Abbreviations: AVB, atrioventricular block; AVNA, atrioventricular node ablation; BiVP, biventricular pacing; COI, current of injury; CRT, cardiac resynchronization therapy; CS, coronary sinus; CSP, conduction system pacing; EGM, electrogram; HBP, His bundle pacing; LBB, left bundle branch; LBBAP, left bundle branch area pacing; LBBP, left bundle branch pacing; LVSP, left ventricular septal pacing; LVAT, left ventricular activation time; NICM, nonischemic cardiomyopathy; PICM, pacing-induced cardiomyopathy; PoLBB, LBB potential; RVP, right ventricular pacing; RWPT, R-wave peak time; SND, sinus node dysfunction.
Introduction
For decades, right ventricular pacing (RVP) has been the standard of care for nonreversible bradycardia. However, long-term RVP is associated with electrical and mechanical dyssynchrony, thus contributing to the development of cardiomyopathy [1]. Even biventricular pacing (BiVP), which delivers cardiac resynchronization therapy (CRT), has a 30%–40% non-response rate [2]. These deleterious effects of RVP have led to the development of the more physiological conduction system pacing (CSP), which includes His bundle pacing (HBP) and left bundle branch pacing (LBBP) [3–7].
HBP achieves synchronized activation of both ventricles through the native His-Purkinje system and has been widely used. Limitations of HBP include a higher implantation difficulty than conventional pacing and capture threshold, and a risk of lead revision [3–5]. The novel technique of LBBP, first reported by Huang et al. in 2017, achieves synchronization through direct capture of the left bundle branch (LBB), with a low and stable pacing threshold, and a high success rate [6, 7]. Studies on LBBP have steadily increased. This review summarizes the most recent evidence supporting LBBP use in defining and evaluating LBB capture, and discusses its applications.
Definition of LBBP
LBBP is defined as the direct capture of the LBB trunk or its fascicles, together with the left ventricular (LV) septal myocardium, to achieve rapid physiological LV activation at low output [8]. The anatomical basis of LBBP is that LBB fibers lie within the LV septum and underneath the endocardium, and have a broad and expansive structure, thus enabling targeting and capture with a low and stable pacing threshold. The key aspect differentiating LBBP from left ventricular septal pacing (LVSP) is the confirmation of LBB capture [9]. LVSP has a similar narrow QRS morphology to that with LBBP, but it results in slower depolarization of the LV lateral wall by activating only the myocytes [10]. Left bundle branch area pacing (LBBAP) consists of both LBBP and LVSP, without requiring clear evidence of LBB capture (Figure 1).
Evaluation of the Criteria for LBB Capture
Although no compelling evidence comparing the clinical outcomes between LBBP and LVSP has been published, confirmation of LBB capture is essential to optimize synchronized ventricular activation and avoid pacing-related complications [10]. Herein, we review the currently accepted standards for LBB capture, as well as findings from several studies investigating personalized criteria.
General Criteria
Paced QRS Morphology
Paced right bundle branch block (RBBB) morphology and an RBB delay pattern (qR/rSR) in lead V1 can be used as a screening tool for LBBP, by indicating earlier activation of the LV than the right ventricle [11]. However, paced RBBB morphology is necessary but not sufficient to confirm LBB capture. Pacing at the left sided distal His or proximal LBB can result in incomplete RBBB [12]. When the lead is fixed deeper in the interventricular septum (IVS), paced RBBB has also been observed without LBB capture in 23.3% of patients without LBBB and 44.4% of patients with LBBB [8].
LBB Potential and LBB Current of Injury
LBB potential (PoLBB) is often recorded in patients without LBBB during intrinsic rhythm, with a 20–30 ms interval from the potential to the QRS onset [13]. In patients with LBBB, PoLBB is concealed in ventricular electrograms (EGMs) but can be recorded during successful HBP with the restoration of left bundle conduction or during ventricular premature contraction or ventricular escape rhythm in a narrow QRS or RBBB pattern [14]. The demonstration of PoLBB can indicate that the lead was placed at the LBB area, and can help confirm the level of conduction block, but it does not provide direct evidence of LBB capture [13]. Su et al. have recorded LBB current of injury (COI) in 67% (77/115) of patients with LBB potential (Figure 2F) [15]. LBB COI indicates damage to the cell membranes in the LBB as a result of the trauma of electrode pressure, and may indicate that the lead tip is directly adjacent to the left conduction system. All patients with LBB COI have been confirmed to have a low threshold for LBB capture (<1.5 V/0.5 ms) [15]. Hence, the presence of LBB potential and LBB COI provide value in LBB capture confirmation. Detailed procedures of recording PoLBB and COI have been described by Su et al. [15].
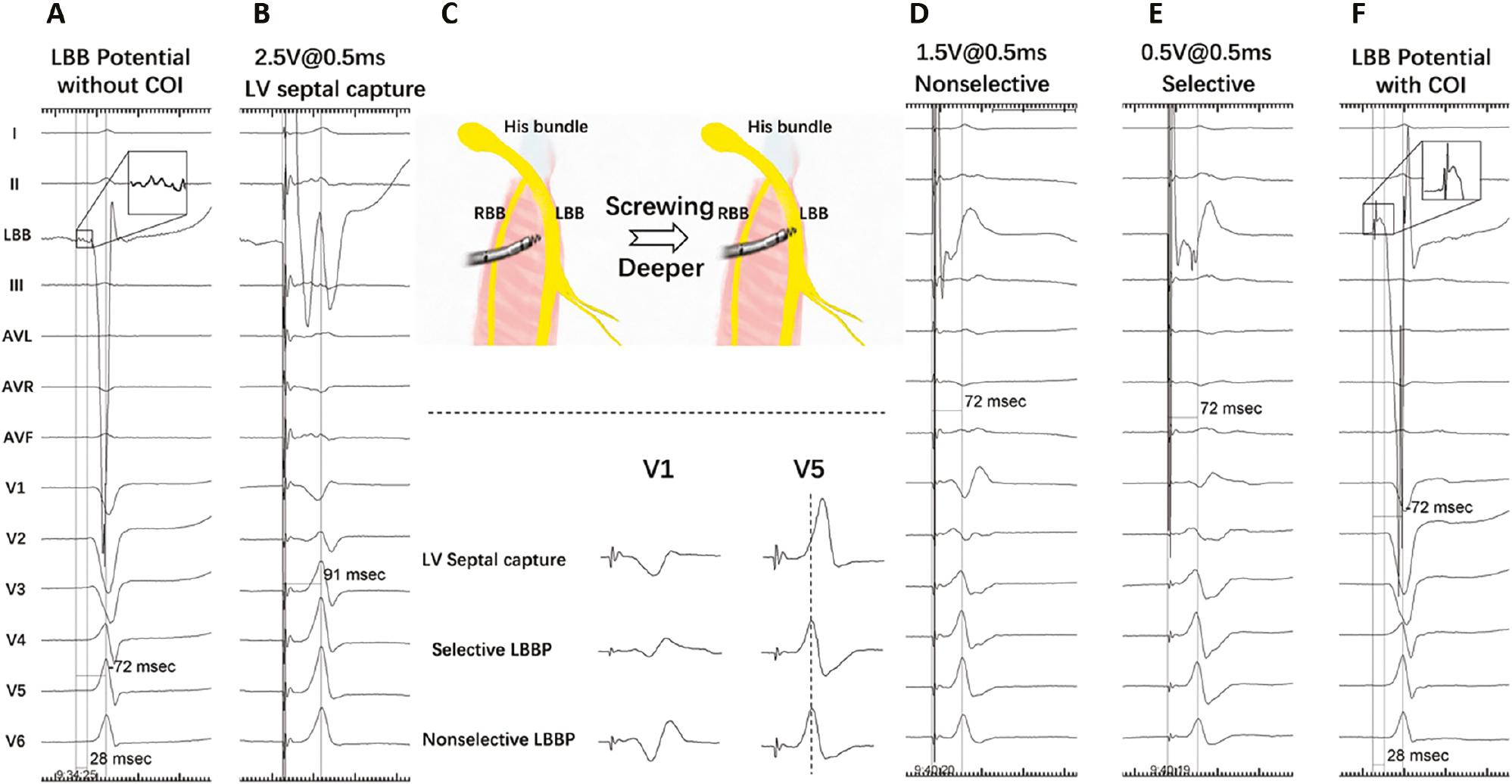
Electrophysiological Characteristics.
(A) LBB potential without COI during intrinsic rhythm, with an LVAT of 72 ms. (B) Stim-LVAT of 91 ms at 2.5 V/0.5 ms during septal capture. (C) Transition from deep septal to LBB capture and comparison of the morphology of V1/V5. (D) Stim-LVAT of 72 ms at 1.5 V/0.5 ms during nonselective LBBP. (E) Stim-LVAT of 72 ms at 0.5 V/0.5 ms during selective LBBP. (F) After screwing, LBB potential with COI was recorded, with an intrinsic LVAT of 72 ms. LVAT, left ventricular activation time; Stim-LVAT, pacing stimulus to left ventricular activation time; P, potential; COI, current of injury; RBB, right bundle branch; LBB, left bundle branch. (Adapted from Su et al. J Cardiovasc Electrophysiol. 2020;31(4):834–842.)
Left Ventricular Activation Time
Left ventricular activation time (LVAT) is measured from the pacing stimulus to the peak of the R wave in lead V5 or V6, and indicates fast activation propagation throughout the LV free wall [8]. After the LBB is captured, LVAT abruptly shortens and remains constant at both high and low outputs (Figure 2D, 2E) [11].
Abrupt changes in LVAT ≥10 ms are observed in the transitions from both non-selective LBBP (NS-LBBP) and selective-LBBP (S-LBBP) to LVSP. These transitions have been demonstrated to have 100% specificity in confirming LBB capture [8, 13]. Huang et al. have reported that an LVAT of 75 ms in patients without LBBB has a specificity of 95% and a sensitivity of 82% in confirming LBB capture, whereas, in patients with LBBB, an LVAT of 85 ms has 93% specificity and 76% sensitivity in confirming LBB capture [8]. Qian et al., in a pilot study investigating the association between LVAT and mechanical synchrony by using SPECT myocardial perfusion imaging, have found that an LVAT <76 ms identifies patients with better LV mechanical synchrony than those with LVAT ≥76 ms and might be a reasonable parameter for defining LBB capture during procedures [16]. The LVAT varies depending on the conditions in individual patients, particularly those with intraventricular conduction diseases, ischemic cardiomyopathy with substantial scarring, or abnormal left ventricular size [17]. A single LVAT cut-off value therefore might not be sufficient to determine LBB capture in all patients.
S-LBBP and NS-LBBP
S-LBBP is defined as capture of only the LBB without local myocardium and is demonstrated by typical RBBB morphology (M or rsR′, wide R′ with a notch in lead V1, and wide and deep S with a notch in leads I, V5, and V6) in electrocardiograms (ECGs) and a discrete component in EGMs, at low pacing output (Figure 2C) [11]. S-LBBP has 100% specificity in confirming LBB capture and is obtained in 39.6%–90% of patients undergoing LBBP [8, 14, 15]. In a large observational study, 75.4% (460/618) of the study population showed S-LBBP at the time of implantation, whereas only 30.9% (191/618) showed S-LBBP during follow-up [18]. This decline might have resulted from changes in the thresholds for LBB capture and local myocardial capture during the follow-up period. Repeated testing after recovery of COI, different rates of pacing and pulse widths, and programmed stimulation can help demonstrate S-LBBP in more patients [8].
NS-LBBP is observed when the LBB and local myocardium are both captured. The paced morphology of NS-LBBP shows QR; a narrow R without a distinct notch in lead V1; a narrow and small S without a notch in leads I, V5, and V6; and no discrete component in the EGM [11]. The LVAT remains short and stable during the transition from NS-LBBP to S-LBBP, whereas a prolonged LVAT is observed during the transition from NS-LBBP to LVSP [8].
Retrograde His Bundle Potential and Anterograde Distal LBB Potential
Capturing the LBB can result in retrograde and anterograde activation of the Purkinje conduction system, regardless of myocardial capture. The interval between the LBB pacing stimulus and the retrograde His potential (Stim-PHis), and the demonstration of the anterograde distal potential of the left bundle branch (PLBB), i.e., the potential of the left conduction system (PLCS), are considered to provide direct support for LBB capture [13]. Huang et al. have reported that Stim-PHis (21.8 ± 5.9 ms) is the same as the intrinsic interval from the His potential to PLBB (21.8 ± 6.9 ms) in patients without LBBB. Furthermore, the Stim-PHis values remain stable during the transition from S-LBBP to NS-LBBP at low and high output. In patients with LBBB, PLCS can be recorded by corrective His pacing or during LBB capture [8].
Programmed Deep Septal Stimulation
Deep septal pacing often results in NS-LBBP, by stimulating the LBB and the local septal myocardium simultaneously. The paced QRS morphologies of LBB capture and deep septal myocardial capture are both relatively narrow and show an RBBB pattern, and consequently are difficult to separate [19]. Programmed deep septal stimulation can reveal changes in QRS morphology, increases in QRSd, axis shift, and prolonged R-wave peak time (RWPT) in leads V5 and V6 (Figure 3) [17]. Jastrzebski et al. have performed programmed deep septal stimulation in 143 patients, with a basic drive train of 600 ms, and have demonstrated LBB capture in 79.7% of patients. The average septal-myocardial effective refractory periods have been determined to be significantly shorter than the effective refractory periods of the LBB (263.0 ± 34.4 ms vs. 318.0 ± 37.4 ms, P < 0.01) [19]. This method can be helpful when the paced QRS morphology and other criteria for LBB capture cannot be demonstrated.
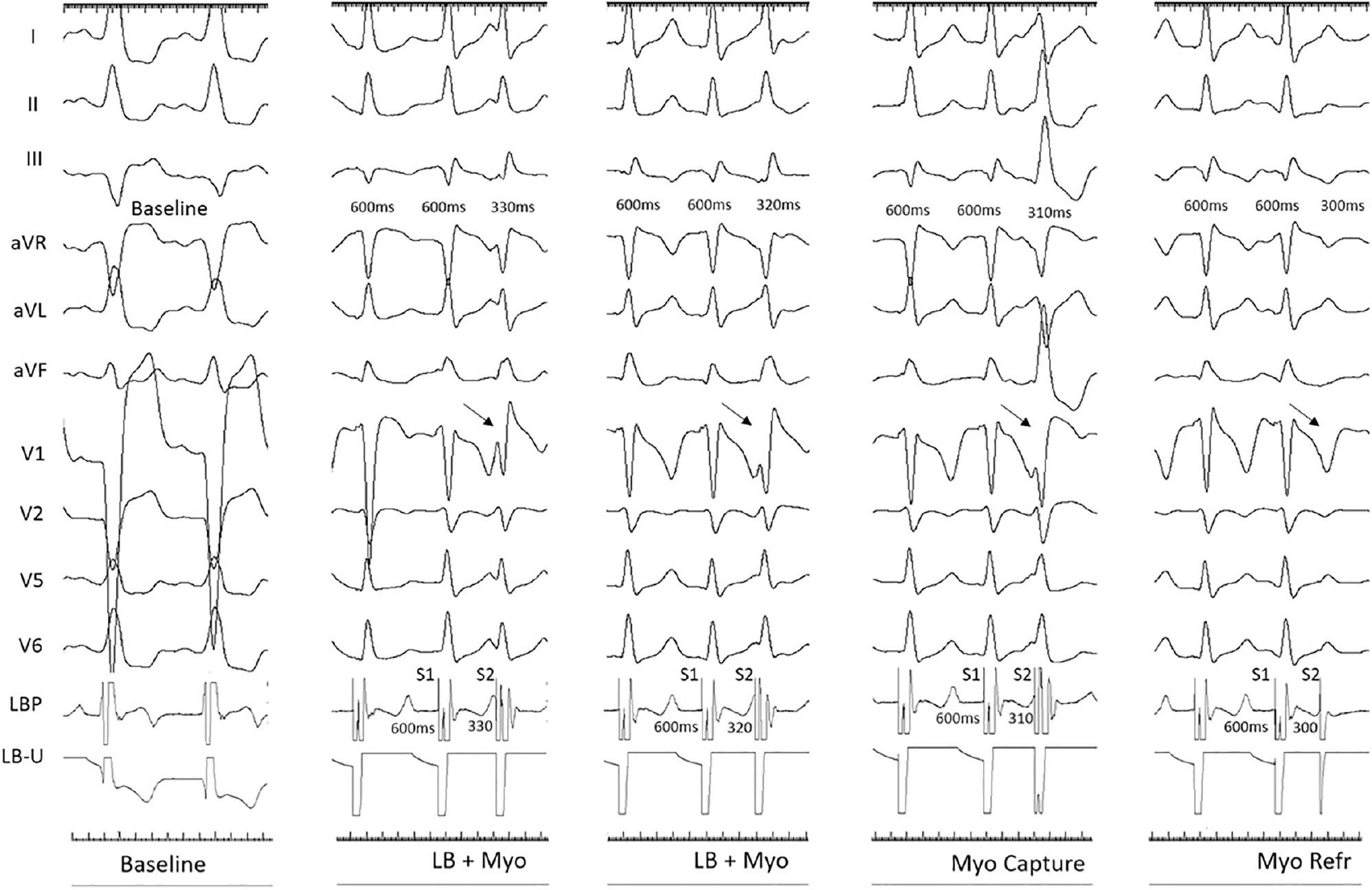
Demonstration of Programmed Deep Septal Stimulation.
Programmed deep septal stimulation was performed with the pacing lead (LBP). In patients with baseline LBBB, the LB refractory period was 320 ms (LB + septal myocardial capture until 320 ms), and the septal myocardial refractory period was 300 ms. Change in QRS morphology, axis, and prolongation of the duration after loss of LB capture at S1 600 ms and S2 310 ms. LB, left bundle; Myo, myocardial; Refr, refractory. (Adapted from Ponnusamy et al. J Cardiovasc Electrophysiol. 2020;31(9):2462–2473.)
Physiology-based Individualized Criteria
Physiology-based Electrocardiographic Criteria
Depending on the dynamic and output-dependent changes in QRS morphology and LVAT, which are considered the gold standard for LBB capture, precise ECG criteria can be developed to differentiate the paced QRS complexes of LBB capture from LVSP. LBB capture can restore the physiological activation of the LV; thus, the native QRS provides a valuable reference for confirming LBB capture [20]. Jastrzebski et al. have analyzed 357 ECGs from 124 patients: 118 with native rhythm, 124 with NS-LBBP, 69 with S-LBBP, and 46 with LVSP. In the patients without LBBB, the paced V6 RWPT, measured from QRS onset, was equal to the native V6 RWPT, thus confirming LBB capture with 98% sensitivity and 86% specificity (Figure 4A). The paced V6 RWPT (measured from the stimulus) was also identical to the interval between the LBB potential to V6 R-wave peak, with a sensitivity of 88.2% and specificity of 95.4%. A 74 ms cut-off value of V6 RWPT has been found to have 100% specificity in confirming LBB capture in patients without LBBB (Figure 4B) [20].
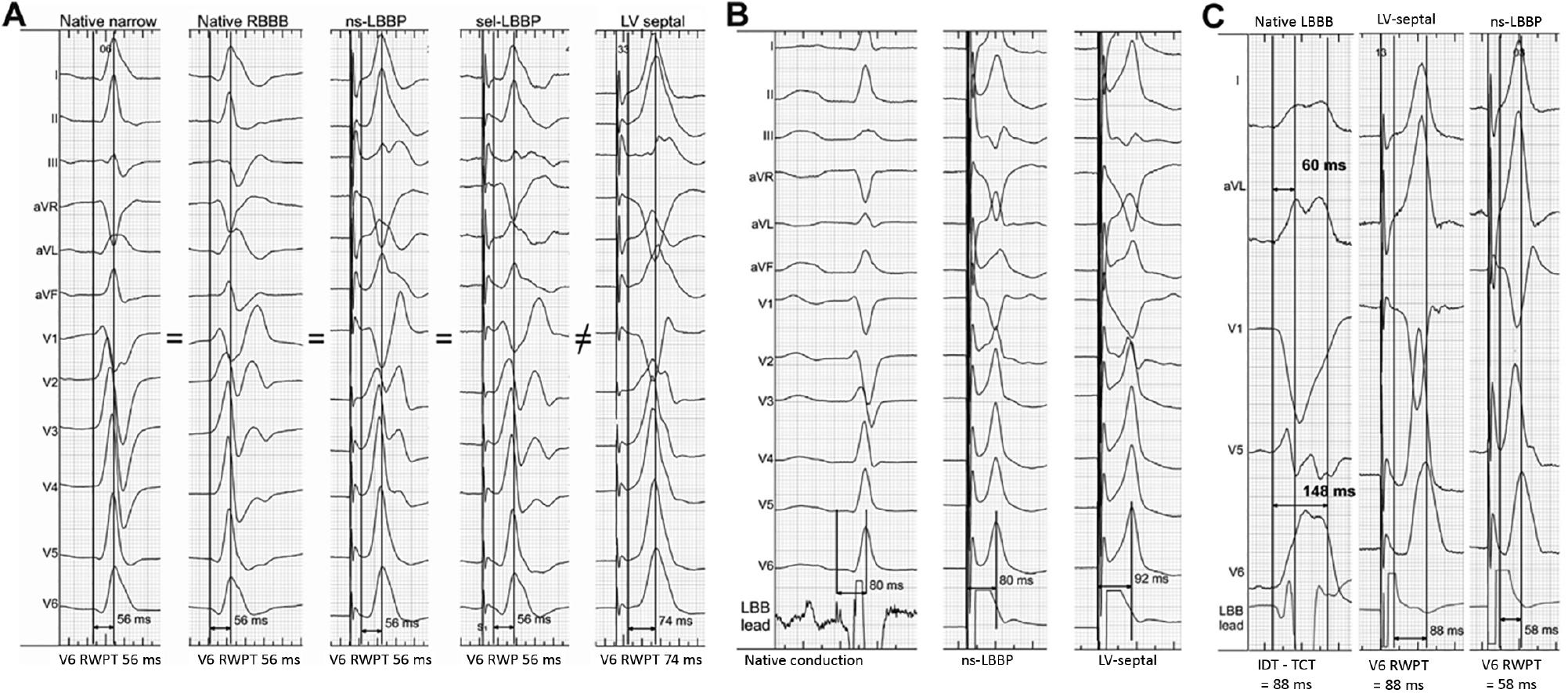
Cases of Physiology-based Electrocardiographic Criteria.
(A) The paced V6 RWPT (measured from the QRS onset) is identical to the V6 RWPT during native non-LBBB rhythm, native RBBB, non-selective LBBP, and selective LBBP, whereas LVSP results in a prolonged V6 RWPT. (B) In patients with non-LBBB rhythm, the paced V6 RWPT (measured from the stimulus) is identical to the PLBB to the V6 R-wave peak, whereas the interval is longer in LVSP. (C) In patients with native LBBB rhythm, a paced V6 RWPT (measured from the stimulus) shorter than IDT-TCT can define LBB capture. RWPT, R-wave peak time; RBBB, right bundle branch block; PLBB, LBB potential; IDT, intrinsicoid deflection time (time from QRS onset to the beginning of the final rapid downsloping phase of the R wave); TCT, transseptal conduction time (time from QRS onset to the arrival of depolarization at the left side of the interventricular septum). (Adapted from Jastrzębski et al. Heart Rhythm. 2021;18(6):935–943.)
In patients with LBBB, the V6 intrinsic deflection time (IDT, interval from QRS onset to the beginning of the final rapid downslope of the R wave) is prolonged, because of a delay in transseptal conduction and the non-physiological activation of LV. Transeptal conduction time (TCT) was defined as the time interval between QRS onset and the beginning of the first notch of the lateral leads. Paced V6 RWPT + 10 ms <IDT - TCT has been demonstrated to have a sensitivity of 77.8% and specificity of 100% in indicating LBB capture (Figure 4C). Paced V6 RWPT ≤80 ms has specificity of 100% in confirming LBB capture [20].
V6-V1 Interpeak Interval
Although several studies have demonstrated the cut-off values of LVAT for LBB capture, extensive LVAT overlap exists between NS-LBBP and LVSP in real-world practice [16, 20]. The LBB paced LVAT might be prolonged by the slow propagation of left conduction system disease, LV dilatation in heart failure (HF), or a combination thereof, thus limiting the sensitivity for detecting LBB capture. Furthermore, criteria based on QRS morphology for differentiating NS-LBBP and S-LBBP are lacking [21]. Jastrzebski et al. have analyzed 124 patients (239 ECGs) and compared the V6 RWPT, V1 RWPT, and V6-V1 interpeak interval among the three types (NS-LBBP, S-LBBP, and LVSP). The longest V6-V1 interval value was observed in S-LBBP (62.3 ± 21.4 ms), followed by NS-LBBP (41.3 ± 14.0 ms), and the shortest was observed in LVSP (26.5 ± 8.6 ms). The optimal V6-V1 interpeak interval value for differentiating NS-LBBP and LVSP was >33 ms, with a sensitivity of 71.8% and a specificity of 90.0%. The 100% specific cut-off value for the V6-V1 interpeak interval was >44 ms. This new criterion is based on the individual intra-QRS time instead of the initial latency after the pacing stimulus (Figure 5) [21]. That study has also indicated that prolongation of approximately 20 ms in V1 RWPT and V6 RWPT represents the loss of myocardial capture and of LBB capture, respectively, thus providing a deeper understanding of the physiology of LBB pacing [21].
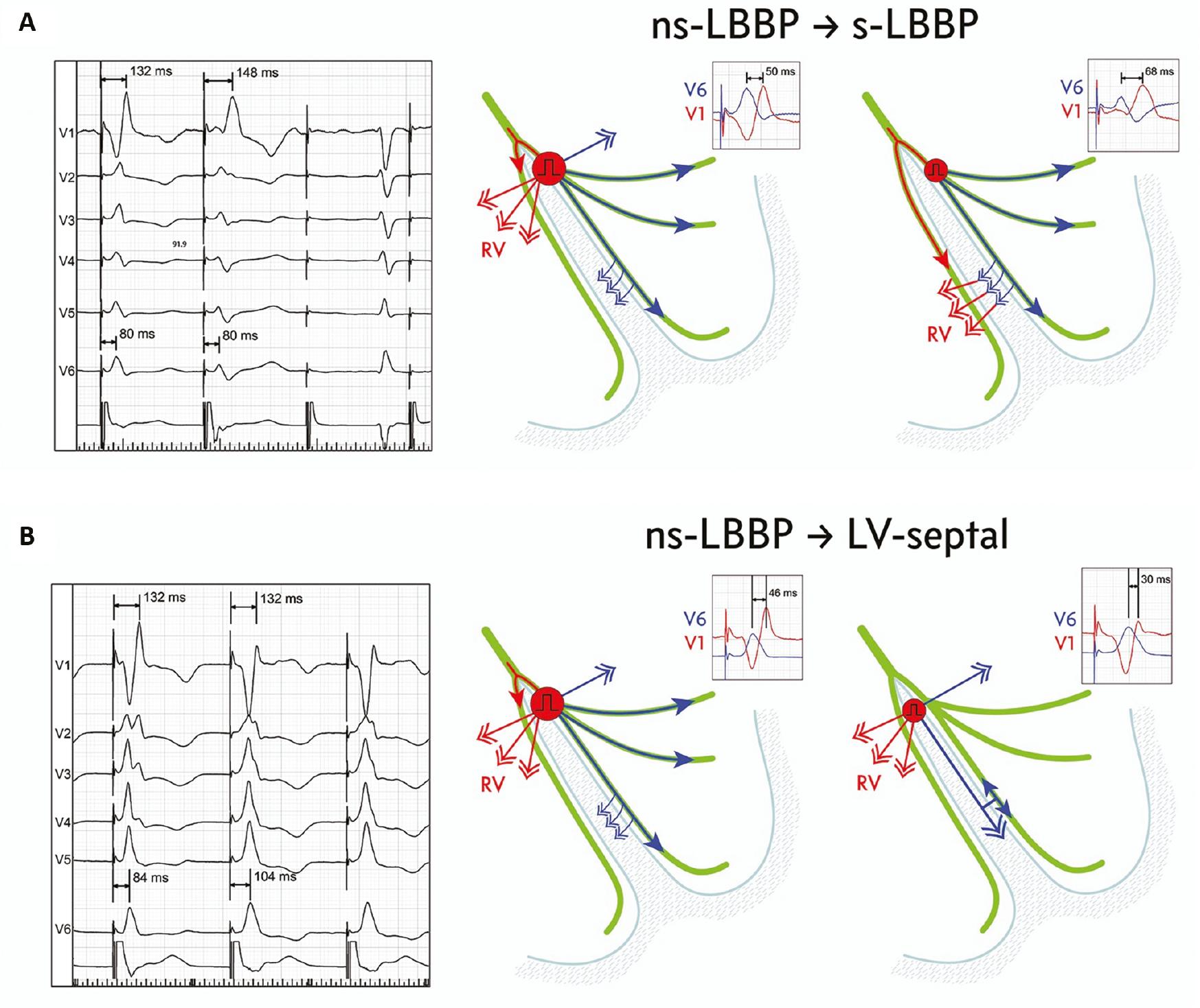
Left Bundle Branch (LBB) Area Pacing with Hypothesized Ventricular Activation Patterns During QRS Morphology Transitions.
(A) Transition from non-selective LBB capture to selective LBB capture results in a delay of right ventricular (RV) activation due to a loss of RV depolarization via direct septal myocardial activation. RV activation proceeds via transseptal conduction from left septal fascicles. Electrocardiographic markers of delayed RV activation are V1 R-wave peak time prolongation and an increased V6-V1 interval. (B) Transition from non-selective to septal LBB capture does not influence right ventricular activation but delays left ventricular activation, owing to a loss of direct LBB capture. (Adapted from JastrzeRbski et al. EP Europace. 2022;24(1):40–47.)
Physiology-based Intracardiac Ventricular Electrogram Criteria
Chen et al. have recorded intracardiac ventricular electrograms with a coronary sinus (CS) catheter in 43 patients in whom LBBP was attempted [22]. In 27 patients with successful LBBP, the LV activation sequences were identical to their intrinsic rhythm, whereas in 16 patients who underwent LVSP, intrinsic sequences were not maintained. LBBP can preserve a normal ventricle activation sequence and better electrical synchrony than LVSP. A novel algorithm combining LBB potential with 70 ms and 85 ms cut-off values for LVAT has been developed to differentiate LBBP and LVSP during implantation, with a sensitivity of 95.2% and a specificity of 93.7%. This accurate differentiation may be helpful when patients do not demonstrate S-LBBP or abrupt shortening ≥10 ms of the LVAT [22].
∆LVAT Based on Individual HBP and RVSP
Because LBBP captures the distal HB, the fastest activation of the LV lateral wall and the shortest LVAT have been observed with this method [23]. Qian et al. have developed a personalized LBB capture criterion based on patients’ intrinsic HBP and RVSP electrical parameters during implantation [24]. A total of 105 patients were enrolled: 80 with normal cardiac function and 25 with HF. The difference (∆) in LVAT between HBP and LBBP/LVSP was defined as ∆LVAT1, and the difference in LVAT between RVSP and LBBP/LVSP was defined as ∆LVAT2 (Figure 6). Among patients with normal cardiac function, ∆LVAT1 >12.5 ms had a sensitivity of 73.9% and a specificity of 93.3% as a cutoff for confirming LBB capture, whereas a cutoff of ∆LVAT1% (∆LVAT1/LVATHBP) >9.8% exhibited 92.0% sensitivity and 92.3% specificity. The optimal cutoff of ∆LVAT2% (∆LVAT2/LVATRVSP) for differentiating LBBP from LVSP was 21.2%, with a sensitivity of 84.0% and a specificity of 100%. Among patients with HF, ∆LVAT1 >9.0 ms and ∆LVAT1% >9.8% have been found to have high accuracy in indicating LBB capture [24]. Vijayaraman et al. have also reported that an absolute value of 8 ms for the difference in RWPTs (∆RWPT) during HBP and ns-LBBP/LVSP is highly accurate (sensitivity of 100% and specificity of 93.3%) in confirming LBB capture in patients with LBBB [23]. These criteria provide a reliable reference for confirming LBB capture during the implantation procedure in patients with or without HF.
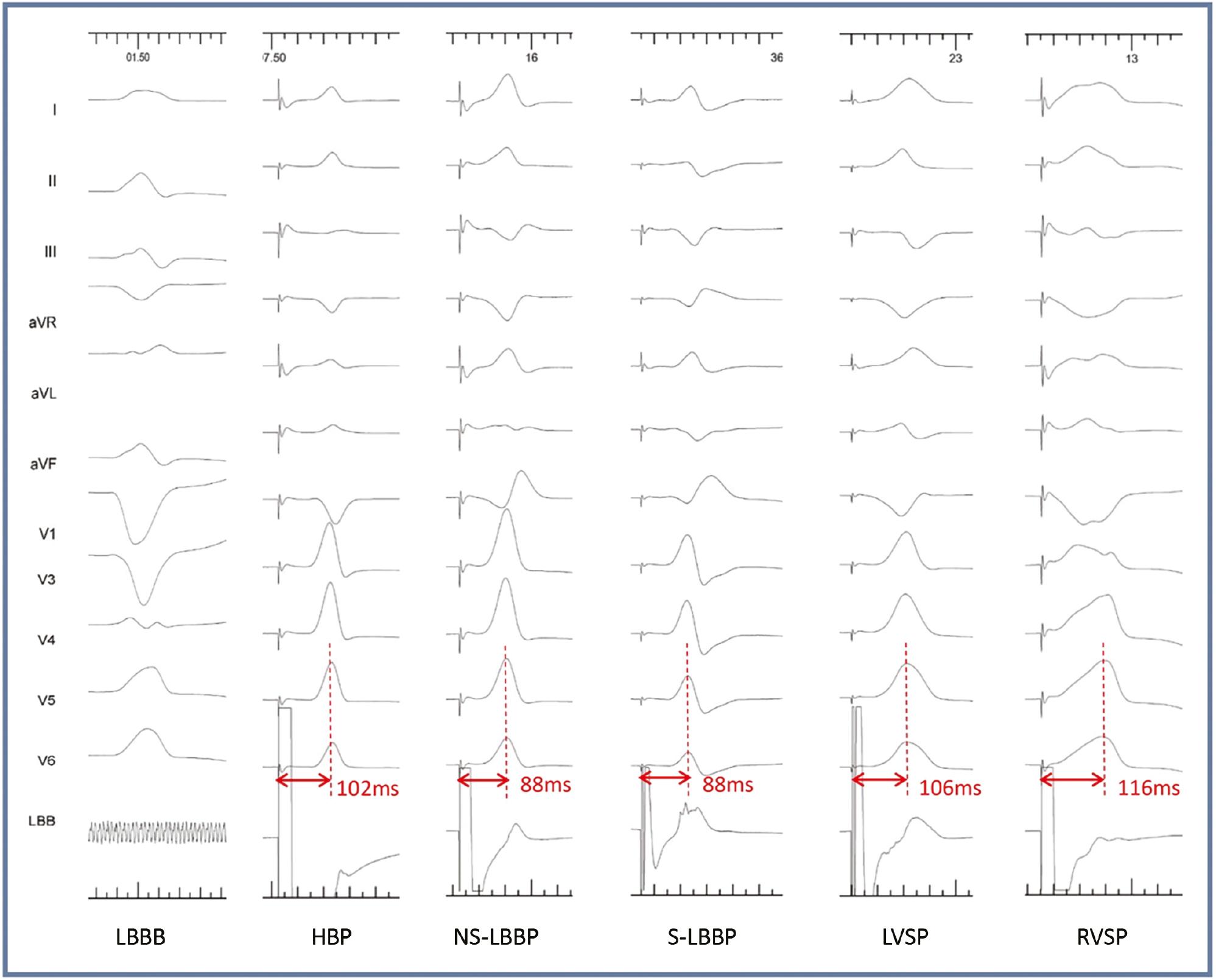
The Case of a Patient with HF and LBBB During Corrective HBP, RVSP, LBBP, and LVSP.
SLBBP or NS-LBBP produced a short LVAT of 88 ms, whereas corrective HBP produced an LVAT of 102 ms. The ∆LVAT1 was 14 ms, and the ∆LVAT1% was 13.7%. LVSP presented a long LVAT of 106 ms with a ∆LVAT1 of 4 ms. The LVAT of RVSP was 116 ms, and the ∆LVAT2% was 24.1% for LBBP. (Adapted from Qian et al. Heart Rhythm. 2022;19(12):1984–1992.)
Recommended Algorithm for Confirming LBB Capture
Although many criteria have been developed, comprehensive consideration and evaluation are essential to confirm LBB capture. We propose an algorithm (Figure 7) combining widely recognized criteria and several physiology-based individualized criteria, which may be helpful when diagnosis is difficult. We recommend that clinicians achieve LBBP in patients needing CRT, particularly those with LBBB and LV dysfunction. For patients with bradycardia and preserved cardiac function, both LBBP and LVSP are acceptable. However, long-term studies are needed to confirm whether the clinical efficacy between LBBP and LVSP differs. Meanwhile, implanters must critically strike a balance between safety and achieving LBB capture.
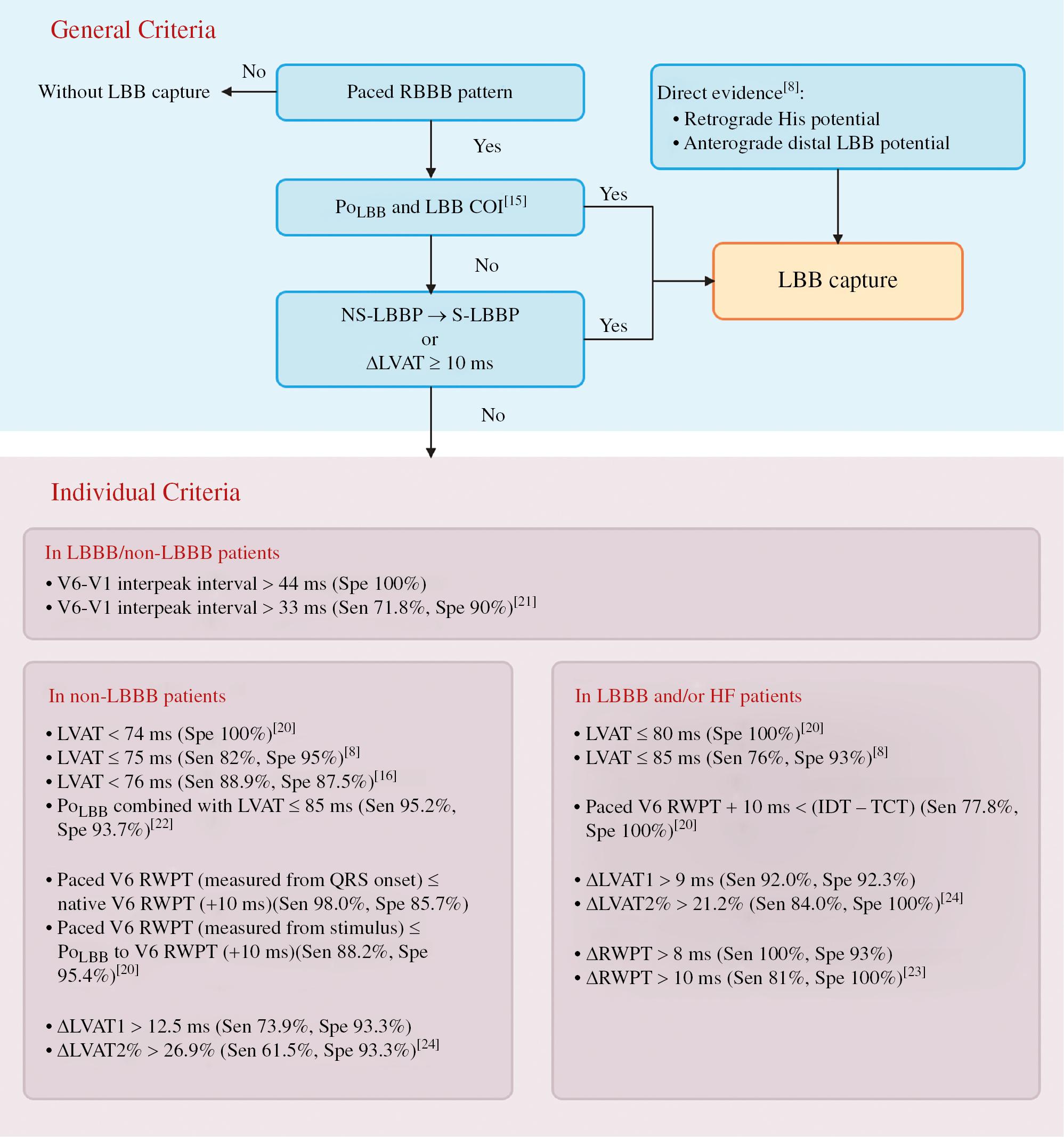
Algorithm to Confirm LBB Capture.
LBB, left bundle branch; LBBB, LBB block; RBBB, right bundle branch block; PoLBB, LBB potential; COI, current of injury; Stim-LVAT, stimulus to left ventricular activation time; ∆, absolute change; IDT, intrinsicoid deflection time (time from QRS onset to the beginning of the final rapid downsloping phase of the R wave); TCT, transseptal conduction time (time from QRS onset to the arrival of depolarization at the left side of the interventricular septum); RWPT, V6 R-wave peak time; ∆RWPT, RWPTHBP - RWPTnsLBBP/LVSP; ∆LVAT1, LVATHBP - LVATLBBP/LVSP; ∆LVAT2%, (LVATRVSP - LVATLBBP/LVSP)/LVATRVSP; Sen, sensitivity; Spe, specificity.
Applications of LBBP
LBBP in Bradycardia
LBBP has been demonstrated to be a safe and feasible treatment for patients with bradycardia in many studies [18, 25–45]. In 2021, Su et al. reported the longest-term observational study of LBBP in patients with pacing indications [18]. LBBP was successful in 618/632 (97.8%) patients. The pacing thresholds remained low and stable during the mean follow-up of 18.6 ± 6.7 months. Improvements in LVEF (57.08 ± 16.60% versus 62.36 ± 12.20%, P < 0.001) and decreases in LVEDD (52.27 ± 7.51 versus 50.73 ± 6.71 mm, P < 0.001) were observed after the 1-year follow-up. LBBP resulted in only a modest increase in QRSd in patients with atrioventricular block (AVB) or atrial fibrillation (AF) (108.41 ± 26.69 versus 111.35 ± 14.41 ms, P = 0.030) and sinus node dysfunction (SND) (98.75 ± 19.13 versus 110.59 ± 14.83 ms, P < 0.001), thus suggesting that LBBP maintains rapid LV activation. Only six patients (1%) had the threshold increased to >3 V or a loss of LBB capture, and two patients required lead revision. The largest study evaluating the outcomes of LBBAP for a variety of indications was published in 2022 by Jastrzębski et al. [40]. That study analyzed 2533 patients from 14 European centers, including 1837 patients with bradyarrhythmia as an indication [40]. The success rate of LBBAP was 92.4% in these patients and 82.2% in patients with HF. Left bundle fascicular capture was the predominant type of LBBAP.
Recently, application of LBBP in patients after transcatheter aortic valve replacement (TAVR) has been reported by Vijayaraman et al. The success rate of LBBAP was 26/28 (93%), a value higher than that of HBP (29/46, 63%) [46]. Gul et al. have also reported LBBAP success in 80/84 (95%) patients with valvular interventions [35]. Niu et al. have compared the procedural and clinical outcomes of CSP, including HBP (n = 10) and LBBP (n = 20), with RVP (n = 30) in patients who developed AVB after TAVR. The success rate of LBBP was significantly higher than that of HBP (95.2% vs. 62.5%, P < 0.001). During a mean follow-up of 15.0 ± 9.1 months, the LVEF in the LBBP group was higher (54.9 ± 6.7% vs. 48.9 ± 9.1%, P < 0.05), and the LVEDD was shorter (49.7 ± 5.6 mm vs. 55.0 ± 7.7 mm, P < 0.05), than that in the RVP group [47]. In these studies, the pacing parameters of LBBP remained stable, and no acute complications occurred. LBBP therefore is feasible and safe for patients with prior valvular interventions, and might maintain better cardiac function than RVP during follow-up [35, 46, 47].
Several studies have reported LBBP in patients with intrinsic RBBB and pacemaker indications [48–50]. Li et al. have attempted LBBAP in 55 patients with bradycardia, 29 of whom had RBBB [49]. LBBAP was successful in 93.1% (27/29) of patients with RBBB and significantly shortened the QRSd (143.1 ± 16.6 ms to 119.5 ± 11.7 ms). Vijayaraman et al. have conducted LBBAP in patients with RBBB, HF, and LV dysfunction. The success rate of LBBAP was 88% (107/121), and the QRSd narrowed from 156 ± 20 ms to 150 ± 24 ms (P = 0.01) [51]. That study has demonstrated the feasibility of LBBP in patients with RBBB. However, further evidence is needed to understand the mechanism of QRSd narrowing of RBBB by LBBP.
RVP, the current standard treatment for bradycardia, can lead to prolonged QRSd, LV dyssynchrony, and cardiac systolic dysfunction in pacemaker-dependent patients, and has been demonstrated to increase the risk of AF and heart failure hospitalization (HFH) [1, 52]. This deleterious effect of RVP on LV remodeling has been termed pacing-induced cardiomyopathy (PICM). HBP has been demonstrated to result in lower HFH risk than RVP by eliciting more physiological activation [53]. LBBP may have similar benefits in the prevention of PICM, but this possibility has not been confirmed in long-term randomized studies. Current knowledge gaps may be addressed by an ongoing multicenter randomized clinical trial (RCT) (OptimPacing, NCT04624763) aimed at comparing the long-term clinical outcomes of LBBP and RVP, and establishing whether LBBP might prevent AF and PICM in patients with AVB.
LBBP for Heart Failure
LBBP for HF and LBBB
As a physiological pacing mode for preserving synchrony in patients with an intrinsically narrow QRS, LBBP has been demonstrated to benefit patients with HF and wide QRS by achieving resynchronization. In 2017, Huang et al. first reported a case of LBBP in a 72-year-old women with 32% LVEF and LBBB [6]. After failures in LV lead implantation and LBBB correction by HBP, the patient underwent attempted LBBP for pacing the LBB beyond the conduction system block. LBBP achieved LBB correction at low output (0.5 volts/0.5 ms) and was found to ameliorate the clinical symptoms and echocardiographic values during a 1-year follow-up [6]. To date, several clinical studies have demonstrated this novel technique’s feasibility and applications in patients with HF [7, 14, 34, 54–63]. Zhang et al. have reported LBBAP in 11 patients with HF with reduced LVEF and LBBB. LBBAP has been found to significantly shorten the QRSd and LVAT after implantation, and to decrease NYHA class, plasma levels of BNP, and improve cardiac structure and function after an average of 6.7 months of follow-up [54]. Huang et al. have conducted a prospective, multicenter study of LBBP in patients with nonischemic cardiomyopathy (NICM) with LVEF less than 50% and LBBB [14]. LBBP was successfully performed in 61 of 63 patients (97%), and the QRSd narrowed from 169 ± 16 ms to 118 ± 12 ms (P < 0.001). After a 1-year follow-up, the LVEF had increased from 33 ± 8% to 55 ± 10% (P < 0.001); the LV end-systolic volume (LVESV) had decreased from 123 ± 61 mL to 67 ± 39 mL (P < 0.001); and 75% of these patients showed LVEF normalization (LVEF >50%) [14]. In addition, Ponnusamy et al. have reported that LBBP decreases the severity, and avoids worsening, of functional mitral regurgitation in patients with NICM and LBBB [62].
Whether LBBP might serve as an alternative to traditional BiVP-CRT is currently under investigation. Wang et al. and Li et al. have conducted two separate small-sample studies [56, 57]. During a 6-month follow-up, LBBP resulted in a greater decrease in QRSd, better recovery in echocardiographic parameters, and a higher response rate than BiVP in both studies. Similar trends have been observed in a non-randomized multicenter study by Chen et al. [60]. The first prospective RCT has recently been published [7]. Forty consecutive patients with NICM and LBBB were enrolled and randomized into an LBBP-CRT group or BiVP-CRT group. Greater improvements in LVEF (mean difference: 5.6%; 95% CI: 0.3–10.9; P = 0.039) and greater decreases in LV end-systolic volume (−24.97 mL; 95% CI: −49.58 to −0.36 mL) were observed in the LBBP-CRT group than the BiVP-CRT group. Favorable reduction in NT-proBNP and NYHA class and improvement in 6-minute walk distance and rates of CRT response were observed in the LBBP-CRT group. These encouraging results have demonstrated the superiority of LBBP-CRT in patients with NICM and LBBB. However, future large-scale RCTs are needed. Table 1 summarizes current RCTs comparing LBBP with BiVP.
Completed and Ongoing Randomized Clinical Trials of LBBP vs. BiVP in CRT.
| Status | Study | Year | Number | Intervention | Indication | Follow-up (months) | Outcomes/primary outcome measures |
|---|---|---|---|---|---|---|---|
| Completed | Wang et al. (LBBP-RESYNC, NCT04110431) | 2019–2021 | 20 in LBBP20 in BiVP | LBBP vs. BiVP | HF, NICM, LVEF ≤40%, typical LBBB | 6 | Greater LVEF improvement and a favorable decrease in LVESV and NT-proBNP with LBBP-CRT compared with BiVP-CRT |
| Liang et al. (NCT04505384) | 2020–2021 | 25 had both LV and LBBP lead | LBBP vs. BiVP | HF, LVEF ≤35%, LBBB | 0 | Superior acute electrical and mechanical resynchronization and hemodynamic improvement with LBBP compared with BiVP | |
| Ongoing | Atabekov et al. (NCT05769036) | 2023–2028 | 60 | LBBP vs. BiVP | HF, ICM, NICM | 24 | All-cause mortality or worsening of heart failure requiring unplanned hospitalization |
| Huang et al. (NCT05553626) | 2022–2025 | 160 | LBBP vs. BiVP | HF, LVEF <50%, II-III AVB | 12 | Change in LVEF | |
| Vinther et al. (NCT04409119) | 2020–2024 | 125 | HIS/LBB pacing vs. LV pacing | HF, LVEF ≤35%, LBBB | 6 | Change in LVESV | |
| Žižek et al. (NCT05467163) | 2023–2026 | 82 | CSP + AVNA vs. BiVP + AVNA | HF, LVEF <50%, symptomatic AF, narrow QRS scheduled for AVNA | 6 | Change in LVEF | |
| Žižek et al. (NCT05155865) | 2022–2024 | 60 | CSP vs. BiVP | HF, LVEF ≤35%, LBBB | 6–12 | Change in left ventricular volume, LVEF, heart failure class, pro-BNP, 6-minute walk test distance, EQ-5D index | |
| Fan et al. (NCT05549544) | 2022–2024 | 60 | LBBAP vs. BiVP | HF, LVEF <50%, permanent AF (AVNA or with slow ventricular rate) | 6–12 | Change in LVEF | |
| Waroux et al. (NCT05365568) | 2022–2024 | 170 | LBBAP vs. BiVP | HF, LBBB, QRS > 150 ms | 12 | Number of days elapsed from study procedure to first occurrence of one of the combined endpoint or to study close-out in the total patient population. | |
| Khan et al. (NCT05428787) | 2022–2024 | 284 | LBBAP + AVNA vs. BiVP + AVNA | HF, AF | 6 | Change in NT-proBNP | |
| Mont et al. (NCT05187611) | 2022–2024 | 130 | CSP vs. BiVP | HF, LVEF ≤35%, LBBB, QRS > 150 ms, AVB | 12 | Composite endpoint: all-cause mortality, cardiac transplantation, heart failure hospitalization, and LVEF improvement <5 points. | |
| Polanczyk et al. (NCT05572736) | 2022–2023 | 304 | CSP vs. BiVP | HF, LVEF ≤35%, LBBB | 12 | Non-inferiority of clinical benefit, hierarchical composite of all-cause death, any hospitalization for heart failure, any urgent heart failure visit, and left ventricular ejection fraction change at 12 months | |
| Atwater et al. (NCT05652218) | 2023–2024 | 20 | LBBP vs. BiVP | HF, LVEF > 35%, LBBB | 3 | Change in myocardial performance index |
AF, atrial fibrillation; AVB, atrioventricular block; AVNA, atrioventricular node ablation; BiVP, biventricular pacing; CRT, cardiac resynchronization therapy; CSP, conduction system pacing; HF, heart failure; LBBB, left bundle branch block; LBBP, left bundle branch pacing; LV, left ventricle; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; NT-proBNP, N-terminal proB-type natriuretic peptide; NYHA, New York Heart Association.
LBBP is superior to HBP in delivering CRT. HBP, the most physiological pacing strategy, has been demonstrated to achieve clinical and physical improvements similar to those with BiV-CRT. However, HBP has several limitations, such as difficulty in fixation of the pacing lead and a risk of loss of capture. Wu et al. have recruited 32 patients who underwent LBBP and compared their treatment outcomes with those in patients receiving HBP and BiVP for delivering CRT [59]. The absolute increase in LVEF (∆LVEF, +23.9% vs +24%, P = 0.977) and the rate of LVEF normalized to 50% (74.4% vs 70.0%, P = 0.881) between HBP and LBBP showed similarities, and both were higher than those in patients receiving BiVP (∆LVEF +16.7% and LVEF normalized rate of 44.9%, P < 0.005). Moreover, LBBP had stable pacing thresholds lower than those with HBP [59]. The higher success rate, lower pacing parameters, and greater clinical benefits with LBBP support its broader application prospects in the future [7, 59, 64–67].
Traditional BiVP-CRT including the CS LV lead has been well established in patients with HF with bundle branch block, but it carries risks of unsuccessful CS lead implantation and nonresponse to BiVP. Vijayaraman et al. have assessed the feasibility of LBBAP in patients in whom BiVP has failed [63]; LBBAP was successfully conducted in 200 patients, including 156 with CS lead implantation failure and 44 nonresponders. The treatment significantly narrowed the QRSd (170 ± 28 ms to 139 ± 25 ms (P < .001) and improved the LVEF from 29 ± 10% to 40 ± 12% (P < .001) during follow-up. This study supports that LBBP may serve as an alternative to failed traditional BiVP, although further investigations are necessary.
LBBP After AV Node Ablation
Wang et al. have reported on 52 patients who underwent successful CSP, including 44 with HBP and 8 with LBBP, combined with atrioventricular node ablation (AVNA). These patients had less medication use, lower incidence of HFH or death, and greater LVEF during follow-up than the group receiving medical therapy combined with ICD implantation (n = 31). AVNA combined with CSP appears to be a safe and feasible strategy that effectively improves cardiac function and prevents inappropriate shock caused by AF with a rapid ventricular rate [68]. Several studies have examined small samples of patients who underwent LBBP after AVNA [18, 34, 69]. Jin et al. have reported on 46 patients with successful LBBAP, and have indicated that the right atrial diameter, status of tricuspid regurgitation, and thickness of IVS may be associated with the success rate of LBBAP [70]. Pillai et al. have compared outcomes between HBP and LBBP in patients undergoing AVNA [71]. LBBAP had a higher success rate and fewer complications than HBP, whereas both preserved cardiac function in these patients.
A notable recent study by Huang et al. has focused on the long-term safety and efficacy of LBBP combined with AVNA. This single-center prospective study enrolled 99 patients and observed a 100% success rate of LBBP implantation and AVNA. The LVEF improved from 30.3 ± 4.9% to 47.3 ± 14.5% (P < 0.001) in patients with HFrEF, and from 56.3 ± 12.1% to 62.3 ± 9.1% (P < 0.001) in patients with HFpEF after a 1-year follow-up; moreover, further improvements were observed in 2-year and 3-year follow-up. A total of 86 patients who underwent LBBP and AVNA were successfuly propensity score-matched with 86 patients who had permanent HBP with AVNA. LBBP presented similar clinical outcomes to those of HBP, but achieved shorter procedural times, fewer ablation sites, and better pacing parameters. An additional ventricular lead was implanted in 54.7% of patients, and 97.7% of patients underwent LBBP and HBP for defibrillation, synchronization, or safety backup. During follow-up, five patients in the HBP group had HB capture loss, for the high capture thresholds of both HB and local cardiac tissue. Only 1 patient who underwent LBBP experienced an increased threshold of LBB capture [72].
LBBP for PICM
For the treatment of PICM, CRT has received only a class 2a recommendation in the recent guidelines [73]. Qian et al. have evaluated the efficacy of LBBP upgrade in 27 patients with PICM or with HF after RV pacing with LVEF ≥50% [74]. After a mean of 10.4 ± 6.1 months of follow-up, LVEF significantly increased from 40.3 ± 5.2% to 48.1 ± 9.5% in patients with PICM, whereas the NYHA functional class decreased from 2.5 ± 0.5 to 1.7 ± 0.8 (P < 0.0001). In patients with LVEF ≥50%, LVEF also increased after an upgrade (59.1 ± 4.2% vs. 61.4 ± 4.3%, P = 0.009). Thirteen patients each experienced at least one HFH in 6 months before the LBBP upgrade, whereas only four patients experienced an HFH after the upgrade. This study has demonstrated that LBBP is a feasible and effective upgrading strategy for patients with PICM, to achieve electrical and mechanical synchrony before further damage from dyssynchrony caused by RV pacing. A similar trend has also been demonstrated in several small studies [75–77]. Future large randomized trials comparing CSP and BiVP for upgrade of PICM are needed.
Complications of LBBP
LBBP related complications, such as perforation to LV, lead dislodgement, and an increased capture threshold, may be observed, despite their low likelihood [40, 69]. Chen et al. have reported a very low incidence of complications in 612 consecutive patients who underwent LBBP (10/612, 1.63%) [78]. In a study conducted by Jastrzębski et al., the rates of all complications and those associated with lead implantation were 11.7% and 8.3%, respectively, in 2533 patients receiving LBBAP [40]. Acute perforation to LV was the most common complication, accounting for 3.7%.
Indications for LBBP
Although emphasizing that LBBP is a promising treatment, the 2021 ESC guidelines for CRT do not make any recommendations for LBBP, because evidence from randomized trials focusing on the safety and efficacy of LBBP remain lacking [79]. However, LBBP use has rapidly increased. According to the 2023 HRS/APHRS/LAHRS guidelines released recently, several consensus recommendations for different pacing indications are outlined [80].
First, cardiac physiological pacing, including CRT, HBP, and LBBAP, is recommended for patients with permanent pacing indications and requiring substantial amounts of ventricular pacing to prevent PICM. The application class is 2a in patients with LVEF 36%–50% and 2b in patients with normal LVEF. However, in patients with LVEF >35% requiring minimal ventricular pacing, traditional RVP is reasonable (class 2a). Second, in patients with LVEF ≤35%, NYHA class II–IV, and LBBB with QRS duration ≥150 ms, CSP (including HBP and LBBAP) is recommended (class 2a) when BiVP cannot achieve CRT. Third, in patients with treatment-refractory AF undergoing AVNA, data on the efficacy of LBBAP are limited. Implanting an LBBAP lead might be a reasonable option (class 2b). Finally, although CRT with BiVP pacing is recommended (class 1), CSP can be beneficial for cardiac recovery in patients with PICM (class 2a) [80]. We summarized the current evidences of LBBP in bradycardia or HF in Table 2 and Table 3. The range of indications and levels of recommendation may change if stronger evidence is reported in the future.
Future Directions
Despite promising advances in LBBP, several important questions must be answered. Because LBBP is a novel technique with only 5 years of development, long-term and large-scale randomized clinical trials are essential, particularly those focusing on mortality and HF rehospitalization, to establish whether LBBP can serve as an alternative to traditional RVP for bradycardia and BVP for HF. Despite the superior resynchronization and greater physiological activation achieved by LBBP, whether the efficacy of LBBP and LVSP over longer time periods might differ has not been well studied. LBB-optimized CRT (LOT-CRT), on the basis of LBBP and combined with CS-LV pacing, has been proposed as a pacing strategy in patients with complex conduction system disease in whom LBBP did not decrease or only modestly decreased the QRSd. Although the feasibility of this approach has been reported, randomization is necessary to determine whether LOT-CRT is better than BiVP-CRT. Would a CS-LV lead be necessary in these patients? Few studies have reported the application of LBBP in patients with HF with mildly reduced EF (HFmrEF) or HF with preserved EF (HFpEF). Can LBBP improve clinical symptoms and reverse cardiac function before further deterioration? These questions remain to be explored.
Conclusion
LBBP is a safe and feasible pacing strategy in patients with RV pacing or CRT indications. A comprehensive assessment of LBB capture is necessary, and the hemodynamic and clinical outcomes of LBBP and LVSP should be compared in future studies. Although LBBP has been widely reported, prospective randomized trials and studies focused on its long-term efficacy are needed for different indications.
Studies Using LBBP for Bradycardia or a Variety of Indications.
| Study | Year | Type of study | Number | Success rate (%) | Indication | Outcomes |
|---|---|---|---|---|---|---|
| Chen et al. [25] | 2018 | Single center cohort | 20 | NA | SND, AVB, HF | LBBP produces much narrower QRSd than RVP, with comparable and stable pacing parameters. |
| Hasumi et al. [81] | 2019 | Single center cohort | 21 | 81% | AVB, failed HBP | LBP/peri-LBP is an alternative ventricular pacing method in patients with AVB with failure of His-bundle pacing, with <15 min procedure time and no complications. |
| Hou et al. [26] | 2019 | Single center cohort | 56 | NA | SND, AVB, AF with slow ventricular rate | LBBP, similarly to HBP, preserves better electrical and mechanical synchrony than RVSP, on the basis of analysis of QRSd, LVAT and mechanical synchrony, as evaluated by SPECT myocardial perfusion imaging. |
| Li et al. [27] | 2019 | Single center cohort | 33 | 90.90% | AVB | LBBAP is a promising strategy for physiological pacing in patients with AVB, with s stable threshold, narrow QRSd, preserved LV synchrony, and few complications. |
| Li et al. [28] | 2019 | Single center cohort | 87 | 80.50% | SND, AVB | LBBP results in a narrow QRSd (<120 ms), and can be safely and successfully performed in most patients. |
| Vijayaraman et al. [29] | 2019 | Single center cohort | 100 | 93% | SND, AVB, AVN ablation, HF (failed LV lead) | LBBAP is feasible, with a high success rate and low thresholds during acute follow-up. |
| Zhang et al. [30] | 2019 | Single center cohort | 23 | 87% | SND, AVB, HF | LBBAP achieves better electrical synchronization than RVP. |
| Cai et al. [31] | 2020 | Single center cohort | 40 | 90% | SND | LBBP preserves favorable LV synchrony and hemodynamic effects. |
| Chen et al. [32] | 2020 | Single center cohort | 250 | 94.80% | SND, AVB, HF | LBBP is feasible in patients with bradycardia; pacing parameters remained stable during an 18-month follow-up. |
| Das et al. [33] | 2020 | Single center cohort | 22 | 88% | SND, AVB | LBBAP results in a narrow QRS complex and BBB correction. |
| Qian et al. [34] | 2020 | Single center cohort | 185 | 95.90% | SND, AVB, AVN ablation, HF indications | Both LBBP and HBP result in favorable electrical synchrony and maintain normal cardiac function in patients with bradycardia. |
| Ravi et al. [69] | 2020 | Single center cohort | 57 | 97% | SND, AVB, AVN ablation, HF indications | Lead-associated complications was observed in 12.3% of successful LBBP procedures. |
| Vijayaraman et al. [46] | 2020 | Muticenter cohort | 26 | 93% | SND, AVB, AVN ablation, HF indications, post-TAVR | HPSP, including LBBP, is feasible in most pacing-indicated patients with post-TAVR. |
| Wang et al. [82] | 2020 | Single center randomized | 61 | 92.40% | SND, AVB, AF with slow ventricular rate | LBBAP results in better depolarization-repolarization reserve than RVP, and is associated with lower risks of ventricular arrhythmia and sudden cardiac death. |
| Li et al. [49] | 2020 | Single center cohort | 55 | 83.6% | SND, AVB, LVEF ≥40%, LBBB/RBBB | LBBAP completely or partially narrows the QRSd of RBBB and corrects LBBB in patients with bradycardia. |
| Chen et al. [78] | 2021 | Single center cohort | 612 | NA | SND, AVB, AVN ablation, HF indications | The incidence of LBBP procedure-associated complications is low, with no adverse clinical outcomes. |
| Gul et al. [35] | 2021 | Muticenter cohort | 84 | 95% | SND, AVB, AVN ablation, HF indications, post vascular interventions | LBBAP is feasible and safe in patients with prior interventions for valvular heart disease. Two patients had loss of LBB capture within 1 month. |
| Hu et al. [36] | 2021 | Single center cohort | 25 | 88% | AVB | LBBP is indicated as a physiological pacing therapy for patients with AVB. |
| Li et al. [37] | 2021 | Muticenter cohort | 246 | 95.50% | AVB | LBBAP, compared with traditional RVP, might be a favorable pacing strategy to decrease the risk of HF events or upgrade to biventricular pacing in patients with high burden of VP. |
| Ponnusamy et al. [83] | 2021 | Single center cohort | 11 | 91% | ≥80 years of age, SND, AVB, HF | LBBP is safe and provides effective resynchronization therapy in patients ≥80 years of age. |
| Ponnusamy et al. [38] | 2021 | Single center cohort | 99 | 94% | SND, AVB, AVN ablation, HF indications | LBBP is as safe and effective a strategy as physiological pacing and might overcome the limitations of HBP. |
| Su et al. [18] | 2021 | Single center cohort | 632 | 97.80% | SND, AVB, AVN ablation, HF indications | During a mean 18.6 ± 6.7 months of follow-up, LBBP was feasible, with high success and low complication rates. |
| Wang et al. [84] | 2021 | Single center cohort | 52 | 94.20% | Persistent AF, LVEF > 35% | LBBAP narrows the QRSd, slightly decreases the rate of death and HFH, and reverses LA diameter in patients with persistent AF. |
| Zhu et al. [39] | 2021 | Single center cohort | 406 | 92.60% | SND, AVB | LBBAP was associated with a decrease in LAD, whereas RVP decreased LVEF during a mean follow-up of 13.6 ± 7.8 months. LBBAP may result in greater preservation than RVP. |
| Rademakers et al. [43] | 2021 | Single center cohort | 100 | 83% | SND, AVB, AVN ablation, HF indications | The success rate of LBBP in CRT-eligible patients tended to be low (69%). The pacing parameters remained stable, with no complications during a 6-month follow-up. |
| Niu et al. [47] | 2021 | Single center cohort | 20 | 95.20% | AVB, post-TAVR | LBBP and HBP achieved shorter paced QRSd and better LVEF and LVEDD than RVP in patients who developed AVB after TAVR. |
| Hu et al. [41] | 2022 | Single center cohort | 91 | NA | SND, AVB, AF with slow ventricular rate | Twenty-eight patients had unchanged or decreased LVEF after an 18-month follow-up. Baseline LVEF, ΔPaced QRSd, and corrected longitudinal distance were negatively associated with LVEF improvement. |
| Jastrzębski et al. [40] | 2022 | Muticenter cohort | 2533 | 89.60% | SND, AVB, AF, HF indications | LBBAP is feasible for bradycardia and HF as a primary pacing strategy. HF history, broad QRS, and wide LVEDD at baseline are independent predictors of LBBAP failure. The complication rate of LBBAP was 11.7% |
| Okubo et al. [42] | 2022 | Single center cohort | 43 | NA | AVB, HF (EF > 40%) | LBBAP maintains physiological activation of ventricles and decreases NT-proBNP in patients with HfpEF or HfmrEF. |
| Raymond-Paquin et al. [44] | 2022 | Muticenter cohort | 364 | 93% | SND, AVB | Procedural success rates were similar among indications. Four acute LBBAP lead dislodgments were observed. |
| Sharma et al. [45] | 2022 | Muticenter cohort | 321 | NA | SND, AVB | Patients, who underwent LBBAP and with ventricular pacing burden >20%, had lower risk of primary outcome than those underwent RVP. |
AF, atrial fibrillation; AVB, atrioventricular block; AVNA, AV node ablation; CRT, cardiac resynchronization therapy; HBP, His bundle pacing; HF, heart failure; HFrEF, HF with reduced ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFH, heart failure hospitalization; HPCSP, His-Purkinje conduction system pacing; LA, left atrium; LAD, LA dimension; LBBB, left bundle branch block; LBBP, left bundle branch pacing; LV, left ventricle; LVEF, left ventricular ejection fraction; LVEDD, LV end-diastolic dimension; NA, not available; NT-proBNP, N-terminal proB-type natriuretic peptide; NYHA, New York Heart Association; RVP, right ventricular pacing; RVSP, right ventricular septal pacing; SND, sinus node dysfunction; TAVR, transcatheter aortic valve replacement.
Studies Using LBBP for HF.
| Study | Year | Type of study | Number | Success rate (%) | Indication | Outcomes |
|---|---|---|---|---|---|---|
| Zhang et al. [54] | 2019 | Single center cohort | 11 | NA | HFrEF + LBBB, CRT-eligible | LBBAP is a new strategy for CRT to correct LBBB, achieve LV synchrony, and improve cardiac function and clinical symptoms in HF patients with reduced LVEF and LBBB. |
| Wang et al. [68] | 2019 | Single center cohort | 8 | NA | AF + HF (ICD and AVNA indication) | HPSP (HBP/LBBP) + AVNA is safe and feasible in patients with AF and HF for preventing inappropriate shocks and improving LV function. |
| Huang et al. [14] | 2020 | Muticenter cohort | 63 | 97% | CRT-eligible, NICM, LBBB, LVEF≤50% | LBBP is feasible and effective in achieving electric resynchronization, improvement in cardiac structure and function, and correction of LBBB in patients with NICM with LBBB. LBBP may be superior to HBP for CRT, because of its stable and low pacing parameters. |
| Qian et al. [34] | 2020 | Single center cohort | 185 | 95.90% | SND, AVB, AVN ablation, HF indications | Both LBBP and HBP can reverse cardiac structure and improve function in patients with HF. |
| Li et al. [55] | 2020 | Single center cohort | 25 | NA | CRT-eligible | LBBAP can serve as an alternative to CRT in patients with failure of LV lead placement and a prior option in patients with LBBB and HF. |
| Li et al. [57] | 2020 | Single center cohort | 37 | 81.1% | CRT-eligible, LBBB, LVEF≤35% | LBBAP delivers a greater decrease in QRSd, and increase in LVEF and super echocardiographic response, than BiVP for CRT. |
| Wang et al. [56] | 2020 | Single center cohort | 10 | NA | CRT-eligible, LBBB, LVEF≤35% | Ten patients who underwent LBBAP were matched through propensity score matching with 30 patients who underwent BiVP. LBBAP was demonstrated to better optimize electrical synchrony and improve cardiac function. |
| Vijayaraman et al. [58] | 2021 | Muticenter cohort | 325 | 85% | CRT-eligible, LVEF≤50% | After a mean follow-up of 6 ± 5 months, 72% and 73% of patients had clinical and echocardiographic responses, respectively. Baseline LBBB and LVEDD independently predicted the response. |
| Wu et al. [59] | 2021 | Single center cohort | 32 | NA | CRT-eligible, LBBB, LVEF≤40% | LBBP and HBP resulted in similar improvements in symptoms and LV function: 74.4% and 70.0% of patients had normalized LVEF, respectively, both of which were higher than those in the BVP group (44.9%). |
| Qian et al. [74] | 2021 | Single center cohort | 27 | 93% | PICM, symptomatic HF with LVEF ≥50% | Upgrading to LBBP improves electrical synchrony and reverses acute LV remodeling, according to SPECT in patients with PICM. Patients with PICM and those with HF not meeting standards for PICM benefit from LBBP, thus avoiding further RV pacing damage. |
| Yang et al. [75] | 2021 | Single center cohort | 5 | NA | PICM | HPSP improves cardiac function and reverses LV remodeling in patients with PICM with or without AF. |
| Ye et al. [76] | 2021 | Single center cohort | 20 | 95% | PICM | LBBP is a reasonable alternative to CRT via CS-LV lead in patients with PICM. |
| Chen et al. [60] | 2022 | Muticenter cohort | 49 | 98% | CRT-eligible, LBBB, LVEF≤35% | LBBP-CRT achieved better improvement in LVEF, a higher super-response rate, and a lower threshold than BVP-aCRT. |
| Hua et al. [61] | 2022 | Single center cohort | 21 | NA | CRT-eligible, LBBB, HF | LBBAP had lower thresholds and shorter procedure times than BiVP. Both LBBAP and BiVP improved symptoms and cardiac function. |
| Jin et al. [70] | 2022 | Single center cohort | 56 | 0.82 | AF + HF, AVNA indication | LBBAP + AVNA is safe and effective in patients with AF and HF. The right atrial diameter, degree of TR, and IVS may affect the success rate of LBBAP. |
| Pillai et al. [71] | 2022 | Single center cohort | 50 | NA | AF, AVNA indication | AVNA combined with CSP preserves LV function; LBBAP is particularly associated with a high success rate and diminished complications. |
| Ponnusamy et al. [62] | 2022 | Single center cohort | 73 | 94% | NICM, LBBB | LBBP decreases functional mitral regurgitation severity in most patients with NICM and LBBB. No worsening of FMR was observed in any patients. |
| Vijayaraman et al. [63] | 2022 | Muticenter cohort | 200 | 94% | CRT-eligible, CV lead failure, nonresponse to BVP | LBBP is a feasible alternative to CRT in patients with failed BVP due to unsuccessful CS-LV lead implantation or nonresponse to BiVP-CRT. |
| Vijayaraman et al. [85] | 2022 | Muticenter cohort | 171 | NA | CRT-eligible, LVEF≤35% | In this large cohort of CRT-eligible patients, CSP (including HBP and LBBAP) resulted in greater LVEF improvement and significantly lower HFH than BVP for CRT. |
| Wang et al. [7] | 2022 | Muticenter randomized | 20 | 90% | CRT-eligible, NICM, LBBB, LVEF≤40% | In this randomized trial, LBBP-CRT showed significantly higher LVEF improvement, and greater decreases in LVESV and NT-proBNP, than BiVP-CRT in patients with NICM and LBBB. |
| Cai et al. [72] | 2022 | Single center cohort | 99 | 100% | AF + HF, AVNA indication | LBBP and AVNA are safe and feasible. After PS matching, LBBP had similar benefits, better pacing parameters and lower risks of complications than HBP. |
| Liang et al. [67] | 2022 | Single center randomized | 25 | 96% | CRT-eligible, LVEF≤35%, LBBB | LBBP delivers superior acute electrical and mechanical resynchronization and hemodynamic improvement to BiVP. |
| Rademakers et al. [77] | 2022 | Single center cohort | 20 | 100% | PICM | LVEF increased from 32% ± 6% to 47% ± 8%, and NYHA class decreased from 2.8 ± 0.4 to 1.4 ± 0.5 at 6 months. No pacing-associated complications, death, or HFH occurred. |
| Vijayaraman et al. [51] | 2022 | Muticenter cohort | 121 | 88% | HF, LVEF <50%, RBBB, CRT-eligible or ventricular pacing indication | LBBAP is a feasible strategy in patients with HF and RBBB for CRT or physiological ventricular pacing. |


