Overview and Progress in Wire-Free Coronary Physiology Assessment
Conventionally, myocardial ischemia is attributed to “flow-limiting” epicardial coronary artery stenoses of >70% that are identified angiographically. However, in addition to the severity of stenoses, the area of the myocardium being supplied affects the functional significance of coronary artery lesions. Percutaneous coronary intervention (PCI), for instance, is a contemporary procedure to relieve flow-limiting stenosis, thus enhancing perfusion to the myocardium in patients with stable angina who are unresponsive to medication. According to recent studies, the presence of ischemia in intermediate-severity coronary lesions remains the most critical factor determining the clinical benefits of revascularization in the context of stable coronary artery disease (CAD), and an initial invasive strategy of functionally non-significant lesions does not confer any additional benefits over optimal medical therapy [1, 2]. PCI of non-flow limiting lesions has also been associated with major adverse cardiac events and the need for repeat revascularization [1]. Overall, clinical trial data support a primary pharmacological approach in this clinical scenario and recommend delaying revascularization in coronary artery stenoses without functionally significant ischemia [3–5]. Hence, recommendations have suggested identifying and quantifying ischemia before considering revascularization for stable angina [6, 7]. Nonetheless, no standard approach exists for performing physiological assessment of all coronary stenoses with intermediate severity before revascularization.
Before the development of an invasive wire-based technology that objectively measures pressure gradient differences across a coronary lesion (driving pressure for microcirculatory flow distal to the stenosis (Pd) in relation to the coronary pressure available in the absence of stenosis, aortic pressure ratio (Pa)), visual assessment of intermediate-degree stenoses remained the standard approach for revascularization decision-making [8]. Fractional flow reserve (FFR), a technique pioneered 30 years ago, is based on Poiseuille’s law, in which the distal coronary driving pressure measured in maximal hyperemia is directly proportional to the maximum vasodilated perfusion. To quantify the percentage contribution of coronary stenosis to cardiac flow limitation, the downstream arteries with resistance must be dilated with pharmaceutical drugs [9].
The acceptance of FFR is limited, although FFR assessment is increasingly used in interventional cardiology, and abundant scientific evidence supports its prognostic ability and cost-effectiveness. This limited acceptance is associated with perceptions that FFR is associated with additional procedure costs, time, and pressure wire instrumentation, as well as patient discomfort from the pharmacological medicines used to induce hyperemia [10]. Consequently, non-hyperemic pressure ratios (NHPR) or diastolic-only pressure ratios (dPR) have been developed in the search for simpler patient-friendly methods to increase the use of physiologically guided revascularization. These methods do not require hyperemia and avoid concerns regarding the best time to measure FFR during hyperemia or how to handle the adverse effects of hyperemia-inducing drugs such as adenosine. However, landmark studies of these applications have varied in their use of wire-based modalities and their assessment of various types of lesions in a heterogeneous population, thus yielding a range of results [11–14]. Different delivery techniques for NHPR, such as instantaneous wave-free ratio (iFR), have been demonstrated to be non-inferior to FFR and to provide a straightforward assessment of resting Pd/Pa in the diastolic period. The diastolic hyperemia-free ratio, resting full-cycle ratio and diastolic pressure ratio (DPR/dPR) are other diastolic indices from various vendors that have been validated with FFR [11–14].
Given that the adoption of wire-based coronary physiology assessment remains low, numerous groups have begun to develop wire-free angiography-based procedures using artificial intelligence to assess the functional importance of intermediate lesions, even in the absence of hyperemia induction. This review provides an overview and scientific background for various angiography-based FFR technologies, current evidence supporting their use in clinical practice and perspectives on future directions.
Concept of Angiography-Based FFR
Wire-free image-based FFR is software-based and has been enabled by the development of three-dimensional quantitative coronary angiography (3-D QCA) and computational fluid dynamics (CFD). Most programs use paired two-dimensional angiograms to reconstruct the three-dimensional vascular anatomy of the arteries. Because the Navier-Stokes equation, which uses CFD to explain fluid motion, can be time- and resource-intensive, simplified assessments using equations based on the pioneering work of Young, Tsai and Gould have been suggested [15, 16]. The Gould formula estimates the pressure drop for each stenosis segment by using geometry and the mean hyperemic flow velocity, by modelling a range of flow rates to determine the pressure difference at rest and during an assumed hyperemic state. The relationship between coronary pressure and flow velocity is predicated on the assumption that blood is a homogeneous Newtonian fluid with predetermined outflow boundary conditions. The mass flow rate across a stenosis section is then calculated by using the vessel dimensions and mean flow velocity from 3D QCA [17].
Available Angiography-Based FFR Solutions
Angiography-based FFR or angio-derived FFR from diagnostic angiography was previously complicated and time-consuming without the use of a conventional pressure wire or hyperemia induction. However, technology has made these processes faster, particularly in terms of computational data processing speed and capabilities, thus enabling the application of FFR approaches derived from angiography in clinical settings. Angio-derived FFR analysis can now be available within minutes after performing conventional coronary angiography, and its diagnostic performance is comparable to that of wire-based FFR. Numerous methods have been developed, many of which are currently commercially available. Table 1 provides a summary and comparison of available angiography-based FFR techniques [18–32].
Summary and Comparison among Current Available Angiography-Based FFR Technologies.
| # | Index | Technological description | Potential advantages | Pitfalls/limitations |
|---|---|---|---|---|
| 1. | Computational pressure-flow dynamics derived FFR (caFFR) | This method incorporates real-time aortic pressure monitoring in CFD by using a pressure transducer to determine the pressure drop across the stenosis. | This method considers the dynamic nature of blood pressure to determine hemodynamically significant coronary artery stenoses. | This method has high cost. Randomized clinical trial outcomes are limited, and trials are currently ongoing. |
| 2. | Quantitative flow ratio (QFR) | FFR calculation relies on the contrast flow frame count and 3D reconstruction of the stenotic channel created from two angiographic projections. The measurement of blood flow velocity is combined with a 3D reconstruction of the arteries. | This method decreases the time and cost, and eliminates the risks associated with pressure wire instrumentation in a coronary artery as well as induction of hyperemia. Analysis can be performed offline or online. | This method has technical difficulties in aortic-ostial lesions, severe tortuosity or overlapping vessels. This method has not been validated for the assessment of bifurcations with stenosis in both the side branch and the proximal main vessel. |
| 3. | Murray’s law based QFR (μQFR) | This method creates a step-down reference diameter tapering function and the lumen outlines of the epicardial coronary arteries. | FFR calculation requires only one angiographic image. Computation time is shortened. Fractal geometry is incorporated into CFD simulation at side branches and bifurcations. | Highly eccentric lesions may compromise accuracy. |
| 4. | FFRangio | This method enables 3D reconstruction of the whole coronary tree with color-coded FFR values at different epicardial locations, by using two distinct angiographic projections at least 30° apart. | Simultaneous evaluation of the whole coronary tree is performed. | Excellent angiographic views are required. This method is relatively less accurate in microvascular dysfunction. |
| 5. | Vessel fractional flow reserve (vFFR) | This method provides a virtual pullback, wherein pressure is shown at each location in a branching coronary artery tree in a single study. | This method has advantages and limitations that may be similar to those of procedures based on coronary angiography. | Excellent angiographic views are required. This method is relatively less accurate in microvascular dysfunction. This method has a limited amount of supporting evidence, in contrast to QFR. Computational times may be long. |
| 6. | Intravascular ultrasound (IVUS)-derived FFR (UFR) | Luminal geometry and a 3D model are reconstructed on the basis of electrocardiogram-gated-IVUS images. Machine learning then uses patient-specific hemodynamic conditions to compute the IVUS-based FFR. | Maximal hyperemia is not required. This method assesses the entire vessel wall while providing anatomical and functional evaluations without a need for wire exchange. This method provides flow along with qualitative and quantitative variables. Assessment can be performed in ostial lesions and is not affected by vessel tortuosity. | Evidence and clinical data are currently limited. Vessel instrumentation is required. This method has high cost. |
| 7. | Optical coherence tomography (OCT)-derived FFR (OFR) | This method provides images with high resolution and high contrast between the vessel wall and lumen. This method reconstructs 3D models and machine-learning generated FFR similar to IVUS-FFR and provides a feasible alternative to IVUS-based FFR | This method has the advantages of both high-definition anatomical imaging and functional assessment during PCI direction and planning (stent length selection/serial lesions selection). Post-PCI results can be evaluated both anatomically (OCT) and physiologically (OCT-FFR). | Evidence and clinical data are currently limited. Vessel instrumentation is required. This method has high cost. Side branch segmentation might be inaccurate in certain situations. For example, side branch ostial disease may confound the vessel tapering model’s quantification of side branch sizing. |
Abbreviations: 3D, three-dimensional; FFR, fractional flow ratio; CFD, computational fluid dynamics; QFR, quantitative flow reserve; PCI, percutaneous coronary intervention.
Invasively, to recreate a 3D QCA model, computational pressure-flow dynamics derived FFR (caFFR) (Rainmed Ltd, Suzhou, China) uses two angiographic projections at various angles (of at least 30° apart). The FlashPressure pressure transducer attached to the guide catheter records the real-time aortic pressure and transmits it to the FlashAngio console, which automatically calculates the mean aortic pressure over the third to eighth cycles after angiography. The resting coronary flow velocity is determined via the angiographic thrombolysis in myocardial infarction (TIMI) frame count [30]. The FlashAngio software then uses this information to calculate the pressure drop along the generated location of the coronary artery. An example of caFFR analysis showing a hemodynamically significant intermediate coronary lesion is shown in Figure 1. Unlike caFFR, other CFD software does not consider real-time invasive pressure in computing the pressure drop across coronary artery stenosis. To account for energy diffusion in the lumen before and after the investigated coronary artery stenosis, the dynamic nature of blood pressure is considered. The Stokes-Navier equation is solved for the data through the CFD approach to estimate the flow rate and pressure gradient [29]. CaFFR has been demonstrated to be extremely accurate in the FLASH FFR, by using wire-based FFR as a reference; this method requires 1 minute of processing time and a total operational time less than 5 minutes [29]. The multicenter, prospective, blinded, randomized, non-inferiority FLASH FFR II trial is now being conducted to compare the clinical results of pressure-wire FFR-guided PCI versus caFFR-guided PCI.

Angiography Showing an Intermediate Lesion in the Right Coronary Artery.
caFFR analysis was 0.71, thus indicating the hemodynamic significance of the lesion. caFFR also incorporates invasive aortic pressure monitoring in the computation of angiography derived FFR.
The quantitative flow ratio (QFR) (Medis Medical Imaging System, Leiden, the Netherlands and Pulse Medical Imaging Technology, Shanghai, China) reconstructs a 3D vessel model with two orthogonal diagnostic angiographic projections in the optimal end-diastolic frame selected by automated electrocardiography triggering. In the QFR model, the coronary pressure decreases, and the mean hyperemic flow velocity is calculated for each stenotic segment with the Gould formula, on the basis of the assumption that blood is a homogeneous Newtonian fluid [27, 28]. Following that, 3D QCA predicts the mass flow rate at each position along the examined vessel using this formula and vessel dimension [27, 28]. In the Functional Diagnostic Accuracy of Quantitative Flow Ratio in Online Assessment of Coronary Stenosis (FAVOR) pilot study, three flow models were initially used to evaluate the computation performance of the hyperemic flow: (1) fixed empiric hyperemic flow velocity (fixed-flow QFR (fQFR)); (2) modelled hyperemic flow velocity based on the TIMI frame count analysis without pharmacologically induced hyperemia (contrast-flow QFR (cQFR)); and (3) measured hyperemic flow velocity derived from coronary angiography during adenosine-induced hyperemia (adenosine-flow QFR (aQFR)) [26]. The authors observed good agreement with FFR for all measurements acquired with or without pharmacological hyperemia (fQFR 0.003±0.068, cQFR 0.001±0.059 and aQFR 0.001±0.065), thus confirming that computation of cQFR without pharmacological hyperemia-induction is feasible and has potential for broader use [26]. Figure 2 shows an example of QFR analysis illustrating an intermediate coronary lesion that was not functionally significant; the residual QFR predicts the post-PCI FFR by simulating the effects of stent implantation in a coronary lesion segment. Despite a substantial learning curve, the online computation time for QFR has been reported to be significantly shorter than FFR in the FAVOR II Europe Japan (5.0 vs 7.0 min, P<0.001) and FAVOR III China (3.9±1.4 min) studies, and has shown good agreement with the gold standard of FFR [24, 25]. The multicenter, blinded, prospective FAVOR III China study, an entirely Asian population-based randomized controlled trial conducted in China, has demonstrated better 12 month clinical outcomes of QFR-guided PCI than angiography-guided PCI [24]. The outcomes of the ongoing FAVOR III Europe Japan clinical trial dedicated to transferring and extending these findings to other populations are keenly anticipated, given the promising outcomes achieved in the China setting.
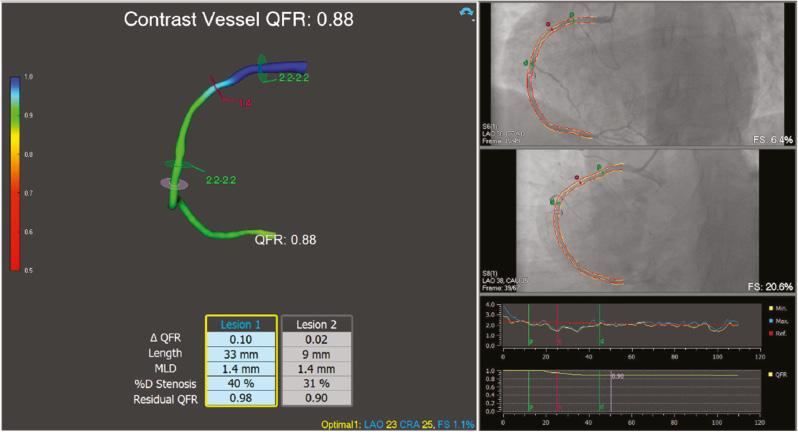
An Intermediate Lesion in the Right Coronary Artery, Analyzed with QFR.
A three-dimensional reconstruction of the coronary artery and computational analysis using two angiographic projections 30° apart indicates that the lesion is hemodynamically non-significant, 0.88.
Murray’s law-based QFR (μQFR) (Pulse Medical Imaging Technology, Shanghai, China) uses CFD to recreate the lumen outlines of epicardial coronary arteries and a step-down reference diameter tapering function. The Murray bifurcation fractal law also considers side branches in the CFD bifurcation and includes an exclusive bifurcation lesion analysis [23]. Hence, only a single angiographic view is required for rapid computation of the FFR. Validation of this Murray’s law-based QFR in the FAVOR II China study cohort has indicated that single view μQFR assessment to identify functionally important coronary lesions by using a single angiographic projection has comparatively good accuracy with respect to that of two-view three-DμQFR angiographic projections, which can also be made available in the analysis workflow [23]. Figure 3 shows an example of μQFR analysis, illustrating the assessment of an intermediate coronary lesion that was hemodynamically significant and computed with only a single angiographic projection. The virtual stenting technique simulates the effect of stent implantation in selected segments by using the predicted value of residual QFR.

An Intermediate Lesion in the Right Coronary Artery, Assessed with μQFR.
Only a single angiographic projection is required for three-dimensional reconstruction and computational assessment of the left anterior descending (LAD). Side branches and bifurcations are considered in the functional assessment of coronary stenosis. The analysis showed hemodynamically significant coronary stenosis in the proximal LAD, and the respective FFR value was calculated by using the virtual stenting function.
FFRangio (CathWorks, Ltd, Kfar Saba, Israel) is an angiography-based index that enables functional angiographic mapping of the complete coronary tree while the patient is at rest, without the use of adenosine. It provides a 3D reconstruction of the coronary tree with color-coded FFR values at different epicardial locations, by using two distinct angiographic projections at least 30° apart. The hyperemic flow ratio is derived from a lumped model system contributed by overall resistance and the patient’s hemodynamic data [22]. FFRangio is subsequently calculated as the ratio of maximal flow in the presence and absence of stenotic lesions in two different models of coronary trees [23]. The prospective, multicenter FAST-FFR trial has provided primary evidence validating FFRangio and has indicated a sensitivity of 94%, specificity of 91% and accuracy of 92%, with respect to conventional pressure wire-based FFR [22].
Vessel fractional flow reserve (vFFR) (CAAS, Pie Medical Imaging, Maastricht, the Netherlands) uses a pseudo transient mathematical model and CFD approach to process two angiographic views with at least a 30° difference in rotation/angulation. The CAAS workstation uses an intrusive non-dimensional indicator called the pressure drop coefficient, which incorporates viscous resistance and separation of lesser effects, on the basis of well-validated CFD approaches, to evaluate coronary flow behavior [21]. vFFR then integrates measured aortic root pressure and an automated algorithm that assumes maximum hyperemic blood flow to determine the hemodynamic significance of an intermediate coronary lesion. vFFR then uses an automated algorithm that assumes maximum hyperemic blood flow to determine the hemodynamic significance of an intermediate coronary lesion. The single-center observational FAST study has indicated a high diagnostic accuracy of vFFR compared with pressure wire-based FFR (area under the curve 0.93, 0.88–0.97 95% confidence interval) with low interobserver variability (R=0.95; P<0.001) [21]. The large international, prospective, observational FAST II trial showed a good correlation between both online and core laboratory-assessed vFFR, compared with conventional pressure wire-based FFR (R=0.74; P<0.001; mean bias 0.0029±0.0642), for intermediate coronary artery lesions in patients with stable CAD [20]. The FAST III study is an ongoing multicenter trial comparing clinical outcomes of a vFFR-guided strategy compared with FFR-guided coronary revascularization.
Intravascular ultrasound (IVUS)-derived FFR (UFR) and optical coherence tomography (OCT)-derived FFR (OFR) (Pulse Medical Imaging Technology, Shanghai, China) while simulating virtual FFR from CFD, can interrogate both coronary anatomy in high definition as well as the hemodynamic significance of an intermediate lesion to aid in revascularization planning. A reconstruction of the coronary channel is performed after the intracoronary catheter image pullback. RefineNet, a deep learning artificial intelligence method, was initially used to capture blood flow speckles, and delineate the intima and arterial lumen contours in 3D [33]. Second, according to bifurcation fractal principles, the ostia and reference lumen size of side branches are reconstructed and quantified perpendicular to the centerlines of the side branches [17, 34]. A fluid dynamic algorithm that considers a downstream volumetric flow rate by multiplying the reference lumen area with a virtual hyperemic flow velocity then determines the pressure drop along the vessel of interest to obtain UFR/OFR pullback, by using a validated computational FFR method [18, 19, 35, 36]. This method examines the inertial and frictional losses inherent to the flow’s abrupt change as it crosses a stenosis [35]. Prior wired-FFR validation studies using this technology have demonstrated diagnostic concordance; the vessel-level diagnostic accuracy for OFR was 90–93%, with a sensitivity and specificity of 87% and 92%, respectively, and low inter-observer variability [18, 19]. Whereas QFR fits well in the diagnostic catheterization laboratory and in ordinary PCI procedures, UFR/OFR can complement the QFR portfolio in patients with complex coronary anatomy (left main lesion/complex bifurcation lesions with significant vessel foreshortening and overlapping), and can aid in assessing stent malpositioning or underexpansion. UFR/OFR analysis is appealing for assessing and optimizing complex PCI procedures without being constrained by the invasive nature of repeated instrumentations. Although IVUS has poorer resolution than OCT, UFR has shown comparable accuracy to OFR, owing to IVUS’s superior tissue penetration [37]. More details may become available from future studies comparing the two modalities.
Compared with wire-based FFR, angio-derived FFR has both advantages and drawbacks. Without use of a wire and hyperemic medication, patients experience less discomfort and shorter operation times, and the method poses no risk of wire instrumentation or pressure wire drift, which can lead to erroneous coronary pressure measurement in as many as one-third of procedures [38]. The speed of computing and the flexibility of online and offline analysis are arguably the two crucial points favoring use of this modality. In the FAVOR II Europe Japan (Diagnostic Accuracy of Online Quantitative Flow Ratio) study, the median time for QFR computation of the entire procedure was significantly shorter than that for FFR (5.0 min vs. 7.0 min) [25]. Whether these variations can be reproduced outside a clinical trial setting remains to be seen. Additionally, most angio-derived FFRs include the ability to calculate post-PCI FFR through virtual stent analysis while considering data from the pre-PCI FFR baseline. In the presence of tandem lesions, this software assesses the entire length of the vessel to predict which part of the vessel should be treated, to derive the maximum post-PCI FFR [39, 40]. Preliminary retrospective analyses have yielded promising results, and further prospective studies to validate this strategy are awaited.
Currently, the major drawback is the lack of results from a large randomized controlled trial evaluating clinical outcomes compared with established FFR-guided PCI strategies. However, such trials are ongoing in the FAVOR III Europe Japan Study and FAST III study [41, 42]. Specific lesion types, such as left main coronary artery bifurcation lesions, serial lesions, diffuse disease as well as ostial lesions, are known confounders, because the laminar flow in coronary arteries is pulsatile and not straight, unlike the presumed straight conduit, thereby resulting in differences in the interpretation of any associated coronary stenosis. Furthermore, the functional analysis is heavily reliant on the quality of acquisition of angiographic views (optimal angle difference, no vessel overlap/foreshortening, optimal contrast filling and avoidance of panning). The contour of the vessel can still be manually corrected in the computed analysis and thus could influence interobserver variability in the FFR results. Hence, operators are recommended to undergo proper training and certification in this technique to minimize the effects of this phenomenon. The main limitations of QFR are inherent to all non-invasive parameters, as described above.
In summary, these techniques represent a milestone in non-invasive physiological assessment and have applicability in daily coronary assessment in cardiac catheterization laboratories.
Research Landscape of Wire-Free Coronary Physiology Assessment
A systematic search was performed to understand the research landscape and gaps in evidence regarding wire-free coronary physiology assessment, as a complement to the above narrative of existing FFR trials focusing on wire-free FFR. For many practicing physicians, coronary physiology involves the measurement of coronary pressure for the calculation of FFR, and non-invasive methods encompass imaging procedures such as computed tomography or magnetic resonance imaging [43]. Therefore, this review focusses on wire-free FFR. A search strategy was developed to identify all studies relevant to wire-free coronary physiology assessment, particularly those on FFR. Broad search terms (comprehensive search strategy in supplementary material Table S1) were used to identify the maximum number of studies eligible for inclusion from the electronic databases of PubMed, ScienceDirect, Cochrane and journals indexed in Radcliffe Cardiology. Various combinations of the following keywords were used: “coronary,” “coronary physiology,” “coronary circulation,” “fractional flow reserve, myocardial,” “FFR,” and “wire-free” [44, 45]. Systematic searching and analysis yielded 62 citations after the elimination of duplicates, in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Figure 4). Initial screening of these 62 articles yielded 38 articles for full-text review; 14 articles were subsequently included in the final review.

PRISMA Flow Diagram Illustrating the Selection Process for Articles on the Application of the Wire-Free Fractional Flow Ratio in Coronary Physiology Assessment.
An overview of the studies on wire-free FFR application in coronary physiology assessment is shown in Table 2. All reviewed publications were published in the past 5 years, most of which were efficacy studies evaluating the performance of wire-free FFR in different population cohorts, by using wire-based FFR as an intervention or comparator. Two articles used wire-free FFR as a companion method to index of microcirculatory resistance (IMR) and angiography-derived index of microcirculatory resistance (angio-IMR) analyses. More than half of the reviewed publications reported original research performed European researchers affiliated with universities. QFR had the most recent publications among angio-derived FFR, and the key FAVOR III China randomized control trial extended the utility of this modality to specific clinical settings. This was the first wire-free FFR modality assessed in a prospective, multi-center head-to-head outcome study to strengthen the evidence of the benefits of non-invasive physiological guidance versus angiography-guided PCI at 12 months [24].
Published Studies on Wire-Free Fractional Flow Ratio Application in Coronary Physiology Assessment in the Past 10 Years (2013–2022).
| # | Lead authors | Study aim | Study design (with information on population, intervention and comparator) | Findings/outcomes/recommendations |
|---|---|---|---|---|
| 1. | Albaghdadi, M. and Jaffer, F.A., 2018 [46] | This opinion article discusses the Wire-Free Functional Imaging (WIFI II) study. | NA | Expanding the use of coronary physiology assessment, a critical approach for the best management of coronary artery diseases requires the availability of rapid, effortless estimates of FFR. |
| 2. | Kołtowski, Ł., Zaleska, M., et al., 2018 [47] | This study evaluates the diagnostic accuracy of QFR. | Population: Patients with stable coronary artery disease who previously received FFR evaluation (n=268) Intervention: Retrospectively computed QFR with corresponding FFR Comparator: FFR | The best comparable QFR model was the QFR value at the pressure transducer position. iQFR is distinguished by its high diagnostic accuracy. |
| 3. | Stegehuis, V.E., Wijntjens, G.W., et al., 2018 [48] | This review details the fundamental basis of clinical coronary physiology and its relevance in the evaluation of coronary stenosis by using coronary pressure or coronary flow. | NA | The coronary pressure and flow have advantages and disadvantages in evaluating coronary stenosis. Both factors are complementary and, when combined, best define the pathophysiological basis of IHD. |
| 4. | Westra, J., Tu, S., Winther, S., et al., 2018 [49] | This study evaluates the feasibility and diagnostic performance of QFR in unselected consecutive patients. | Population: Patients with suspected coronary artery disease (n=362) Intervention: QFR Comparator: FFR | QFR properly categorized 83% of the lesions when the reference standard was an 0.80 FFR cut-off. |
| 5. | Zaleska, M., Koltowski, L., et al., 2019 [50] | This article evaluates the influence of laboratory and clinical variables on QFR-FFR mismatch. | Population: Patients with lesions (n=196) Intervention: Computed QFR Comparator: Wire-based FFR | QFR diagnostic accuracy may be compromised by CKD. Larger prospective studies are required to further investigate this potential association. |
| 6. | De Maria, G.L., Scarsini, R., et al., 2020 [51] | This study develops and validates the angiography-derived index of microcirculatory resistance (angio-IMR) as a novel pressure-wire-free index to facilitate assessment of coronary microcirculation. | Population: Patients with STEMI (n=45) Intervention: angio-IMR (QFR was assessed in IRA to generate and validate angio-IMR as a pressure-wire-free alternative to IMR for the measurement of coronary microvasculature) Comparator: IMR | angio-IMR is promising as a tool for the assessment of coronary microcirculation. |
| 7. | Sejr-Hansen, M., Westra, J., et al., 2020 [52] | This study tests the hypothesis that QFR and FFR perform equally when MPS and CMR are used as a reference to identify myocardial ischemia in patients with stable angina pectoris. | Population: Patients with suspected coronary artery disease by CCTA (n=176) Intervention: Patients randomized 1:1 to MPS or CMR Comparator: FFR, QFR | The diagnostic performance of QFR and FFR was comparable but moderate with respect to that of MPS or CMR. |
| 8. | Westra, J. and Tu, S., 2020 [53] | This article reviews the quantitative flow ratio and optical flow ratio. | NA | The use of quantitative flow ratio and optical flow ratio has the potential to facilitate the rapid and safe identification of lesions requiring revascularization, the optimization of PCI, the evaluation of plaque characteristics and the virtual planning of PCI. |
| 9. | Cherukuri, L., Birudaraju, D., et al., 2021 [54] | This article reviews the role of FFRCT according to supporting evidence and describes the challenges in its widespread application for determining hemodynamically significant stenosis, on the basis of the most important research articles. | NA | Non-invasive FFR derived from CCTA is a novel method with a high diagnostic yield for detecting and excluding flow-limiting coronary lesions. |
| 10. | Dan, K., Garcia-Garcia, H.M., et al., 2021 [55] | This article explores the relationship between target vessel vulnerability by OCT and wire-free functional assessment with QFR. | Population: Patients with stable angina (n=25) and NSTEMI (n=24) in the final prospective study cohort from the DECODE study Intervention: VVS Comparator: Vessel QFR | A significant inverse linear correlation existed between VVS and QFR (r=−0.34, P=0.009). Plaque distribution determined by OCT was comparable between stable angina and NSTEMI. The vulnerability of the target vessel was greater in patients with lower QFR values. |
| 11. | Mejia-Renteria, H., Lee, J.M., et al., 2021 [56] | This study develops a novel wire- and adenosine-free microcirculatory resistive index from functional angiography (angio-IMR). The aims were to estimate coronary microcirculatory resistance and to investigate how this method can improve clinical interpretation of physiological stenosis assessment with QFR. | Population: Patients with clinically indicated coronary angiography and stable coronary artery stenoses tested with intracoronary pressure and thermodilution-derived flow measurements (n=104) Intervention: (1) development of a formula (angio-IMR) to estimate IMR from resting angiograms and aortic pressure and (2) validation of the method in a clinical population by using invasively measured IMR as a reference, and evaluation of the diagnostic efficacy of QFR in light of the angio-IMR results Comparator: Invasively measured IMR | IMR can be estimated without a physiological wire and adenosine. In arteries with low angio-IMR, coronary microcirculatory dysfunction causing high IMR can enable rule out with high confidence. Knowledge of angio-IMR improves clinical interpretation of functional stenosis assessment with QFR. |
| 12. | Bharmal, M., Kern, M.J., et al., 2022 [57] | This review summarizes evidence and discusses commonly used methods in multivessel coronary artery stenosis physiologic assessment. | NA | Coronary physiologic assessment in multivessel disease decreases observer bias in visual estimation of stenosis, modifies therapeutic therapy and enhances patient outcomes. |
| 13. | Bhavnani, C.D., Fong, A.Y.Y., et al., 2022 [58] | This article assesses the performance and 12-month clinical outcomes in patients with CAFFR-guided PCI deferral. | Population: Patients (93 vessels) with 30–90% angiographic stenosis and estimated vessel diameters of 2 mm in the stenotic segment (n=69) Intervention: Wire-free FFR Comparator: Wire-based FFR | CAFFR demonstrated excellent concordance with wire-based FFR, and 12-month outcomes demonstrated that CAFFR-guided PCI deferral was safe and equivalent to wire-based FFR guidance. |
| 14. | Sejr-Hansen, M., Christiansen, E.H., et al., 2022 [59] | This article evaluates the diagnostic performance of QFR pre-TAVI in patients with aortic valve stenosis and CAD. Post-TAVI FFR and iFR were used as references. | Population: Patients undergoing TAVI (n=28) Intervention: QFR measured pre-TAVI Comparator: FFR post-TAVI | Pre-TAVI QFR diagnostic performance was better than that of post-TAVI FFR as a reference. |
Abbreviations: NA, not applicable; QFR, quantitative flow reserve; FFR, fractional flow ratio; MPS, myocardial perfusion scintigraphy; CMR, cardiovascular magnetic resonance; CCTA, coronary computed tomography angiography; CKD, chronic kidney disease; IMR, index of microcirculatory resistance; OCT, optical coherence tomography; VVS, vulnerable vessel score; iQFR, QFR value at the pressure transducer position; TAVI, transcatheter aortic valve implantation; CAD, coronary artery disease; iFR, instantaneous wave-free ratio; PCI, percutaneous coronary intervention, CAFFR, coronary angiography-derived fractional flow reserve; FFRCT, fractional flow ratio derived from CT; CT, computed tomography; IRA, infarct related artery; NSTEMI, A non-ST-elevation myocardial infarction; IHD, ischemic heart disease.
In the WIFI II study by Westra et al., which evaluated the diagnostic performance and feasibility of wire-free QFR, good agreement and diagnostic accuracy were observed with respect to those of FFR. QFR correctly classified 83% of lesions when the reference standard had an 0.80 FFR cut-off [49]. Currently, most published evidence from large randomized studies has included only Western populations, with the exception of the exclusively Asian study cohorts in the ground-breaking FAVOR III China and a prospective study from Malaysia demonstrating favorable 12 month clinical outcomes of caFFR-guided PCI [58]. Hence, a clear need exists for more local Asia Pacific (APAC) real-world data to be generated, given the unique patient makeup, treatment strategies and techniques, lesion complexity, access to coronary physiology and imaging devices, and differently structured healthcare systems in this region.
One of the many factors contributing to the inconsistent use of physiological assessment in the APAC region is interventional cardiologists’ misconception that physiologically guided PCI might have limited overall value through requiring more time in the cardiac catheterization laboratory and decreasing service efficacy [60]. A lack of equipment, reimbursement and in-country consensus has also resulted in physicians relying more on their experience than on guidelines for therapeutic strategies [60].
Numerous opportunities exist for local clinical outcome trials in the APAC region that can compare results with non-APAC populations. In-country registry data and health economic data could be generated to strengthen the recommendations and reimbursement policies, and increase the use of physiologically guided PCI in the APAC. Future APAC studies should aim to produce multicenter, prospective, randomized, long-term prognostic data and cost-benefit analyses to validate various wire-free FFR technologies. A head-to-head comparison evaluating the diagnostic performance of various wire-free FFR technologies might not influence the development of new guidelines yet could add value by demonstrating the potential applicability of each modality in various centers, on the basis of individual resources and capabilities. The next challenge would be generating clinical evidence to integrate the assessment of microvascular dysfunction in practice. The applicability of new generations of QFR technologies in interventional practice that further minimize analytic variability and computing time (automatic creation of angiographic-FFR with only a single angiographic projection) is eagerly awaited.
Actual Clinical Applications
The diagnostic performance of angiography-based FFR has been evaluated and shown to be equivalent to that of wire-based FFR in specific clinical scenarios. The largest body of evidence was derived from patients with stable CAD, as discussed above. Moreover, in acute ST elevation myocardial infarction (STEMI) and non-ST elevation myocardial infarction (NSTEMI), angio-derived FFR in non-culprit vessels appears to be practical, efficient and cost-effective [61]. Often, for revascularization of the culprit artery in acute coronary syndrome, no treatment or physiological assessment is performed on the non-culprit arteries, because most interventional cardiologists opt for a rapid, less complicated procedure during emergencies. Consequently, staged procedures may be performed to address the other non-culprit arteries; these procedures result in additional risks and costs, which could be avoided by integrating angio-derived FFR during the index procedure [62]. In the APAC region, access to a cardiac catheterization laboratory or a tertiary cardiology center may be a challenge for most patients, particularly those residing far from a facility. Therefore, any means to improve the diagnostic assessment of non-culprit lesions in patients with acute myocardial infarction transferred to such facilities during the index admission could positively affect long-term care.
In real-world situations, angiography-based FFR values are often near the cut-off (0.80). To consolidate stenosis severity in various situations, the idea of using a “gray zone” in which wired-FFR assessment might be used as an addendum has been suggested [63, 64]. Although the clinical implications of angiography-based FFR using a binary cut-off are currently being investigated in ongoing large clinical outcome trials, such as the FAVOR III Europe Japan and FAST III trials, future research is necessary to explore the clinical relevance of discordance between angiography-based FFR and FFR.
The accuracy of computed QFR in complex vessels (for example, bifurcations, heavily calcified vessels or diffusely diseased vessels) and adjunct clinical conditions (aortic stenosis, increased left ventricular end-diastolic pressure, post transcatheter aortic valve implantation (TAVI), diabetes mellitus, irregular heart rhythm or large vessels) remains to be investigated in larger patient cohorts.
Future Perspective and Challenges
In cardiac catheterization laboratories, FFR is the cornerstone index for coronary physiological assessment, because of its substantial evidence base. However, NHPRs can be considered an alternative to FFR, because discordance between FFR and iFR has not translated to differences in outcomes in the two large randomized trials [65, 66].
For hemodynamically significant stenosis, defined as FFR ≤0.80, angio-derived FFR exhibits comparable prognostic performance to traditional wire-based FFR, particularly with early revascularization. However, after an index revascularization procedure by PCI, a post-PCI FFR may improve short and long term outcomes. Assessment of this concept in clinical trials has become more feasible with the emergence and availability of angio-derived physiology indices. A series of studies have shown that post-stenting FFR >0.90 and drug-coated balloon treatment after successful balloon angioplasty with a resultant FFR value ≥0.75 is safe and effective [67–69]. However, the adoption of post-PCI FFR utilization remains modest, and cardiologists in the APAC region use this method in <25% of physiology procedures [60]. Of note, present-generation virtual stenting QFR or vFFR functions cannot account for conditions such as heavy calcification or stent under-expansion. The clinical effects of physiology-guided PCI optimization using various applications remain to be demonstrated.
Finally, evidence is limited regarding the measurement of myocardial dysfunction and the extent to which variations in the microcirculation affect coronary flow (for example, in infarct-associated arteries in patients with STEMI or in stable patient populations with substantial microvascular abnormalities). Coronary microcirculation and resistance are complicated models, but other non-invasive modalities may improve the diagnostic capabilities of this discipline.
The choice of physiological index used for clinical decision-making in cases with discordant values remains challenging, and further studies with larger sample sizes are warranted to provide longer-term clinical evidence. To date, angio-derived FFR techniques have been found to be reassuringly safe, with minimal procedural complications.
Conclusion
After three decades of use of FFR derived from wire-based technologies, the advent of wire-free techniques to assess the functional significance of coronary stenosis has a growing base of clinical evidence supporting its greater use in clinical use at cardiac catheterization laboratories. In many APAC countries, where the morbidity and mortality associated with CAD are high, and access to tertiary cardiology centers is limited, wire-free FFR techniques stand to play a greater role in improving the management of CAD, at the individual patient level and in resource planning at the healthcare service provision level.

