Introduction
Coronary artery disease (CAD) is the second leading cause of cardiac death in the Chinese population [1]. In China, the mortality due to CAD is lower than that in Western countries, but in recent years, the burden of CAD has been increasing [2], and the number of patients accepting percutaneous coronary intervention (PCI) therapy has increased rapidly in China. Most of these early PCI procedures were performed with balloon angioplasty [3] and drug eluting stents, which formally entered the Chinese market in 2002. To satisfy the growing market, studies on XIENCE V, ENDEAVOR, CYPHER, and FIREBIRD drug-eluting stents have been performed in China, and clinical trials involving the EXCEL sirolimus biodegradable eluting scaffold have also been performed [4–7].
The clinical efficacy of the XIENCE V Everolimus Elution Coronary Stent System (EECSS) had been well validated in both Chinese premarket and post-market trials, including the SPIRIT V single cohort study in 63 patients and the SPIRIT women single cohort study in 50 patients [8]. In addition, Abbott Vascular had completed two Chinese clinical trials, including a XIENCE V Chinese randomized controlled trial [RCT] and a XIENCE V single arm study [SAS] in 2011 [4]. No stent thrombosis (ST) was observed in any patients, and the success rates of both clinical instruments and operation were 100%.
The XIENCE PRIME EECSS is an improved generation of drug-eluting stents of the XIENCE V EECSS, and both of them have similar efficacy and safety. The XIENCE PRIME EECSS consists of two adjustable parts: a base bracket (multi-link VISION) coronary stent system (CSS) or Multi-link VISION CSS and a drug (everolimus in a polymer coating). The Multi-Link VISION® stent uses medical grade L-605 cobalt-chromium (CoCr) alloy. The efficacy and safety of XIENCE PRIME have been validated in the Spirit Prime study, which has confirmed a 1-year outcome of target lesion failure (TLF) consistent with the conclusions of the XIENCE V study in the USA [9]. However, the longer follow-up data for the EECSS remained insufficient. This study is the first clinical trial assessing the long-term effectiveness and safety of the XIENCE PRIME EECSS in a real-world population after its launch in China.
Materials and Methods
Patient Population and Study Design
Patients were recruited from 45 Chinese cardiovascular research centers, and more than 2000 patients with general cardiovascular disorders with interventional treatment were enrolled in the clinical study. This multicenter study was registered at https://clinicaltrials.gov/ct2/show/NCT01894152 (XIENCE PRIME Everolimus Eluting Coronary Stent System (EECSS) China Single-Arm Study (XP China SAS)). From December 2013 to May 2014, a total of 108 hospitalized patients (with 127 lesions) were implanted with XIENCE PRIME EECSS stents at the Department of Cardiology, Shanghai Tenth People’s Hospital, where this research was conducted. The inclusion criteria were as follows: (1) age ≥18 years; (2) patients or their legally authorized representatives signing an informed consent form approved by the Ethics Committee indicating consent to participate in the study (for three patients who underwent emergency surgery, their family members did not arrive in time to provide a signature; therefore, the oral consent of the family members was obtained before surgery, and a supplementary signature was obtained after surgery); and (3) no other stent implantation performed during the procedure. Clinical follow-up was conducted by the investigator/authorizer at 1, 2, and 5 years, either in the hospital/outpatient department or by telephone. The detailed inclusion and procedures for follow-up are shown in Figure 1. The median follow-up duration was 5.1 years. Of the 108 participants, follow-up was completed in 96.3% (104 patients) at 1 year, 93.5% (101 patients) at 2 years, and 83.3% (90 patients) at 5 years (Figure 2). The distribution of underlying diseases was as follows: 69 cases of hypertension, 15 cases of diabetes mellitus (4 of which were insulin-treated diabetes), 14 cases of dyslipidaemia, 3 cases of renal dysfunction, and 10 cases of chronic obstructive pulmonary disease (COPD). The clinical study was approved by the ethics committee of the Shanghai Tenth Hospital, and written informed consent was obtained from all participants.
Procedures
Dual-antiplatelet therapy (DAPT) with aspirin (100 mg/day) plus clopidogrel (75 mg/day) or ticagrelor (90 mg/day) was administered to all patients receiving XIENCE PRIME EECSS stents for at least 1 year, and subsequent treatment strategies were determined by the researchers. If some patients developed adverse clinical symptoms, such as bleeding after antiplatelet therapy, the investigator could decide to decrease the dose or replace it with other antiplatelet treatments. When at least one antiplatelet drug was discontinued for more than 2 months between two successive surveys, persistent discontinuation of DAPT was recorded.
Endpoint Definitions
The primary clinical endpoints were ST and TLF. The other clinical outcome measures contained major adverse cardiovascular events (MACE, including myocardial infarction (MI), stroke, and cardiac death), all-cause death, and target lesion revascularization (TLR); and any coronary revascularization, new coronary revascularization, or complications of massive hemorrhage (including severe and moderate bleeding). The causes of death included cardiac, vascular, and non-vascular causes. ST was defined according to the Academic Research Consortium [10], and divided into four stages (acute, subacute, late, and very late), on the basis of the duration between the intra-stent thrombosis and catheter withdrawal. TLF was composed of cardiac death, target vessel MI, or clinically driven TLR [11]. Myocardial infarction is widely considered a cardiac disease of myocardial ischemia along with elevated myocardial markers. Target lesion revascularization was recorded when revascularization occurred within 5 mm proximal to 5 mm distal to the stent [12].
Statistical Analysis
For dichotomous variables, results were counted as frequencies and percentages of patients. For continuous variables such as age, the statistical results are presented as mean ± standard deviation (SD). Furthermore, Kaplan-Meier analysis and Cox multivariate analysis were applied to evaluate the cumulative rates of clinical events. All detailed data were analyzed by GraphPad Prism version 7.0 (GraphPad Software, USA).
Results
Participant Characteristics
A total of 108 hospitalized patients (with 127 lesions) were enrolled in this single-center clinical study, 76.9% of whom were men. The average age of participants was 65.6 ± 10.8 years, and 25.0% of patients were older than 75 years. Moreover, 30.6% and 8.3% patients had previous PCI and MI, respectively, and 15.7% had a history of stroke. The clinical classifications comprised stable angina (38.0%), unstable angina (28.7%), and acute MI (33.3%); most patients (90.7%) were diagnosed with single-vessel disease. During PCI operation, the use rates of low molecular heparin and GP IIb/IIIa inhibitor were 17.6% and 23.1%, respectively. Furthermore, DAPT (aspirin plus clopidogrel or ticagrelor) was initiated in nearly all participants at hospital discharge, and patients who could not tolerate DAPT were given thienopyridine instead (Table 1).
Baseline Patient Characteristics.
| Patient, number | 108 |
| Patient characteristics | |
| Age, years | 65.6 ± 10.8 |
| Age ≥ 75 | 27 (25.0) |
| Male sex | 83 (76.9) |
| Hypertension | 69 (63.9) |
| Diabetes mellitus | 15 (13.9) |
| Insulin-treated diabetes | 4 (3.7) |
| Dyslipidemia | 14 (13.0) |
| History of smoking | 37 (34.3) |
| Renal dysfunction | 3 (2.8) |
| COPD | 10 (9.3) |
| Ejection fraction, % | 61.2 ± 7.3 |
| Ejection fraction<45% | 3 (2.8) |
| Previous PCI | 33 (30.6) |
| Previous MI | 9 (8.3) |
| History of stroke | 17 (15.7) |
| Clinical characteristics | |
| Clinical presentation | |
| Stable coronary artery disease | 41 (38.0) |
| Unstable angina pectoris | 31 (28.7) |
| Acute MI | 36 (33.3) |
| Extent of coronary artery disease | |
| Single-vessel disease | 98 (90.7) |
| Double-vessel disease | 13 (12.0) |
| Triple-vessel disease | 0 (0) |
| Left main disease | 5 (4.6) |
| Intraoperative medication | |
| Low molecular heparin | 19 (17.6) |
| GP IIb/IIIa inhibitor | 25 (23.1) |
| Myocardium markers | |
| Preoperative TnT | 3.1 ± 3.2 |
| Postoperative TnT | 1.1 ± 2.2 |
| Medications at hospital discharge | |
| Aspirin | 98 (90.7) |
| Clopidogrel | 89 (82.4) |
| Thienopyridine | 2 (18.5) |
| Ticagrelor | 13 (12.0) |
Continuous variables are shown as mean ± SD; categorical variables are presented as frequency counts (proportion).
COPD, chronic obstructive pulmonary disease; PCI, percutaneous coronary intervention; MI, myocardial infarction.
Baseline Characteristics of Lesions and Anti-Platelet Therapy
At baseline, the incidence of culprit lesions in the left anterior descending artery was highest (56.7%), and was followed by the right coronary artery (25.2%), left circumflex artery (14.2%), and left main coronary artery (3.9%). Two (1.6%) of the 127 lesions were treated with the two-stenting strategy. According to the American College of Cardiology/American Heart Association (ACC/AHA) classification, most target lesions were B1 (29.1%), or B2 or C type (68.5%) (Table 2).
Baseline Lesion, Angiographic, and Procedural Characteristics.
| Lesion | 127 |
| Lesion and procedural characteristics | |
| Target lesion | |
| Right coronary artery | 32 (25.2) |
| Left main coronary artery | 5 (3.9) |
| Left anterior descending artery | 72 (56.7) |
| Left circumflex artery | 18 (14.2) |
| Chronic total occlusion | 35 (27.6) |
| In-stent restenosis | 8 (6.3) |
| Ostial lesion | 24 (18.9) |
| Severe angulation | 11 (8.7) |
| Severe calcification | 6 (4.7) |
| Bifurcation | 15 (11.8) |
| Long lesion (LL>30 mm) | 29 (22.8) |
| Small vessel (RVD<2.75 mm) | 14 (11.0) |
| ACC/AHA classification | |
| A | 3 (2.4) |
| B1 | 37 (29.1) |
| B2/C | 87 (68.5) |
| Profile | |
| Smooth | 18 (14.2) |
| Irregular | 109 (85.8) |
| Mean number of stents | 1.1 ± 0.1 |
| Diameter stenosis, % | 86.6 ± 1.0 |
| Lesion length, mm | 23.3 ± 9.4 |
| Procedure characteristics | |
| Total stent length, mm | 27.2 ± 4.3 |
| Two-stenting | 2 (1.6) |
Continuous variables are shown as mean ± SD, and categorical variables are presented as frequency counts (proportion).
Before stent implantation, aspirin (100 mg/day), clopidogrel (75 mg/day), and ticagrelor (90 mg/day) were used in 93.5%, 87.0%, and 10.2% of patients, respectively. After EECSS implantation, the utilization rate of aspirin was decreased to 90.4%, 89.1%, and 81.1%, and the utilization rate of clopidogrel was decreased to 78.8%, 57.4%, and 38.5%, at 1, 2, and 5 years, respectively. However, the utilization rate of ticagrelor was initially increased to 13.5% at 1 year, then gradually decreased to 11.9% and 7.8% at 2 and 5 years (Table 3).
Use of Anticoagulant Drugs in the 2 Years After PCI.
| Index procedure | 0 year | 1 year | 2 year | 5 year | |
|---|---|---|---|---|---|
| Aspirin (100 mg/day) | No. of patients with events | 101 | 94 | 90 | 73 |
| Cumulative incidence | 93.5% | 90.4% | 89.1% | 81.1% | |
| Clopidogrel (75 mg/day) | No. of patients with events | 94 | 82 | 58 | 35 |
| Cumulative incidence | 87.0% | 78.8% | 57.4% | 38.9% | |
| Ticagrelor (90 mg/day) | No. of patients with events | 11 | 14 | 12 | 7 |
| Cumulative incidence | 10.2% | 13.5% | 11.9% | 7.8% |
The incidence of clinical events was evaluated with Kaplan-Meier analysis.
Clinical Outcomes
The median duration of follow-up was 5.1 years, and angiography was performed if cardiac symptoms or new lesion revascularization was observed. The 5-year cumulative incidence of any coronary revascularization was 16.7%, the rate of new lesion revascularization was 11.1%, the rate of any TLR was 6.7%, and the rate of TLF was 3.3% (Figure 3, Table 4).
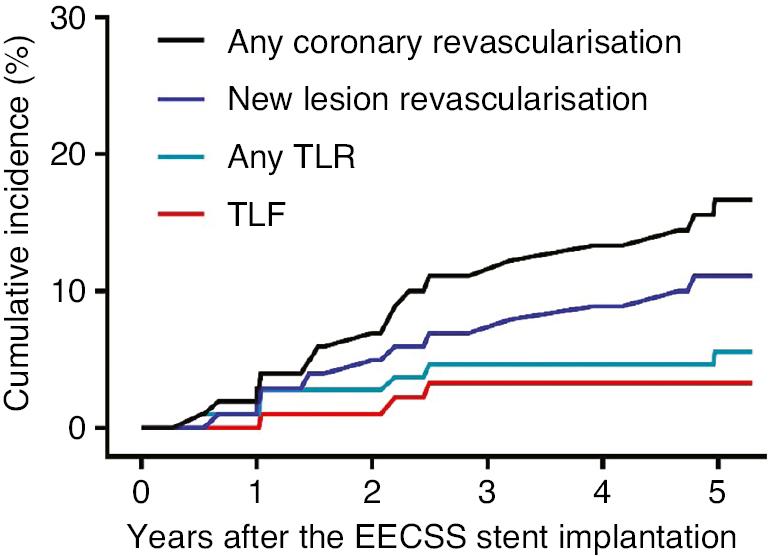
Cumulative Incidence of Any Coronary Revascularization, New Lesion Revascularization, Any TLR, and TLF.
TLR, Target Lesion Revascularization; TLF, target lesion failure.
Cumulative Incidence of Any Coronary Revascularization, New Lesion Revascularization, and Any Target Lesion Revascularization (TLR).
| 1 year No. of events incidence (%) | 2 year No. of events incidence (%) | 5 year No. of events incidence (%) | |
|---|---|---|---|
| Any coronary revascularization | 3 (2.9) | 7 (6.9) | 15 (16.7) |
| New lesion revascularization | 2 (1.9) | 5 (5.0) | 10 (11.1) |
| Any TLR | 1 (1.0) | 3 (3.0) | 6 (6.7) |
| TLF | 0 (0) | 1 (1.0) | 3 (3.3) |
| MACE | 2 (1.9) | 4 (4.0) | 9 (10.0) |
| All-cause death | 1 (1.0) | 2 (2.0) | 5 (5.6) |
| Cardiac death | 0 (0) | 1 (1.0) | 2 (2.2) |
| Myocardial infarction | 0 (0) | 2 (2.2) | 3 (3.3) |
| ST | |||
| Definite ST | 0 (0) | 0 (0) | 0 (0) |
| Probable ST | 0 (0) | 0 (0) | 0 (0) |
| Possible ST | 0 (0) | 0 (0) | 1 (1.1) |
| Bleeding | |||
| BARC ≥ 3 | 1 (1.0) | 3 (3.0) | 4 (4.4) |
| BARC ≥ 2 | 3 (2.9) | 6 (5.9) | 9 (10.0) |
The incidence of clinical events was evaluated with Kaplan-Meier analysis.
TLR, target lesion revascularization; TLF, target lesion failure; MACE, major adverse cardiovascular events; ST, stent thrombosis; BARC, Bleeding Academic Research Consortium.
The cumulative rate of MACE was 1.9% at 1 year, which gradually increased to 4.0% at 2 years and finally to 10.0% at 5 years. We measured other clinical outcomes including all-cause death (5.6%), cardiac death (2.2%), and MI (3.3%) (Figure 4, Table 4). Only five deaths occurred during the entire follow-up: two patients died of cardiovascular disorders, and the others died of respiratory failure and liver cancer. During the 5-year follow-up duration, no definite or probable ST was observed, and only one possible ST appeared at a very late stage.
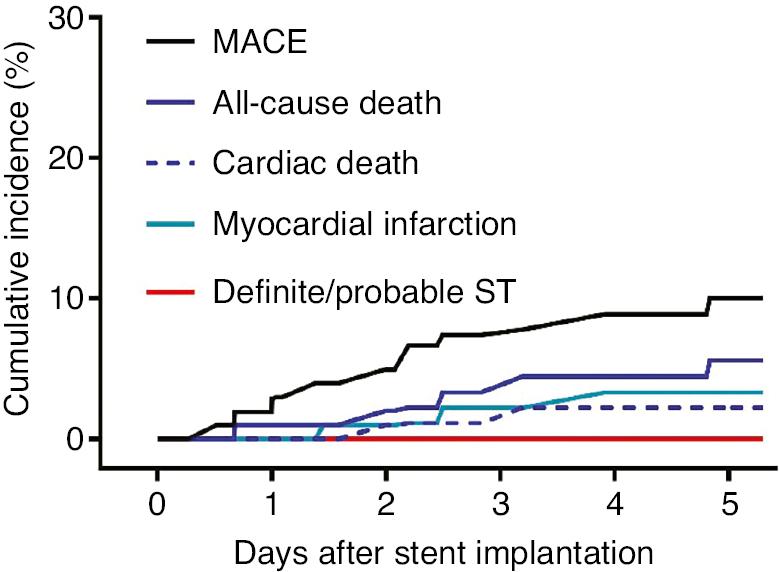
Cumulative Incidence of MACE, All-Cause Death, Cardiac Death, Myocardial Infarction, and Definite/Probable ST.
MACE, major adverse cardiovascular events.
Furthermore, the cumulative rate of bleeding events (BARC ≥ 2) was 5.9% at 2 years, and increased by approximately 1.4% per year in the 2 years after the stent implantation. The incidence of major bleeding events (BARC ≥ 3) was 4.4% at 5 years and increased approximately 0.9% per year after the PCI surgery (Figure 5, Table 4). Three patients had aspirin-induced gastrointestinal bleeding, two patients had intracranial bleeding, and one patient experienced stroke hemiplegia.
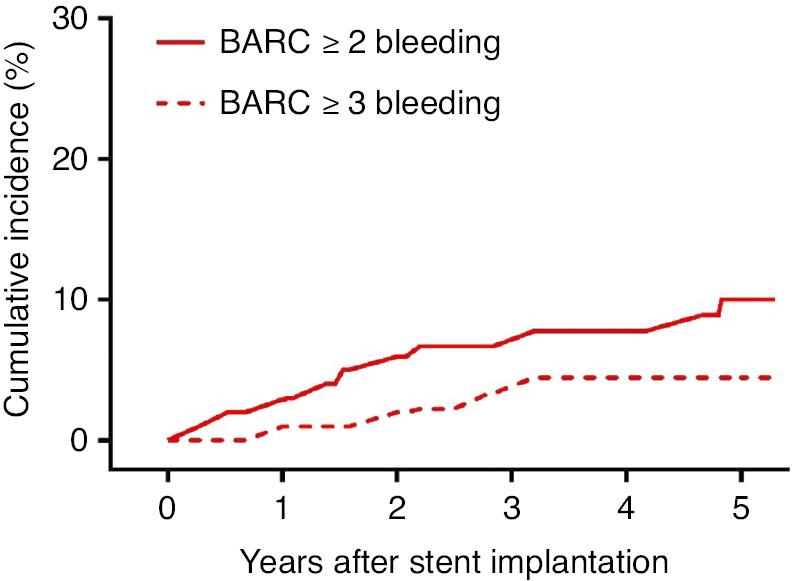
Cumulative Incidence of Bleeding Events According to the Bleeding Academic Research Consortium (BARC) Classification.
The Cox multivariate model was used for prognostic analysis (Table 5). With the number of days of survival from MACE as the time variable; the occurrence of MACE as outcome variable; and age, sex, history of smoking, alcohol, diabetes, hypertension, hyperlipidemia, COPD, history of stroke, ejection fraction, number of lesioned vessels, preoperative TnT, and DAPT as concomitant variables, the results revealed a significant positive association between the occurrence of MACE and history of stroke (RR, 5.812; 95% CI, 1.127–9.967, P=0.035) and preoperative TnT (RR, 1.430; 95% CI, 1.150–1.778, P=0.001), and a significant inverse association between the occurrence of MACE and DAPT (RR, 0.156; 95% CI, 0.030–0.801, P=0.026) (Table 5).
Results of Cox Multivariate Analysis for Factors Correlated with Major Adverse Cardiovascular Events.
| Factors All participants (n=108) | RR | 95% CI | P value |
|---|---|---|---|
| History of stroke | 5.812 | 1.127–9.967 | 0.035* |
| Postoperative TnT | 1.430 | 1.150–1.778 | 0.001** |
| DAPT | 0.156 | 0.030–0.801 | 0.026* |
RR, relative risk; CI, confidence interval; TnT, troponin t; DAPT, dual-antiplatelet therapy, with aspirin (100 mg/day), and clopidogrel (75 mg/day) or ticagrelor (90 mg/day) for at least 1 year. *indicates statistically significant P-values; *P < 0.05, **P < 0.01.
Discussion
In this study, we implanted XIENCE PRIME EECSS in 108 patients with CAD, and performed a clinical follow-up for 5 years. We obtained the following findings: (1) the 5-year clinical outcomes after EECSS implantation were favorable, and ST and TLF occurred in only several patients; (2) although the cumulative rate of new revascularization increased, this trend gradually slowed and reached a plateau; (3) the incidence of major bleeding events gradually increased but maintained a relatively low rate; and (4) after Cox regression analysis, history of stroke, postoperative TnT, and DAPT were independent risk factors for the occurrence of MACE.
Stent thrombosis is a life-threatening clinical syndrome after stent implantation [13]. Aoki et al. [14] have studied prognosis and safety of 1704 patients with cobalt-chromium everolimus-eluting stents (Co-Cr EES) for 5 years, and observed only nine cases (0.5%) of stent thrombosis. Among the 108 patients enrolled in this study, no definite ST cases, and only one case of possible late stent thrombosis were observed; these rates are lower than those in all studies of drug-eluting stents in other patient groups, including the Resolute study (randomized, controlled, noninferiority comparison of the Resolute stent research) (0.7%) [15], Compare study (second generation EES versus paclitaxel-eluting stent in real world) (0.7%) [16], and XIENCE V USA study (0.96%) [17]. No late definite ST occurred, although the use of DAPT was less than 40% in the fifth year of this study. Notably, the cumulative rate of TLF was only 3.3% at 5 years, a value was much lower than that in the previous XIENCE V study (10.2%) [14].
MACE occurred in only nine patients (10.0%) until the fifth year, a value slightly lower than the 10.7% reported in the previous study Co-Cr EES (XIENCE V/PROMUS) in Japan [14]. This finding might be because XIENCE V and XIENCE PRIME had similar efficacy and safety, according to the safety evaluation primary report of the XIENCE V USA study (XIENCE V Everolimus Eluting Coronary Stent System Condition-of-Approval Post-Market study) [9], and it took similar time in these two studies. Moreover, 3.3% of patients had any MI at 5 years, a higher percentage than that in the Japanese Co-Cr EES study, whereas cardiac death was 2.2% lower than that in the Co-Cr EES (3.8%). Furthermore, other clinical endpoints including any TLR (6.7%) were also lower than observed in previous clinical research on biodegradable polymer-coated biolimus-eluting stents (16.3%) [18] and Co-Cr EES (7.2%) [14] in Japan. Above all, these results demonstrated that the XIENCE PRIME EECSS has good safety in Chinese patients. Moreover, the incidence of new lesion revascularization was nearly double that of TLR, similarly to the results of a long-term observational study in Japan [18] but inconsistent with Stone et al.’s conclusion that the incidence of subsequent cardiovascular events is almost equal for culprit and non-culprit lesions [19], possibly because some patients with poly-vascular disease and diabetes were enrolled in the study. In addition, the cumulative rate of major bleeding events increased progressively and eventually reached a relatively low level of 4.4% at 5 years. Although DAPT has been shown to greatly increase the probability of major bleeding [20], its utilization was less than 40% in the fifth year of this study.
In contrast to the aforementioned studies on other drug-eluting stents, this study showed better safety and efficacy, possibly because of the following reasons. First, the application rate of DAPT in the first year was above 90% in this study, a percentage higher than those in most similar studies, and Cox regression analysis revealed that the application of DAPT significantly decreased the incidence of MACE during the 5-year follow-up. Second, the XIENCE PRIME EECSS is an upgraded version of the XIENCE V EECSS with better propulsion and flexibility. Third, the study participants were enrolled in 2013–2014, later than most of the above studies; therefore, the better available patient management, intervention strategies and techniques, and cardiovascular medicine might have positively influenced clinical efficacy. Finally, this was a single-center study in Shanghai, where the medical care and patient compliance were higher than the overall average in other studies.
This study has several limitations. The observational, non-randomized, one-arm design is the first limitation of this study, although the design is comparable to those of similar clinical trials of drug eluting stents in Japan and the USA. Second, this was only a single-center study with a small sample size. Finally, more than 2 years after stent implantation, coronary angiography was performed only if cardiac symptoms appeared, thus resulting in statistically lower values of clinical events than the actual values.
Conclusion
The 5-year clinical outcomes after XIENCE PRIME EECSS implantation were favorable, and ST and TLF occurred in a very small percentage of patients. Although the cumulative rate of new revascularization has increased over time, this increase has gradually slowed and reached a plateau. The incidence of major bleeding events gradually increased but remained moderate. History of stroke, postoperative TnT, and DAPT were identified as independent risk factors for the occurrence of 5-year MACE. Therefore, XIENCE PRIME EECSS implantation is safe and feasible for the treatment of CAD.



