Significance Statement Carcinoid heart disease (CHD), a rare form of valvular heart disease, is associated with poor prognosis in patients with carcinoid syndrome. The initial presentation of CHD is often subtle and insidious. Cardiac biomarkers and imaging aid in screening and early diagnosis. A multidisciplinary team of cardiologists, oncologists, cardiac surgeons and gastroenterologists is necessary for comprehensive management of CHD.
Introduction and Epidemiology
Neuro-endocrine tumors (NET), which were once considered relatively rare, are a heterogeneous group of slow-growing neoplasms originating mainly from enterochromaffin cells. The incidence and prevalence of NETs have continued to increase, owing to increased diagnosis of early stage tumors and possibly stage migration [1]. In the United States, the age-adjusted annual incidence of NETs rose 6.4-fold over the past 20 years, to 6.98 per 100,000 people [1, 2]. Carcinoid tumors are well differentiated NETs that typically originate from the gastro-intestinal tract or bronchopulmonary system, and can cause carcinoid syndrome through the secretion of various vasoactive substances.
Carcinoid heart disease (CHD) eventually occurs in as many as 50% of patients with carcinoid syndrome and can be the primary disease manifestation in as many as 20% of these patients [3, 4]. Valvular involvement in CHD often results in heart failure, and is a major cause of morbidity and mortality [5]. The estimated 3-year survival is 31%, which is approximately half that in people with carcinoid syndrome without CHD [5, 6]. Sufficient evidence indicates that early diagnosis and treatment of CHD improve symptoms and longevity [7]. To achieve early diagnosis and treatment, a multidisciplinary team approach, involving oncologists, cardiologists, endocrinologists, anesthesiologists and cardio-thoracic surgeons, is required [8].
In this article, we review the current literature on pathophysiology, clinical presentation, epidemiologic characteristics, diagnostic tools and novel treatment strategies in CHD management.
Pathophysiology
NETs are typically indolent and secrete a myriad of vaso-active compounds, such as serotonin, 5-hydroxy tryptophan (5-HTE), histamine, bradykinins, growth factors and prostaglandins. These compounds are often deactivated in the liver and lungs, thus protecting the heart. Cardiac involvement, typically in the right side of the heart, occurs when: a) tumors metastasize to the liver, thus bypassing its deactivating mechanisms, or b) the overwhelming burden of secreted vasoactive compounds exceeds the degradation capability of the liver and lungs. In addition, left sided involvement occurs with bronchopulmonary carcinoid or in the presence of a right to left intracardiac shunt, such as a patent foramen ovale or atrial septal defect [3, 9, 10]. In these scenarios, the heart is exposed to paraneoplastic effects of circulating serotonin, growth factors, and other vasoactive peptides, as well as cross-talk among them [11]. Consequently, pro-inflammatory cytokines, such as transforming growth factor beta, are generated through the stimulation of serotonin receptors in the heart (primarily 5HT2B) [10–12]. Consequently, plaque-like fibrous tissue and extracellular matrix deposition occur in the endocardium of valvular cusps, leaflets, tendinous cords, papillary muscles, cardiac chambers, and occasionally the intima of the pulmonary arteries or the aorta [10]. Eventually, these deposits cause structural deformation and progressive dysfunction of the pulmonary and tricuspid valves, right ventricular dysfunction and heart failure over the course of months [4].
Experimental in vivo and in vitro studies that have advanced understanding of pathogenesis are shown in Supplementary Table S1 [13–17].
Among these vasoactive substances, the strongest evidence supports the critical roles of serotonin and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) in the pathogenesis, diagnosis and prognosis of CHD [3, 18–21]. In a large cross-sectional study (n=187), patients with CHD (n=37) have been found to have significantly higher levels of serum chromogranin A/B, neurokinin-A and 5-HIAA than those without CHD (187-37: 150 patients with NET and/or carcinoid syndrome but no cardiac involvement). NT-pro-BNP (AUC: 0.82; CI: 0.74–0.90, P<0.0001) and 5HIAA (AUC: 0.85; CI: 0.78–0.92, P<0.0001) have been identified as the best predictive markers for CHD development [18]. In contrast, no significant correlation has been detected between transforming growth factor beta and fibroblast growth factor levels and CHD [19]. Furthermore, insights from the pathophysiology of drug-induced valvular heart disease have reinforced the role of serotonin in CHD pathogenesis. Ergot derivatives, such as ergotamine; methysergide; and dopamine antagonists, such as pergolide and cabergoline, are structurally similar to serotonin and are believed to cause valve disease by stimulating serotonin 5-HT 2B receptors [22–24].
Clinical Presentation and Diagnosis
The initial presentation of CHD is often subtle and insidious. On an average, CHD develops approximately 2 years after the initial diagnosis of carcinoid syndrome [6]. The symptoms and signs are usually right sided heart failure, such as worsening dyspnea, peripheral edema, ascites with physical examination revealing jugular venous distention, palpable RV impulse, pulsatile hepatomegaly, and audible tricuspid and pulmonic regurgitation murmurs. Rarer presentations include coronary artery spasm in patients with non-occlusive coronary artery disease, direct tumor metastasis to cardiac muscle, left sided heart failure, and supraventricular and ventricular arrhythmias [3, 25–27].
Laboratory Diagnosis
A 24-hour urine measurement of 5-HIAA >300 mmol is an important biomarker in CHD prediction and prognostication. In a meta-analysis by Joish et al. [28], every 10-unit increase in urinary 5-HIAA levels has been found to correspond to an 11.8% (95% CI 8.9%–17.0%) increase in 12-month mortality. Plasma 5-HIAA levels may serve as a surrogate for urine measurement, but serotonin rich foods and commonly used medications can result in false positive (e.g., acetaminophen, nicotine and caffeine) and false negative (e.g., aspirin) results. NT-pro-BNP levels are useful for screening (levels greater than 260 pg/mL can diagnose CHD with a sensitivity of 92% and a specificity of 91%), and correlate well with CHD disease severity, as compared with 5-HIAA [29]. Hence, in addition to clinical evaluation, serum NT-Pro BNP measurement is recommended every 6–12 months in all patients with metastatic NETs every 6–12 months [8]. Chromogranin A and activin A lack specificity in the detection of CHD [30, 31].
Imaging
Two-dimensional transthoracic echocardiography (TTE) remains the primary imaging modality enabling both morphological and functional assessment. The recently published American College of Cardiology consensus statement recommends echocardiography screening in all patients with suspected CHD [8]. The current literature also advocates for repeat TTE every 6 months in patients with mild disease and every 3 months in patients with moderate-severe disease [8, 32]. Pathognomonic plaque-like fibrous deposits involving the valvular and sub-valvular apparatus decrease the dynamic motion of the leaflet, thereby leading to a characteristic “board-like” pattern during diastole. In severe cases, the tricuspid valve is fixed and retraced, and does not coapt, thus resulting in a distinctive “dagger-shaped” Doppler profile [32] (Figure 1). Advanced echocardiographic techniques, such as three-dimensional echocardiography and transesophageal echocardiography, may be useful in assessing pulmonic and tricuspid valves in some cases [33]. Cardiac magnetic resonance imaging (MRI) and cardiac computed tomography (CT) are other valuable adjuncts that can be used when echocardiographic image findings are equivocal [34]. Surgical planning and technique can be optimized by using cardiac MRI and cardiac CT, because they can also quantify right ventricular volumes and function, and identify myocardial metastasis and their relation to coronary arteries [32, 34].
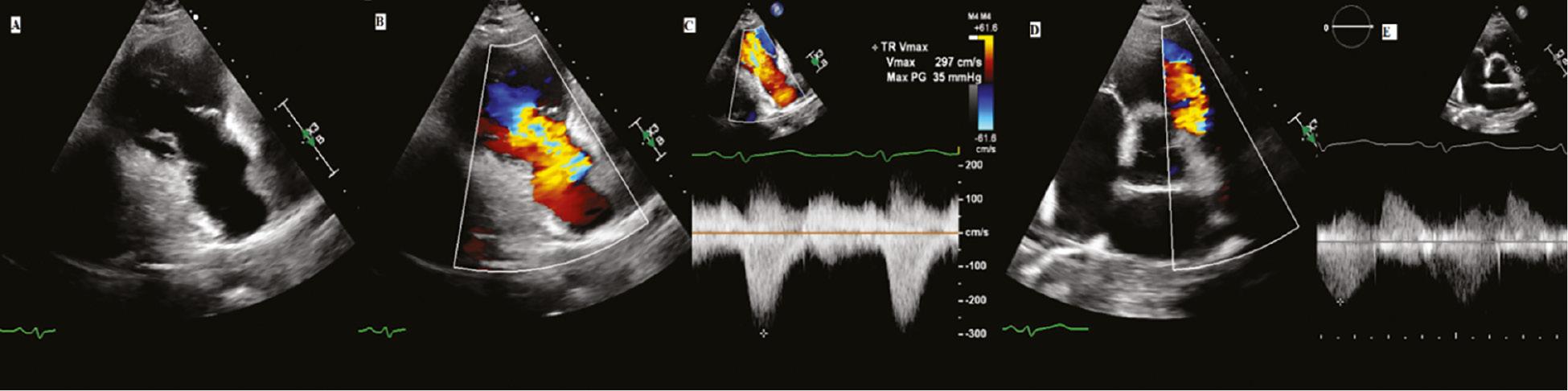
Echo Diagnosis of Right Sided Carcinoid Heart Disease.
A) RV inflow view of the tricuspid valve shows thickened leaflets that are open during mid systole due to significant fibrosis and adhesions. B) Color Doppler shows severe wide open tricuspid regurgitation. C) Continuous wave Doppler shows classic “V cut off sign”. D) Severe pulmonic regurgitation by Color Doppler in parasternal short axis view at aortic valve level. E) Continuous wave Doppler depicts the short pressure half time of pulmonary regurgitation jet.
Several echocardiographic scoring systems have been developed to diagnose CHD and determine its progression (Table 1). The current literature recommends the use of scoring systems focusing on valve anatomy and regurgitation for screening, and reserving more complex scoring systems for monitoring the progression of CHD [37].
Echocardiographic scoring systems validated in carcinoid heart disease.
| Name | Number of patients and type of study | Parameters | Outcomes | Limitations |
|---|---|---|---|---|
| Denney et al. [35] 1998 | 23; prospective single center cohort study | Carcinoid valvular heart disease percentage (CVHD%) score: | ||
| Moller et al. [21]. 2003 | 71; retrospective single center cohort study | Cardiac score: tricuspid/pulmonic: valve anatomy, regurgitation severity, right ventricular (RV) size/systolic function, presence/absence of diastolic forward flow in the pulmonary artery and systolic flow reversal in hepatic veins | ||
| Bhattacharyya et al. [29]. 2008 | 200; prospective single center cohort study | Carcinoid score: tricuspid/pulmonary valve: thickening, mobility, morphology, stenosis/regurgitation severity, RV diameter and function |
| |
| Westberg et al. [5], 2001 | 62; cross-sectional single center study | Tricuspid/pulmonary: Doppler estimation of valvular regurgitation and flow profiles | ||
| Mansencal et al. [36], 2010 | 80; prospective single center cohort study |
|
Management
A comprehensive approach involves addressing the tumor burden, progression of valve disease, heart failure symptoms and timely surgical intervention in selected individuals (Figure 2) [8].
Pharmacotherapy for CHD Related Heart Failure
Owing to the rarity of this condition, standard heart failure medications, such as beta blockers, angiotensin-converting enzyme inhibitors (ACE-I) and angiotensin-2 receptor antagonists, digoxin and vasodilators, have not been studied in the management of CHD related heart failure. Similarly to other heart failure states, diuretics/aldosterone antagonists can be used to optimize fluid status. Caution is required to avoid intravascular volume depletion in isolated right heart failure, which can further decrease cardiac output.
Management of Carcinoid Syndrome in CHD
The mainstay of management of CHD is decreasing tumor metabolites. Somatostatin analogues (SSAs) designed to decrease circulating tumor metabolites are the standard of care for carcinoid syndrome. Long-acting SSAs, such as octreotide LAR or lanreotide/paserotide, are increasingly being used in asymptomatic patients, owing to their demonstrated efficacy in retarding tumor growth and preventing the progression of CHD [38, 39]. Therapy is guided by 5-HIAA levels, and a level <300 mmol/24 hrs is the treatment goal in CHD. These medications achieve symptomatic improvement as well as biochemical response in 60%–70% of patients but have not been shown to reverse existing valvular heart disease [8]. Patients with refractory symptoms can be treated with higher doses/increased frequency of SSAs or with short acting SSAs in addition to depot formulations. If symptoms persist, other options include the following:
Interferon alpha (INF-α): When used along with SSAs, IFN-α may control symptoms and achieve a biochemical response. However its use is limited by substantial adverse effects [40].
Peptide receptor radionuclide therapy (PRRT): This radioisotope therapy, used in combination with octreotide, has been shown to elicit a symptomatic response and antiproliferative activity, mainly in advanced NET and somatostatin-receptor positive lesions [41]. However, concomitant use of peptide and fluid infusions with PPRT can complicate care in patients with decompensated heart failure, thereby resulting in fluid overload.
Telotristat: This oral tryptophan hydroxylase inhibitor with SSAs ameliorates diarrhea associated with carcinoid syndrome and urinary 5-HIAA levels [42].
Everolimus: This oral inhibitor of mammalian target of rapamycin (mTOR) and octreotide has been found to prolong progression free survival in patients with advanced NETs associated with carcinoid syndrome, in the RADIANT-2 trial [43].
Capecatibine/temozolomide (CAPTEM): This combination chemotherapy has shown efficacy in pancreatic neuroendocrine tumors. Papaxoinis et al., in a retrospective analysis, have studied the efficacy of CAPTEM chemotherapy in the management of advanced pulmonary carcinoid syndrome. Although the response rate was relatively low (18%), the progression free survival was longer (9 months) than that with temozolomide monotherapy [44].
Non-cardiac Interventions to Decrease Tumor Burden
Hepatic interventions can aid in decreasing vasoactive substances and tumor burden. Transcatheter arterial embolization and hepatic cytoreductive surgery are two effective treatment options. Transcatheter arterial embolization of hepatic lesions is a less invasive approach that can elicit a clinical response in a substantial number of patients [45]. Hepatic cytoreductive surgery is a more invasive option. However, the peri-operative mortality and morbidity associated with hepatic intervention can be significantly elevated in patients with right heart dysfunction and congestive heart failure. Hence, in such cases, cardiac valve repair and optimization of right heart function may be necessary before hepatic surgery.
Cardiovascular Surgery
Valvular intervention is the only definitive therapy for the treatment of symptomatic CHD. When performed in a timely manner in experienced centers, this therapy improves symptoms and prognosis [7]. Valve surgery, generally replacement, should be considered in patients with severe symptomatic valvular stenosis/regurgitation; asymptomatic patients with ventricular dysfunction and well controlled systemic disease; or patients before hepatic cytoreductive surgery. Cardiac surgery requires careful patient selection and planning, because most patients require multivalvular surgery, often in the setting of right ventricular dysfunction. Tricuspid and/or pulmonary valve replacement surgery can be performed together with closure of a patent foramen ovale and removal of intracardiac carcinoid metastasis. Studies reporting outcomes with surgical treatment of CHD are described in Table 2. In the largest operative experience published to date (n=240), 73% of patients required multivalvular surgery (two or more valves), and 16% of patients required left sided valve operation; the cumulative 30-day mortality has been 9% over the past 3 decades [50]. However, early operative mortality has decreased over time with increasing surgical experience, from 29% between 1985 and 1994; to 7% over the next 10 years; and finally to 5% from 2005 onward. Older age (HR: 1.55, P<0.001), higher New York Heart Association functional class (HR: 1.53, P<0.004) and non-linear variability of creatinine (HR: 0.81, P=0.83) have emerged as independent predictors of overall mortality. Interestingly, the number of valves replaced has not been found to be associated with mortality [50]. The choice of valve prosthesis is a matter of debate; however, bio-prosthetic valves are favored over mechanical valves for several reasons [8, 51]. Bioprosthetic valves are associated with a lower risk of valve thrombosis, do not require long term anticoagulation and are associated with longer than expected survival in most patients with CHD [52]. However, they are not free from complications. For instance, in a study by Nguyen et al., three patients required surgical reintervention after valve thrombosis, and recurrent carcinoid fibrosis of the bioprosthetic valve was identified in three patients. Mechanical valves can be appropriate in patients who already require long term anticoagulation with warfarin for other reasons, or in young patients who have ovarian carcinoids and favorable prognosis after valve surgery and oophorectomy [53]. After surgery, TTE should be performed periodically to detect prosthetic dysfunction. Transcatheter therapies can then be used to treat bioprosthetic valve dysfunction. Continued intensive control of NETs and targeting a 5-HIAA level <300 mmol/L remain mainstays of treatment after valve surgery.
Studies describing surgical outcomes in patients with CHD.
| Name | Number of patients | Type of study | Outcomes | Limitations |
|---|---|---|---|---|
| Connolly et al. [7], 1995 | 26 | Cross-sectional single center observational study | ||
| Bhattacharya et al. [46], 2011 | 22 | Retrospective single center cohort study | ||
| Arghami et al. [47], 2010 | 7 | Retrospective single center cohort study | ||
| Mokhles et al. [48], 2011 | 19 | Retrospective single center cohort study | ||
| Connolly et al. [49], 2015 | 195 | Retrospective single center cohort study | ||
| Nguyen et al. [50] 2018 | 240 | Retrospective and prospective cohort study |
|
Peri-operatively, patients are at risk of a life-threatening condition carcinoid crisis. Carcinoid crisis is mediated by a sudden release of vasoactive mediators into the intravascular circulation, and can result in hemodynamic instability and arrhythmias. This condition can be prevented by intravenous octreotide infusion at a rate of 50–100 mcg/hr, starting 12 hours before the procedure and continuing until the patient is stable postoperatively [54]. If a patient develops symptoms/signs of carcinoid crisis, treatments include administration of larger doses of octreotide (approximately 1000 micrograms), increasing the infusion rate to 100–200 mcg/h, and administering intravenous fluids. The complexity of this disease necessitates peri-operative planning by a multidisciplinary team of anesthesiologists, oncologists, cardiologists and cardiac surgeons.
Recent evidence suggests that correction of right sided valve disease and resultant congestive hepatopathy may improve noncardiac outcomes. In a series of 17 patients, 5-HIAA levels have been found to decrease by an average of 31% after right sided valve surgery, and to correlate with decreases in IVC diameter and bilirubin levels [55]. Whether this benefit in improved hepatic function and decreased tumoral production of serotonin improves long term prognosis is unknown.
Treatment of Arrhythmias in CHD
Cardiac arrhythmias can complicate the treatment of CHD. Owing to the occurrence of substantial atrioventricular valve regurgitation, atrial dilation and fibrosis are common. In a recently published series of 17 patients with carcinoid tumors (not restricted to those with heart involvement) who underwent catheter ablation, 76.5% had atrial arrhythmias [56]. In this series, ablation was safely performed without mortality in the peri-operative period. However, one case of carcinoid crisis occurred after ablation, thus highlighting the need for careful peri-operative management.
Bradyarrhythmias due to complete heart block can occur postoperatively, with an incidence of 12.5%, according to a recently published series [57]. Pacemaker implantation is safe and can be accomplished with minimal complications. The risk of heart block persists until 6 months after right sided valve surgery; consequently, some authors have recommended prophylactic placement of epicardial leads after surgery [58]. Heart block can rarely occur in the setting of intracardiac metastasis from a carcinoid tumor [59].
Rarer Manifestations of Carcinoid Heart Disease Without Valvular Involvement
Although rare, carcinoid syndrome can affect the heart beyond heart valve involvement, as follows:
Cardiac tumor metastasis is generally considered rare and can occur without valvular involvement. Before the era of advanced multimodal imaging, the incidence was approximately 4% [62]. In a recent series using cardiac CT imaging, the incidence has been found to be as high as 9% [63]. Although patients may remain asymptomatic, sudden death due to heart block or arrhythmias can occur [59]. Metastatectomy is usually considered when cardiac surgery is otherwise being performed to correct valvular diseases. In addition, metastatic carcinoid syndrome can induce coronary vasospasm, particularly in patients with non-obstructive coronary artery disease [25].
Prognosis of Carcinoid Heart Disease
The overall tumor burden (clinically determined by the degree of serotonin metabolite elevation) and the severity of cardiac structural abnormalities are important prognostic factors in CHD [5]. Cardiac valve replacement appears to improve prognosis.


