Introduction
Acute coronary syndrome (ACS) is a critical cardiovascular disease and the main contributor leading to death in people with cardiovascular disease. ACS includes ST segment elevation myocardial infarction (STEMI), non-STEMI and unstable angina. Although the proportion of patients with ACS receiving percutaneous coronary intervention (PCI) is increasing, the occurrence of adverse cardiovascular events is inevitable [1]. A previous study has reported that the incidence of major adverse cardiovascular events (MACE) in patients with ACS treated with PCI is approximately 10% within 1 year [2]. Thus, early risk stratification for patients with ACS after PCI is clinically important to decrease the occurrence of adverse events after PCI.
The ACEF score is composed of three factors: age, serum creatinine and ejection fraction. This risk score, used to predict the operative mortality of patients undergoing coronary artery bypass grafting, was first developed and validated by Ranucci et al. in 2009 [3]. The advantage of this simplified risk model is that it avoids the “overfitting” problem arising from the inclusion of many independent variables. Wykrzykowska et al. have evaluated the ACEF scores of patients receiving PCI in the LEADERS trial and found that this score may be a simple method for predicting the risk of myocardial infarction and mortality in patients treated with PCI [4]. However, a previous study has suggested that combining this risk score with clinical variables provides more reliable accuracy in predicting the clinical outcomes of patients after PCI [5].
Fibrinogen (FIB), an important component of the clotting pathway, binds receptors on the platelet membrane, thus resulting in the formation of acute coronary thrombosis [6]. Peng et al. have reported that the plasma FIB level at admission is an independent predictor of cardiac mortality in patients with coronary artery disease [7]. Ang et al. and Mahmud et al. have shown that elevated baseline levels of FIB, a reactant in the acute phase of inflammation, are associated with long-term MACE after PCI [8, 9]. The purpose of this research was to determine whether the ACEF score combined with FIB might improve the prognostic value for patients with ACS after PCI.
Methods
Study Populations and Study Design
All patients were enrolled at the Heart Center of Beijing Chaoyang Hospital, Capital Medical University. A total of 290 patients who underwent angiography for ACS were recruited between May 2019 and December 2019. The diagnostic criteria for ACS were clinical symptoms, elevated cardiac biomarkers (troponin-I or creatine kinase MB), typical electrocardiogram changes and coronary angiography. The exclusion criteria were as follows: 1) age <18 years old; 2) a history of coronary artery bypass grafting or hybrid coronary-revascularization during the hospitalization; 3) contraindications for, or unsuitability of, PCI; and 4) incomplete data for calculating the ACEF score.
Blood samples were collected from each patient in a fasting state on the first morning after admission. All laboratory indices, including FIB, leukocytes, platelets, troponin I, creatine kinase-MB (CK-MB), type b natriuretic peptide (BNP), C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), D-dimer, creatinine, high-density lipoprotein cholesterol (HDL-c), low-density lipoprotein cholesterol (LDL-c) and triglycerides, were determined at our clinical laboratory center. All patients were examined by echocardiography. All participants underwent coronary angiography and optimized treatments. The baseline and clinical characteristics were gathered from the medical record system.
The ACEF score was calculated with the following formula: age/left ventricular ejection fraction +1 (if creatinine was >2.0 mg/dL). The SYNTAX score was calculated on the basis of coronary angiography. This score can be a useful tool for assessing the severity of coronary artery lesions (http://syntaxscore.com/). The GRACE risk score is a practical tool for risk assessment regarding in-hospital outcomes (http://www.outcomes-umassmed.org/GRACE/).
Statistical Analysis
Categorical variables are reported as frequencies (percentages), and continuous variables are reported as mean ± standard deviation, or median and interquartile range (25th and 75th percentiles). Categorical variables were analyzed with chi-square test or Fisher’s exact test. Continuous variables were tested for differences with one-way ANOVA or the Kruskal-Wallis H test. Continuous variables were tested for normal distribution with the Kolmogorov–Smirnov test.
All patients were systematically followed up through medical records or telephone calls. The primary clinical endpoint was the occurrence of MACE, including all-cause death and rehospitalization for cardiovascular diseases. All relevant clinical factors for MACE were included in the logistic regression analysis. We aimed to assess whether combining the ACEF score with FIB increased the prognostic value. Receiver operating characteristic (ROC) curves were constructed to assess the prognostic value of the risk scores to predict MACE. Net reclassification improvement (NRI) and integrated discrimination improvement (IDI) were used to compare the ability of the new risk score versus other scores to reclassify the risk of MACE. Cumulative event rates were calculated on the basis of Kaplan-Meier survival curves and compared with log-rank test. For all tests, P<0.05 was considered statistically significant. All statistical analyses were performed in IBM-SPSS version 24.0 (IBM, Armonk, NY, USA) and R. (version 4.03).
Results
Baseline Characteristics
Patients were divided according to ACEF score tertiles into a low ACEF group (ACEF≤0.899, N=97), mid ACEF group (0.899<ACEF<1.130, N=100) and high ACEF group (ACEF≥1.130, N=93). The overall patient characteristics are shown in Table 1. Regarding demographic characteristics, the age, sex proportion, body mass index, medical history of myocardial infarction, diabetes mellitus, arrhythmia and stroke among the three groups significantly differed among groups. Patients in the high ACEF group had higher levels of troponin I, CK-MB, BNP, CRP, ESR, fasting glucose, D-dimer and FIB, but lower levels of hemoglobin, platelets and serum albumin, than patients in the other groups. In the evaluation of cardiac function with echocardiography, patients with higher ACEF had lower left ventricular ejection fraction (LVEF). Meanwhile, patients with higher ACEF scores also had higher GRACE and SYNTAX scores.
Basic Clinical, Laboratory and MACE Findings in Patients with ACS According to ACEF Score Groups.
| Low group (N=97) | Mid group (N=100) | High group (N=93) | P | |
|---|---|---|---|---|
| Demography | ||||
| Age, years | 51 (49,52) | 64 (62,65) | 71 (69,73) | <0.001 |
| Male, n, % | 84 (86.6%) | 67 (67.0%) | 69 (74.2%) | 0.005 |
| BMI, kg/m2 | 26.6±3.4 | 25.5±2.9 | 25.2±3.4 | 0.048 |
| Heart rate, bpm | 74±12 | 72±13 | 75±13 | 0.167 |
| Systolic blood pressure, mmHg | 131±17 | 130±17 | 129±22 | 0.427 |
| Diastolic blood pressure, mmHg | 78±12 | 74±11 | 72±11 | 0.001 |
| Previous MI, n, % | 13 (13.4%) | 15 (15.0%) | 32 (34.4%) | <0.001 |
| Previous PCI, n, % | 23 (23.7%) | 32 (32.0%) | 33 (35.5%) | 0.191 |
| Current smoker, n, % | 58 (59.8%) | 36 (36.0%) | 25 (26.9%) | <0.001 |
| Hypertension, n, % | 61 (62.9%) | 56 (56.0%) | 60 (64.5%) | 0.432 |
| Diabetes mellitus, n, % | 32 (33.0%) | 37 (37.0%) | 46 (50.0%) | 0.045 |
| Previous arrhythmia, n, % | 5 (5.2%) | 6 (6.0%) | 21 (22.6%) | <0.001 |
| Previous stroke, n, % | 11 (11.3%) | 5 (5.0%) | 16 (17.2%) | 0.026 |
| Laboratory findings | ||||
| WBC, ×109/L | 8.9±3.1 | 7.7±2.5 | 8.6±3.3 | 0.024 |
| Hemoglobin, g/L | 142.2±14.3 | 133.8±15.2 | 131.0±18.9 | <0.001 |
| Platelets, ×109/L | 218 (193,268) | 202 (175,240) | 202 (164,244) | 0.003 |
| Serum albumin, g/L | 41.6±4.7 | 40.4±5.0 | 39.0±6.4 | <0.001 |
| Total cholesterol, mmol/L | 4.3±1.1 | 4.0±0.9 | 4.2±1.3 | 0.093 |
| HDL, mmol/L | 0.96 (0.78,1.10) | 0.96 (0.82,1.12) | 0.90 (0.77,1.03) | 0.177 |
| LDL, mmol/L | 2.7±1.0 | 2.3±0.8 | 2.6±1.2 | 0.082 |
| Triglycerides, mmol/L | 1.8 (1.2,2.3) | 1.3 (1.0,1.9) | 1.3 (0.9,1.9) | 0.002 |
| Troponin-I, ng/mL | 0.23 (0,.00,19.15) | 0.10 (0.00,14.63) | 4.40 (0.03,41.22) | 0.004 |
| CK-MB, ng/mL | 2.0 (0.7,31.8) | 1.7 (0.8,22.9) | 6.6 (1.5,76.4) | 0.002 |
| BNP, pg/mL | 36.0 (18.0,95.0) | 58.0 (26.0,108.3) | 258.0 (100.0,556.0) | <0.001 |
| ESR, mm/h | 5.0 (2.0,11.5) | 6.5 (2.0,15.0) | 11.0 (5.0,21.0) | 0.001 |
| C-reactive protein, mg/L | 2.4 (0.9,5.9) | 3.2 (1.0,9.8) | 4.4 (1.7,23.7) | 0.024 |
| Serum creatinine, μmol/L | 67.1 (60.8,74.8) | 64.1 (56.4,76.2) | 77.4 (64.6,99.3) | <0.001 |
| BUN, mmol/L | 4.9 (4.3,6.1) | 5.3 (4.3,6.4) | 6.4 (5.2,8.3) | <0.001 |
| K+, mmol/L | 3.9 (3.7,4.1) | 3.9 (3.7,4.1) | 4.0 (3.8,4.3) | 0.021 |
| sTSH, uIU/mL | 1.2 (0.7,2.2) | 1.3 (0.6,2.1) | 1.4 (0.7,2.4) | 0.812 |
| D-dimer, mg/L | 0.19 (0.17,0.26) | 0.23 (0.19,0.50) | 0.44 (0.22,0.81) | <0.001 |
| Fibrinogen, mg/dL | 261.6 (227.6,306.4) | 276.2 (230.7,312.5) | 312.5 (258.9,386.3) | <0.001 |
| SYNTAX score | 20.4±8.7 | 19.9±9.3 | 27.1±9.3 | <0.001 |
| GRACE score | 117.7±22.0 | 139.2±22.5 | 170±27.4 | <0.001 |
| Echocardiography | ||||
| Left atrial diameter, mm | 35.5±4.5 | 36.4±4.2 | 38.7±4.5 | <0.001 |
| LVEDD, mm | 47.3±3.5 | 47.1±4.1 | 49.8±6.7 | 0.004 |
| LVESD, mm | 28.9±3.5 | 29.9±4.7 | 35.0±7.7 | <0.001 |
| LVEF, % | 67.6±5.8 | 63.5±6.8 | 52.0±11.5 | <0.001 |
| MACE | 15 (15.5%) | 19 (19.0%) | 28 (30.1%) | 0.037 |
MACE, major adverse cardiovascular events; BMI, body mass index; WBC, white blood cell count; HDL, high-density lipoprotein; CK-MB, creatine kinase MB; LDL, low-density lipoprotein; BNP, brain natriuretic peptide; ESR, erythrocyte sedimentation rate; BUN, blood urea nitrogen; sTSH, thyroid stimulating hormone; LVESD, left ventricular end systolic diameter; LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction.
Follow-up
During a median follow-up of 14 (12, 16) months, the rates of MACE were 15.5% in the low ACEF group, 19.0% in the mid group and 30.1% in the high ACEF group (P=0.037). The ROC was used to derive the cut-off value of FIB for predicting MACE. The cut-off of 291.1 mg/dL for FIB had a sensitivity of 87.1% and a specificity of 58.3% in predicting MACE. The patients were divided into two groups according to the FIB cut-off (lower group, FIB≤291.1 mg/dL; higher group, FIB>291.1 mg/dL).
Regression Analysis
Table 2 shows the univariate and multivariate logistic regression analyses of MACE for all patients. In the univariate analysis, several potential risk factors were identified, including FIB, BNP, creatinine, left atrial diameter (LAD), left ventricular end systolic diameter (LVESD), LVEF, SYNTAX score, diabetes and previous arrhythmia (P<0.05). However, after multivariate adjustment, only the level of FIB (odds ratio=7.798, 95%CI, 3.44–17.676, P<0.001) and the SYNTAX score (odds ratio=1.034, 95%CI, 1.001–1.069, P=0.041) emerged as independent predictors of MACE.
Logistic Regression Analysis of Clinical Parameters for MACE Prediction.
| Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|
| OR (95%CI) | P | OR (95%CI) | P | |
| Male | 0.39 (0.15,0.95) | 0.038 | ||
| Diabetes mellitus | 2.02 (1.08,3.77) | 0.028 | ||
| Previous arrhythmia | 2.55 (1.12,5.80) | 0.025 | ||
| BNP | 1.00 (1.00,1.10) | 0.021 | ||
| Serum creatinine | 1.00 (1.01,1.20) | 0.031 | ||
| Fibrinogen >291.1 mg/dL | 9.11 (4.41,20.03) | <0.001 | 7.80 (3.44, 17.68) | <0.001 |
| SYNTAX score | 1.05 (1.02,1.08) | 0.002 | 1.03 (1.00,1.07) | 0.041 |
| LAD | 1.09 (1.02,1.16) | 0.014 | ||
| LVESD | 1.05 (1,00,1.09) | 0.049 | ||
| LVEF | 0.95 (0.93,0.98) | 0.001 | ||
OR, odds ratio; CI, confidence interval; BNP, brain natriuretic peptide; LAD, left atrial diameter; LVESD, left ventricular end systolic diameter; LVEF, left ventricular ejection fraction.
The New Model
On the basis of the regression coefficient of FIB, the ACEF-FIB was developed. The score was derived by attributing integer numbers to the variables retained in the multivariable model. We used ROC curves to estimate the prognostic value of ACEF-FIB and other risk scores. The area under the ROC curve of the ACEF-FIB scoring system in predicting MACE after PCI was 0.753 (95%CI 0.688–0.817, P<0.001), a value higher than that of the ACEF score, SYNTAX score and Grace score (0.627, 0.637 and 0.570, respectively) (Figure 1). Compared with other risk scores, the ACEF-FIB also had better discrimination ability, according to NRI and IDI (Table 3).
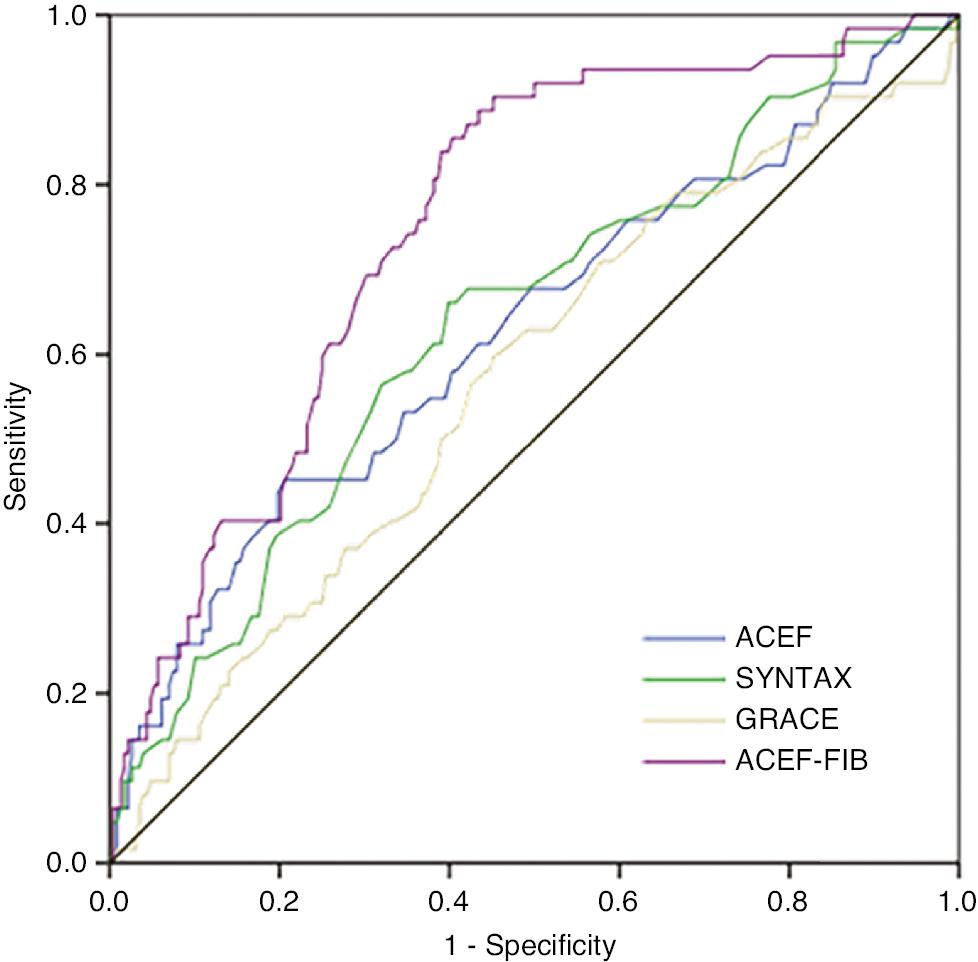
Receiver Operating Characteristic Curve Analysis of Risk Scores in Predicting MACE. MACE, major adverse cardiovascular events.
Reclassification of MACE with ACEF-FIB versus Other Scores.
| NRI or IDI [95% confidence interval] | P value | |
|---|---|---|
| ACEF-FIB score versus ACEF score | ||
| NRI | 0.788 [0.554,1.023] | <0.001 |
| IDI | 0.101 [0.066,0.136] | <0.001 |
| ACEF-FIB score versus SYNTAX score | ||
| NRI | 0.735 [0.487,0.983] | <0.001 |
| IDI | 0.097 [0.057,0.137] | <0.001 |
| ACEF-FIB score versus GRACE score | ||
| NRI | 0.891 [0.681,1.102] | <0.001 |
| IDI | 0.134 [0.099,0.168] | <0.001 |
MACE, major adverse cardiovascular events; NRI, net reclassification improvement; IDI, integrated discrimination improvement.
Kaplan-Meier estimates of MACE according to the ACEF score are shown in Figure 2. The best cut-off for ACEF-FIB for MACE was 1.87, with a sensitivity of 88.7% and a specificity of 56.6%. The new risk score was dichotomized on the basis of a cutoff determined by the Youden index: lower group <1.87 and higher group ≥1.87. Kaplan-Meier survival analysis indicated that patients in the low ACEF group had greater event-free survival rate than those in the high ACEF group (log-rank P<0.001) (Figure 2).
Discussion
This study demonstrated that the ACEF score combined with FIB predicted MACE in patients with ACS after PCI. When FIB and ACEF were jointly used to evaluate MACE, the AUC of the combined prognostic model significantly increased. In addition, the integration of FIB level significantly improved the discriminatory ability and reclassification of ACEF scoring. Therefore, this new score may provide a novel tool for risk stratification of patients with ACS in clinical practice.
With the rapid expansion of PCI indications and the increase in the clinical complexity of patients [1], risk assessment of the overall incidence of MACE after these procedures, particularly mortality, has become a highly important aspect of daily clinical decision-making. Some of risk scores, such as the SYNTAX and GRACE scores, have been widely used in clinical practice to stratify the risk of patients with ACS [10, 11]. However, the SYNTAX score is based on anatomic information and only indirectly incorporates clinical characteristics, because older patients with renal insufficiency generally have more calcified vessels and a wider range of diseases [4, 12]. Furthermore, the GRACE score contains many variables, thus resulting in inaccuracy and overfitting, and it lacks several important predictors of mortality, such as the LVEF [13]. Wu et al. have shown that LVEF after acute STEMI is a reliable and commonly used functional marker of severity of potential myocardial damage [13].
The ACEF score consists of three risk factors, all of which are objective measurement variables [3]. These risk factors represent three important prognostic indicators – age, renal function and cardiac function – which accurately reflect the burden of comorbidities and cardiovascular disease in patients with ACS [13–15]. The LEADERS trial has demonstrated a significant correlation between high ACEF scores and elevated risk of adverse events after coronary revascularization in patients receiving PCI treatment [4]. The predictive power of the ACEF score has been characterized in high-risk patients, such as those with chronic total occlusions, left main artery disease and heavily calcified lesions [16–18]. Our results were consistent with those from previous studies. Patients with higher ACEF scores were more likely to develop MACE, and high ACEF scores were significantly associated with poor prognosis in all patients.
FIB is a serum glycoprotein with a dimeric molecular structure; it is synthesized by the liver and was the first clotting factor discovered [19]. Inflammation is a common precursor of atherosclerosis [20], and FIB plays an important role in inflammation and tissue repair [21]. Previous studies have confirmed that FIB enhances systemic or local vascular inflammation and secondary vascular endothelial injury, and further promotes the accumulation and oxidation of subendothelial low-density lipoprotein, and subsequently the proliferation and migration of vascular smooth muscle cells [22, 23]. These reactions ultimately lead to the formation and vulnerability of atherosclerotic plaques [24]. In addition to being an acute phase reactant in inflammation, FIB is converted into insoluble fibrin by thrombin, thus exposing polymerization sites that promote thrombus formation during the activation of the coagulation cascade, platelet aggregation, and thrombosis [25]. Moreover, blood viscosity and peripheral resistance have been reported to increase with plasma FIB levels, thus resulting in disrupted blood oxygen transport, slow blood flow and aggregation of red blood cells, thereby increasing the risk of thrombosis [26]. Verdoia et al. have found that high FIB levels are an independent predictor of the presence and severity of coronary artery disease [27]. In the ERFC study, Kaptoge et al. have found that evaluating FIB concentrations is associated with a significant improvement in the prediction of cardiovascular adverse events [28].
In our study, FIB levels were higher in the high ACEF group than the low ACEF group. FIB, as expected, predicted poorer clinical outcomes in our ACS cohort. The predictive performance of the ACEF-FIB score was similar to that of the SYNTAX score. Moreover, the new ACEF-FIB model did not violate the simple principles of the original model. In clinical practice, the ACEF-FIB score might reasonably be used as a reliable and updated tool for risk stratification after PCI. However, we do not suggest replacing the original ACEF score or claim that the new model is superior to the existing scores; the new model must first be validated by external verification.
Limitation
This study has several limitations. First, this was a single center study and thus provides a low level of evidence. Second, the sample size of this study was small, and further validation will be necessary in a larger cohort of patients. Finally, the follow-up period was short and must be further extended in the future.


