-directed treatment, or surgical thrombectomy. Previous clinical studies have indicated that the therapeutic effects of systemic thrombolytic therapy for intermediate-risk PE are offset by bleeding risk, thus providing a neutral net clinical benefit. Surgical thrombectomy also has the disadvantages of heavy trauma and high surgical risk [3, 4]. Catheter-based embolectomy has attracted increasing attention from clinicians, owing to its advantages including minimal invasiveness and ability to quickly alleviate the thrombus burden [5]. In comparison to commercially available devices, such as the FlowTriever system (Inari Medical, Irvine, California, USA), the Indigo Thrombectomy System (Penumbra, Alameda, California, USA), and the AngioVac cannula (AngioDynamics, Latham, New York), our device has been demonstrated to result in less bleeding while ensuring efficient thrombus extraction [4]. As a single-arm clinical trial of a device, this study included only 13 patients for clinical safety reasons. Because of the lack of a control group, this study was unable to compare the advantages and disadvantages of this device versus other treatment options. Therefore, caution is necessary in interpreting our conclusions. Although the safety and efficacy of this device require validation in larger clinical studies, catheter-based embolectomy was demonstrated to be an effective and safe treatment for patients with high-risk PE. This treatment approach merits further investigation.

