Abbreviations: KBI, Kissing balloon inflation; POT, Proximal optimization technique; DES, Drug-eluting stents; MI, Myocardial infarction; icECG, Intracoronary electrocardiography; QCA, Quantitative coronary angiography.
Introduction
Revascularization of bifurcation lesions remains challenging in terms of both procedural success and long-term cardiovascular outcomes [1]. In recent years, substantial improvements have been made in the percutaneous treatment of coronary bifurcation stenoses. The introduction of kissing balloon inflation (KBI) and proximal optimization technique (POT) have significantly improved patient clinical outcomes [2].
Furthermore, the development of newer-generation drug-eluting stents (DES) has significantly decreased the rates of restenosis at the bifurcation region and, to a lesser degree, the rates of “hard” endpoints, such as all-cause death, cardiovascular death and spontaneous non-fatal myocardial infarction (MI). Compared with bare metal stents and first-generation DES, second-generation DES have been associated with lower risks of in-stent restenosis, stent thrombosis and MI [3, 4]. The most recent data have indicated relatively low rates of stent thrombosis (1.0%), target lesion failure (5.1%), cardiac death (1.8%) and all-cause death (1.8%) 1 year after bifurcation percutaneous coronary intervention (PCI) [5]. Previously, we hypothesized that these improved PCI results are partially associated with improvements in stent technology (platforms, drugs and drug carriers) [6]. With technological advances, interventional technical performance has improved, as clearly indicated by changes in the recommendations of the European Bifurcation Club over time [7, 8].
In addition, using intracoronary electrocardiography (icECG) guidance may be useful in predicting the functional significance of side branch (SB) stenosis after bifurcation stenting [9]. We have demonstrated that icECG can predict post-procedural increases in troponins after coronary bifurcation lesion stenting [10]. Moreover, icECG has a unique ability to locate possible sources of post-PCI rise in troponin, by detecting zones with ST-segment elevation on icECG at the end of the procedure. However, to our knowledge, the influence of icECG-guidance for bifurcation PCI on all-cause and cardiovascular mortality had not previously been analyzed.
The aim of the current analysis was to evaluate the influence of an icECG-guided revascularization strategy on the clinical outcomes of patients after coronary bifurcation stenting.
Methods
Patient selection
From July 2017 to December 2020, patients with coronary bifurcation stenoses referred for percutaneous revascularization were included in a prospective registry. In the current analysis, patients with acute coronary syndrome, left bundle branch block, left main stenosis, ST-elevation MI, hemodynamic instability and life expectancy <12 months were excluded. In addition, patients with in-hospital events were excluded. No other restrictions for patient inclusion were used.
Procedure
Patients were randomly assigned to receive one of two procedure types:
icECG-guided revascularization
The icECG-guided revascularization strategy was previously demonstrated in the FIESTA study [11]. After obtaining optimal projection, two standard workhorse PCI wires (Runthrough, Terumo, Japan; Sion Blue, Asahi, Japan; or BMW Universal II, Abbot Vascular, USA) were introduced into the main branch and SBs. The tips of the wires were ascertained to be freely moving, without any wedging into the myocardium. The outer ends of the wires were connected to unipolar ECG leads with alligator clips (Supplementary Video 1). The baseline recordings were made before the insertion of any other devices into the target artery. In some cases of interaction signals due to contacts between the wires, a pre-dilatation balloon was inserted to the position of the tip of the guiding catheter. In rare cases, the balloon was inserted into the tip of the bifurcation carina to divide crisscrossing wires. After the baseline signals were obtained, predilatation to the main branch, SB or both, was performed at the operator’s discretion. After stent implantation, a second recording was made from both wires. Proximal optimization technique was recommended, but the decision was left to the surgeon. In cases of ST-segment elevation on icECG from an SB wire, balloon dilatation of the SB ostium (KBI was highly recommended) was performed. Finally, after intracoronary nitroglycerine administration (100–200 mcg), another recording of icECG was made. If the ST-segment elevation persisted, the wire from the SB was slowly pulled back to the ostium of the side vessel, and the ECG was recorded through the whole vessel length. If ST-segment elevation was present only in the distal half of the vessel length, it was considered a result of distal embolization. If ST-segment elevation was also present in the proximal half of the vessel, it was considered a result of diffuse microcirculatory spasm in addition to microembolization. In both cases, patients received heparin and nitroglycerin intravenous infusions for the next 18–24 h. If there was any sign of vessel dissection, more than type B and icECG changes, a stent was implanted into the SB. After POT, if no icECG changes were observed, SBs were balloon dilated only in cases of ≥90% SB ostial diameter stenosis or TIMI flow <2. A case example is illustrated in Figure 1. A detailed description of the icECG analysis is included in the Supplementary Material.
Provisional T-stenting strategy
The provisional T-stenting strategy, also referred to as standard of care (SOC), followed the most recent European Bifurcation Club recommendations [8]. A detailed description of the provisional T-stenting technique is included in the Supplementary Material.
Angiographic analysis
Dedicated bifurcation quantitative coronary angiography (QCA) analysis was performed according to the recommendations of the consensus on QCA methods for bifurcation lesions using General Electric QCA software and MicroDicom QCA software [12]. Bifurcation lesions were classified according to the Medina classification [13]. True bifurcation lesions were defined by a visual percentage diameter stenosis (%DS) >50% at the SB with lesions at the proximal or distal main branch. The minimal luminal diameter, reference vessel diameter and %DS were measured for every segment of the bifurcation (i.e., proximal and distal MV and SB) pre- and post-intervention. Lesion length was measured from the proximal main vessel to the distal main branch (i.e., the beginning and end points where the stent would potentially be implanted). SB lesion length was measured from the ostium to the first normal-appearing part of the vessel. All analyses were performed by two investigators (N.M. and P.P.). In cases of disagreement, a consensus was reached according to additional analysis by the first author (D.V.).
Definition of endpoints
All patients were followed up by telephone contact and/or clinical visit at 30 days, and vital status was subsequently monitored monthly through the National Insurance Institute. Clinical outcomes were defined according to the current recommendations [12]. Cardiovascular death was defined as death due to a clearly determined cardiac origin or an unknown reason. MI after hospitalization was diagnosed according to the fourth definition of MI [14]. Major adverse cardiac events were a combination of cardiovascular death and MIs. Patient oriented cardiac events were a summary of major adverse cardiac events and target vessel failure rates. Target vessel failure was defined as any intervention in the target vessel.
Statistical analysis
All data are presented as means ± standard deviation. Differences between groups were examined with paired or unpaired t-tests as appropriate, for data with a normal distribution. If the data distribution was not normal, Mann-Whitney U-tests were performed. Analysis of variance was used for multiple comparisons of data when parameters were distributed normally. Otherwise, Kruskal-Wallis test was performed. Cox–multiple regression analysis with a backward elimination process was used to identify predictors of death and cardiovascular death. All univariate predictors with P < 0.1 were included in a multivariate model. Chi-square tests were applied for qualitative data. A P-value < 0.05, with 85% power, was accepted as significant. All statistical calculations were performed in the SPSS package, version 25, USA. The study was approved by the local ethics committee.
RESULTS
Demographic and procedural characteristics
Among 1042 patients who underwent coronary bifurcation stenting, 768 were included in the current analysis. A total of 349 patients underwent an icECG-guided strategy, and 419 underwent SOC (Figure 2). The two groups had similar risk profiles (Table 1). The icECG-guided group, compared with the SOC group, had significantly more smokers (54% vs. 34%, P 0.001) and more patients with previous MIs, but a lower rate of chronic kidney disease (GFR 78 ± 26 mL/kg/min vs. 71 ± 23 mL/kg/min, respectively, P < 0.001). Furthermore, the rate of atrial fibrillation was slightly lower in the icECG-guided group than the SOC group (18% vs. 24%, respectively, P = 0.024). Interestingly, the patients with icECG guidance received statins less frequently (93% vs. 98%, P = 0.001), and clopidogrel more frequently (88% vs. 82%, P = 0.019), than those in the SOC group.
Demographic Characteristics.
| Patient characteristics | icECG group | SOC group | P-value |
|---|---|---|---|
| Age (years) | 66 ± 10 | 68 ± 10 | 0.001 |
| Male sex, % (n) | 31% (108) | 31% (130) | 0.998 |
| Hypertension, % (n) | 98% (342) | 99% (414) | 1.0 |
| Hyperlipidemia, % (n) | 93% (324) | 94% (393) | 0.932 |
| Smoking, % (n) | 54% (188) | 34% (142) | 0.001 |
| Diabetes, % (n) | 37% (129) | 42% (175) | 0.231 |
| Renal failure, % (n) | 25% (87) | 37% (142) | 0.001 |
| Previous myocardial infarction, %(n) | 29% (101) | 23% (96) | 0.039 |
| Previous PCI, % (n) | 48% (167) | 49% (205) | 0.978 |
| Atrial fibrillation, % (n) | 18% (62) | 24% (100) | 0.024 |
| Peripheral artery disease, % (n) | 9% (31) | 11% (46) | 0.763 |
| Previous stroke or TIA, % (n) | 14% (49) | 18% (75) | 0.121 |
| Carotid artery disease, % (n) | 5% (17) | 8% (33) | 0.781 |
| COPD, % (n) | 12% (42) | 14% (58) | 0.675 |
| Cancer, % (n) | 7% (24) | 6% (25) | 0.854 |
| Aspirin treatment, % (n) | 91% (318) | 90.5 (381) | 0.882 |
| Clopidogrel treatment, % (n) | 80.2% (280) | 80% (336) | 0.941 |
| Prasugrel treatment, % (n) | 12.8% (44.7) | 12 (50) | 0.792 |
| Ticagrelor treatment, % (n) | 7% (24) | 8% (33.5) | 0.602 |
Renal failure is defined as a glomerular filtration rate, calculated according to the Cockcroft–Gault formula, <60 mL/min.
Abbreviations: TIA, transient ischemic attack; COPD, chronic obstructive pulmonary disease.
We specifically evaluated the differences in patient symptoms, left ventricular function and mitral regurgitation between groups. No difference was observed regarding angina (median CCS class 3, IQR, 2–4), but more patients had dyspnea in the SOC group (NYHA class II–III: 13% vs. 22%, icECG vs. SOC, P = 0.001). Patients with icECG guidance had a higher left ventricular ejection fraction (57 ± 8% vs. 55 ± 10%, P = 0.009) and lower rates of significant mitral regurgitation (14% vs. 23%, P = 0.001).
The procedural and angiographic data are presented in Tables 2 and 3. The icECG-guided group included significantly fewer true bifurcation lesions (50% vs. 63%, P = 0.001), thus probably explaining the higher prevalence of predilatation in the SOC group than the icECG group (83% vs. 76%, respectively, P = 0.034). Although icECG-guidance resulted in a slightly longer procedural time, the X-ray time and the amount of contrast used did not exceed those in the SOC group. The icECG guidance strategy, compared with the PTS strategy, resulted in almost 50% less SB stenting and significantly fewer stents used per procedure overall (1.63 ± 0.81 vs. 1.86 ± 1.00, P = 0.001). Consequently, the total stent length was significantly shorter in the icECG-guided group, regardless of the similar lesion length in the two groups. Importantly, the icECG guidance strategy resulted in higher final SB ostial stenosis (Table 3). The frequency of final SB ostial stenosis >50% was 68% in the icECG group and 38% in the PTS group (P = 0.001). Interestingly, this frequency was not associated with less KBI (Table 2) or any SB post dilatation with a balloon (icECG 53% vs. 50% PTS, P = 0.638).
Procedural Characteristics.
| Procedure details | icECG group | SOC group | P-value |
|---|---|---|---|
| LAD/diagonal, % | 75% | 66% | 0.001 |
| LCX/marginal, % | 20% | 18% | |
| RCA PD/PL, % | 6% | 16% | |
| Multivessel disease, % | 61% | 69% | 0.021 |
| SYNTAX score (mean ± SD) | 11 ± 5 | 12 ± 6 | 0.065 |
| Stent diameter, mm (mean ± ds) | 3.01 ± 0.35 | 3.16 ± 0.36 | 0.001 |
| Stent length, mm (mean ± SD) | 38 ± 22 | 49 ± 27 | 0.001 |
| Kissing balloon inflation,% | 32% | 30% | 0.638 |
| Main vessel POT, % | 67% | 63% | 0.249 |
| Second stent, % | 12% | 21% | 0.001 |
| Guidewires used, % | 71% | 72% | 0.509 |
| Microcatheters used, % | 15% | 17% | 0.311 |
| Procedural time, min (mean ± SD) | 90 ± 34 | 82 ± 38 | 0.003 |
| Scopic time, min (mean ± SD) | 22.5 ± 10.8 | 21.0 ± 8.8 | 0.177 |
| Contrast, mL (mean ± SD) | 194 ± 105 | 204 ± 120 | 0.265 |
Abbreviations: LAD, left anterior descending artery; LCX, left circumflex artery; RCA, right coronary artery; PD, posterior descending artery; PL, postero-lateral branch artery.
Angiographic Results.
| icECG | SOC | P-value | |
|---|---|---|---|
| MV RVD, mm | 3.38 ± 0.39 | 3.30 ± 0.44 | 0.014 |
| MV %DS, % | 55 ± 30 | 56 ± 30 | 0.670 |
| MV %DS final, % | 2 ± 8 | 1 ± 4 | 0.001 |
| MB RVD, mm | 3.09 ± 1.82 | 3.13 ± 1.49 | 0.897 |
| MB %DS, % | 69 ± 23 | 65 ± 24 | 0.009 |
| MB %DS final, % | 2 ± 4 | 2 ± 4 | 0.423 |
| SB RVD, mm | 2.33 ± 0.36 | 2.39 ± 0.98 | 0.332 |
| SB %DS, % | 47 ± 30 | 50 ± 31 | 0.164 |
| SB %DS, post stenting, % | 61 ± 30 | 37 ± 32 | 0.001 |
| SB %DS, final, % | 43 ± 31 | 20 ± 26 | 0.001 |
| Total lesion length, mm | 35 ± 19 | 36 ± 19 | 0.832 |
Abbreviations: icECG, intracoronary electrocardiography; SOC, standard of care; MV, main vessel before side branch, MB, main branch, after side branch, SB, side branch, %DS, percentage diameter stenosis, RVD, reference vessel diameter.
Periprocedural troponin dynamics
We specifically explored the periprocedural troponin dynamics. Twenty-eight percent of the patients in the icECG-guided group had a post-PCI troponin increase ≥5 × the UNL, as compared with 37.5% of the patients in the SOC group (P = 0.009). In addition, the troponin increase post-PCI was significantly lower (77%) in the icECG group than the SOC group (82%, P = 0.046). No difference was observed in the frequency of periprocedural MIs between groups (P = 0.582; Supplementary Table 1).
Clinical outcomes
The median follow-up time was 56 months [IQR 38–60 months]. The rate of all-cause mortality was 23.2% (178/768), and that of cardiovascular mortality was 15.9% (122/768). The type of interventional strategy significantly affected survival: patients with icECG guidance had significantly lower all-cause mortality (20.3% vs. 25.5%, log-rank P = 0.006) and cardiovascular mortality (12.6% vs. 18.6% log-rank P = 0.004, for icECG vs. SOC, respectively; Figure 3).
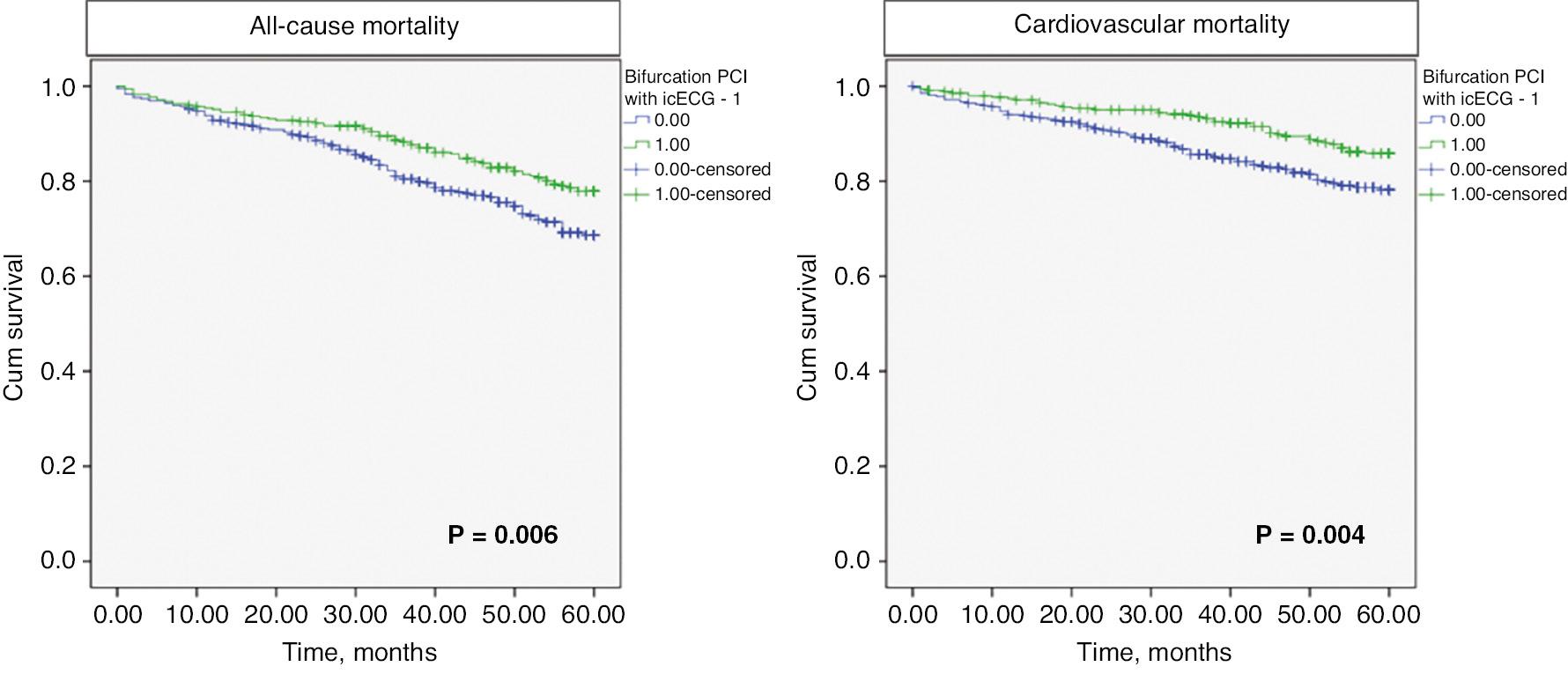
Kaplan-Meier Survival Curves For Events.
Green lines indicate the icECG guided group; blue lines indicate the standard of care (provisional stenting) group.
Because of the strong association observed between icECG guidance and troponin dynamics, we analyzed the effect of this strategy in three troponin strata: 1) normal vs. elevated at baseline; 2) more or less than 5 × UNL post PCI; and 3) presence of absence of an increase post PCI. Kaplan-Meier curves (Figures 4 and 5, and Table 4) indicated that icECG guidance had a significant effect on patient survival, particularly in patients with elevated baseline troponin, those with myocardial injury (with troponin increases post-PCI not reaching the cut-off value for periprocedural infarction) and those without changes in baseline troponin. However, even in the remaining groups, the icECG-guided group had numerically better survival.
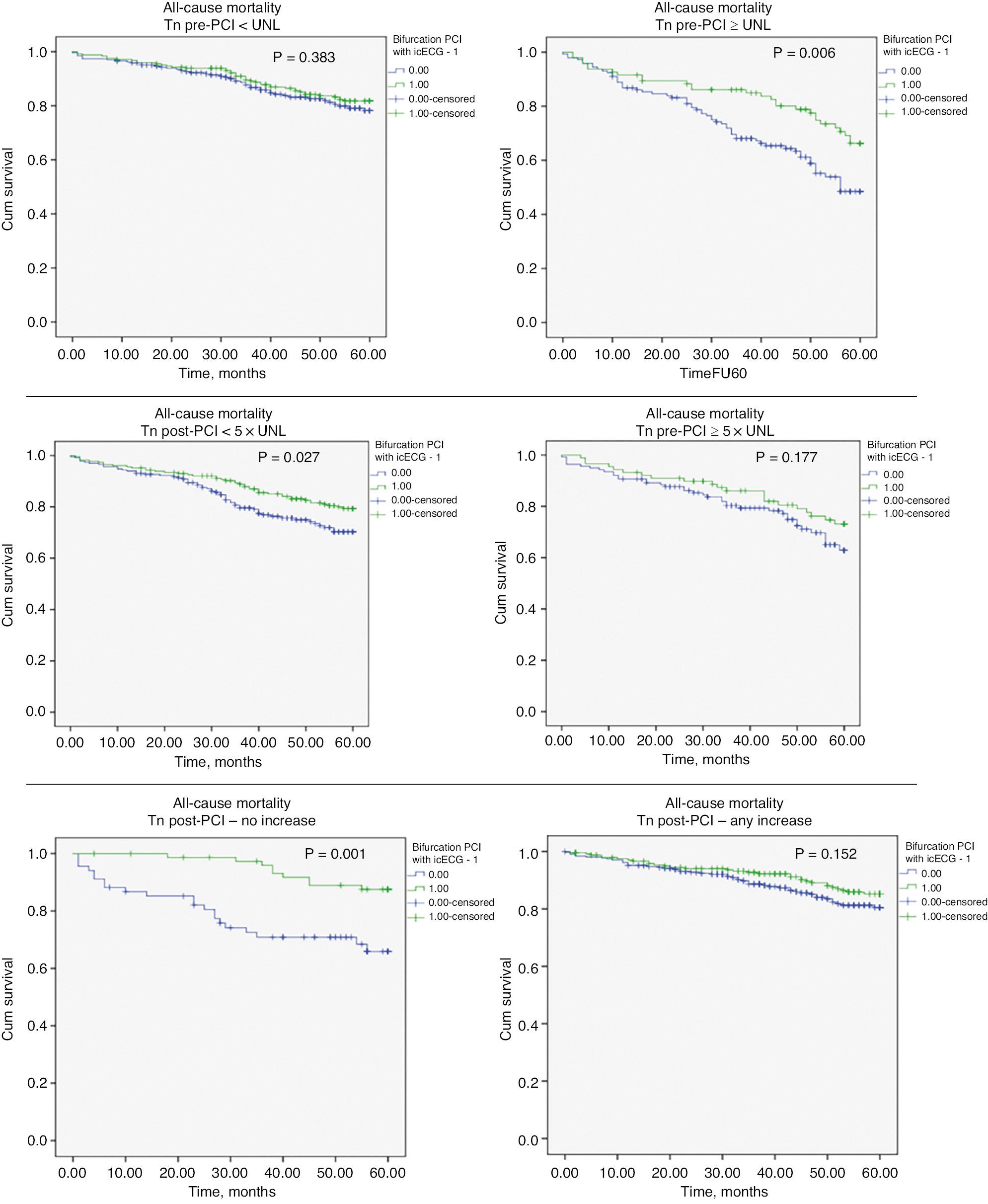
Kaplan-Meier Survival Curves Showing the Effects of an icECG Guided Strategy of Bifurcation Stenting vs. SOC in Different Strata of Troponin Dynamics on All-Cause Death.
Green lines indicate the icECG guided group; blue lines indicate the standard of care (provisional stenting) group.
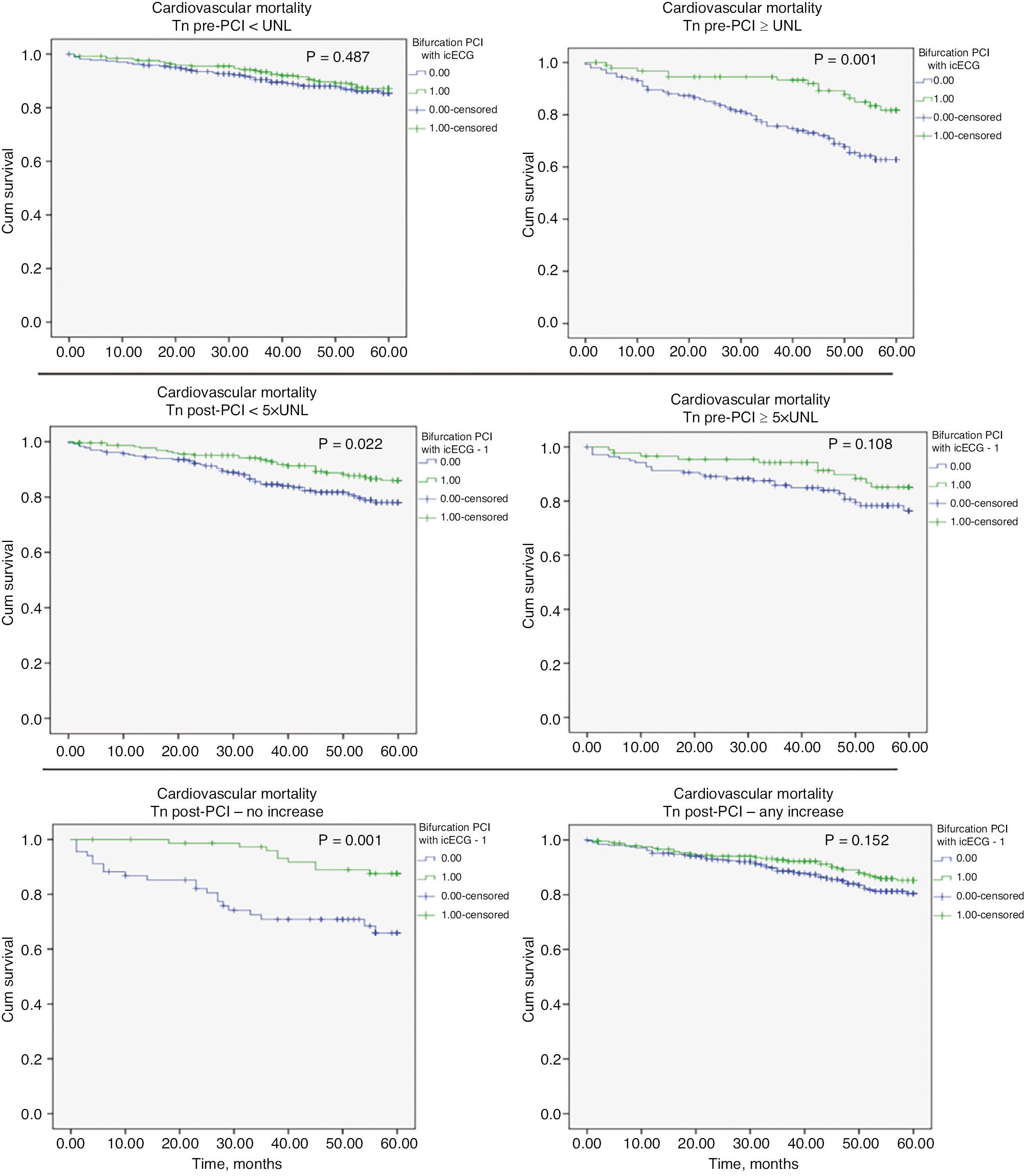
Kaplan-Meier Survival Curves Showing the Effects of an icECG Guided Strategy of Bifurcation Stenting vs. SOC in Different Strata of Troponin Dynamics on Cardiovascular Death.
Mortality Rates among Different Troponin Strata.
| All-cause death | |||
|---|---|---|---|
| Bifurcation PCI strategy | % (n) | P-value | |
| Baseline troponin | |||
| ≤ UNL | SOC | 17.4% (46/264) | 0.383 |
| icECG | 17.1% (42/246) | ||
| > UNL | SOC | 42.4% (61/144) | 0.006 |
| icECG | 29.5% (28/95) | ||
| Troponin post-PCI | |||
| ≤ 5 × UNL | SOC | 25% (58/232) | 0.022 |
| icECG | 19.3% (44/228) | ||
| > 5 × UNL | SOC | 28.1% (39/139) | 0.177 |
| icECG | 23.6% (21/89) | ||
| Any troponin increase post-PCI | |||
| No | SOC | 30.9% (21/68) | 0.001 |
| icECG | 11.7% (9/77) | ||
| Cardiovascular death | |||
|---|---|---|---|
| Bifurcation PCI strategy | % (n) | P-value | |
| Baseline troponin | |||
| ≤ UNL | SOC | 12.5% (33/264) | 0.487 |
| icECG | 11.8% (29/246) | ||
| > UNL | SOC | 31.2% (45/144) | 0.001 |
| icECG | 14.7% (14/95) | ||
| Troponin post-PCI | |||
| ≤ 5 × UNL | SOC | 19% (44/232) | 0.022 |
| icECG | 12.7% (29/228) | ||
| > 5 × UNL | SOC | 19.4% (27/139) | 0.108 |
| icECG | 12.4% (11/89) | ||
| Any troponin increase post-PCI | |||
| No | SOC | 30.9% (21/68) | 0.001 |
| icECG | 11.7% (9/77) | ||
| Yes | SOC | 15.9% (49/309) | 0.152 |
| icECG | 12.8% (31/242) | ||
Abbreviations: UNL, upper normal limit; PCI, percutaneous coronary intervention; SOC, standard of care; icECG, intracoronary electrocardiography.
Multivariate Cox-regression analysis was performed to evaluate independent predictors of mortality (Table 5). In all models, the type of treatment retained in the model independently of the whole model result. A strong trend toward statistical significance was observed between icECG guidance of bifurcation stenting and lower cardiovascular death. Moreover, troponin dynamics strongly influenced survival, such that patients with high postprocedural troponin levels had better survival. In fact, the link between troponin dynamics and survival was observed only when icECG guidance was included in the multivariate model.
Predictors of Mortality in Cox Multivariate Analysis.
| Factor | All-cause death | Cardiovascular death | ||
|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P-value | |
| icECG guidance | 0.844 (0.612–1.163) | 0.300 | 0.673 (0.466–1.041) | 0.074 |
| Age | 1.036 (1.019–1.053) | 0.001 | 1.036 (1.015–1.057) | 0.001 |
| Diabetes | 1.435 (1.056–1.952) | 0.021 | 1.724 (1.175–2.531) | 0.005 |
| Dyslipidemia | 0.576 (0.348–0.953) | 0.032 | 0.410 (0.228–0.737) | 0.003 |
| COPD | 2.025 (1.400–2.929) | 0.001 | 2.368 (1.515–3.700) | 0.001 |
| Serum creatinine, per mmol/l | 1.004 (1.001–1.007) | 0.022 | 1.002 (0.997–1.007) | 0.404 |
| Mitral regurgitation >1st degree | 1.691 (1.191–2.402) | 0.003 | 1.664 (1.060–2.613) | 0.027 |
| Dyspnea (NYHA ≥2 class) | 1.395 (0.968–2.008) | 0.074 | 1.335 (0.845–2.110) | 0.215 |
| Baseline troponin ≥UNL | 1.552 (1.111–2.168) | 0.010 | 1.543 (1.008–2.363) | 0.046 |
| Post PCI increase in troponin | 1.411 (0.965–2.064) | 0.076 | 0.640 (0.411–0.997) | 0.048 |
Abbreviations: HR, hazard ratio; CI, confidence interval; icECG, intracoronary electrocardiography; COPD, chronic obstructive pulmonary disease; NYHA, New York Heart Association; PCI, percutaneous coronary intervention.
Discussion
Our analysis revealed several new findings (Figure 6), as follows: i) The icECG-guided strategy for coronary bifurcation revascularization was associated with lower mortality than SOC. ii) This lower mortality was probably associated with the baseline level of troponin and its dynamics during the intervention. The effect of decreased mortality was most pronounced in patients with no increase or a moderate increase in troponin post-PCI, as well as those with higher-than-normal concentrations. iii) icECG guidance was associated with the use of significantly fewer SB stents, thus leading to fewer total implanted stents and a shorter total stent length for lesions of similar length.
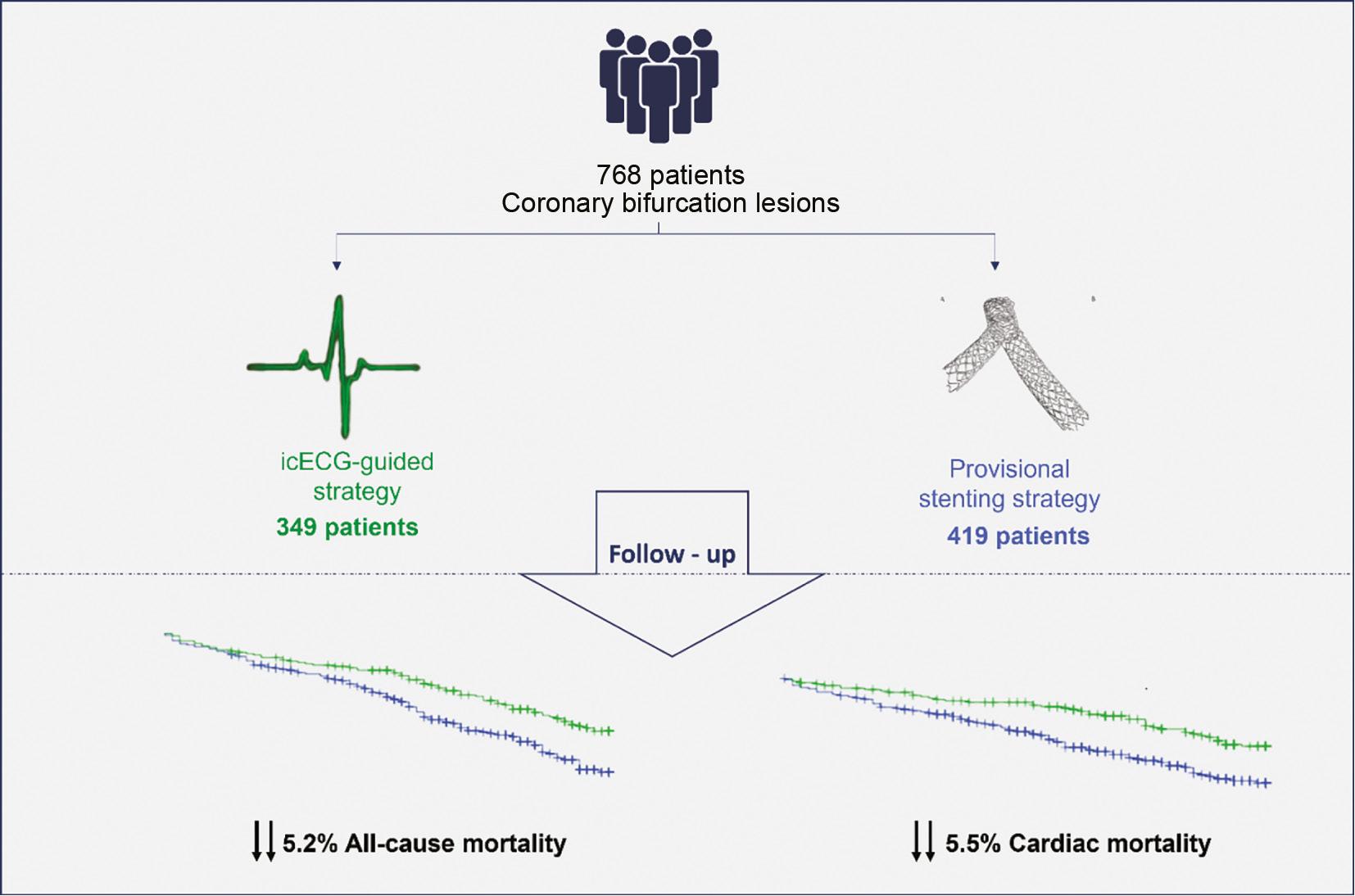
Summary of the Main Findings of the Study, Focusing on the Lower All-Cause and Cardiovascular Mortality in Patients with Intracoronary ECG-Guided Revascularization.
Abbreviation: icECG, intracoronary electrocardiography.
No difference was observed in the baseline risk profiles between patients receiving icECG guidance vs. SOC. A significant difference in patient age was observed between groups, and was higher in the SOC group, thus potentially explaining the different rates of CKD and atrial fibrillation. However, to some extent, this difference was balanced by a higher smoking rate and higher rate of previous MI in the icECG guidance group.
One notable finding of our study was that patients with icECG guidance during bifurcation stenting had higher rates of angiographically significant stenoses in SBs after stenting and at the end of PCI, without an increase in mortality at 5 years. One stent strategy is well known to perform better for coronary bifurcations [8, 14, 15]. Notably, actual ischemia detected with icECG in the SB region is a different entity from a significant fractional flow reserve (FFR) value observed in the same region [11]. As we have demonstrated in the FIESTA study, an FFR ≤0.80 after bifurcation stenting is almost always associated with an icECG ST-segment elevation but can also be associated with no icECG changes [16]. The lack of ECG changes may be associated with normal flow in the region. Furthermore, icECG demonstrated ST-segment elevation in cases without a significant decrease in FFR. The most probable explanation is that distal embolization led to active ischemia and increased peripheral resistance, thus “normalizing” the FFR values. This difference was detectable only with icECG. In addition, in main branches over the length of the vessel, use of a pull-back maneuver was able to detect or exclude active ischemia, thus avoiding unnecessary stenting, in an additional advantage of our method.
We specifically explored periprocedural troponin dynamics and found a relatively smaller increase in troponin post-PCI. These results are in accordance with our previously published findings indicating that icECG guidance decreases periprocedural myonecrosis in bifurcation PCI [10].
We hypothesize that a conservative approach regarding SB ostial stenoses not generating ischemia, and the opposite invasive strategy in stenosis causing ischemia on icECG, resulted in better patient outcomes. The standard approach at the end of a coronary intervention involves observation of patients for chest pain or ST-segment changes on surface ECG. However, surface ECG has been demonstrated to be less sensitive than intracoronary recordings [17]. icECG ischemic changes at the end of a PCI can alert physicians to potential complications that may require additional treatment. The latter could be a probable explanation for the observed effect of icECG guidance on survival.
Whereas functional assessment with FFR and non-hyperemic indexes has questionable value in assessing the ischemic potential of a lesion before PCI, icECG may have a substantial benefit in intraprocedural and post-PCI lesion assessment. This method does not increase the fluoroscopic time or the contrast volume; therefore, it can be used as a complement to FFR to avoid unnecessary interventions and benefit patients’ clinical outcomes.
Limitations
The main limitation of our study is its observational design. In addition, this was a single-center study with no independent adjudication and some restrictions due to cost limitations. However, we carefully selected patients, and, to our knowledge, this is the first study to assess the effects of icECG guidance on clinical outcomes in patients with bifurcation PCI.



