Significance Statement: Right ventricular pacing disrupts atrioventricular synchrony and increases the risk of atrial fibrillation (AF). Whether algorithms for minimizing ventricular pacing reduce the incidence of AF remains controversial. This systematic review and meta-analysis demonstrates that minimizing ventricular pacing significantly reduced the incidence of AF, with an odds ratio of 0.74. This has important implications for the long-term prognosis of these patients.
Introduction
Pacemaker implantation is indicated in bradyarrhythmias, including sinus node dysfunction, bifascicular block, trifascicular block, and atrioventricular (AV) block [1]. Standard ventricular pacing is normally performed from the right ventricular (RV) apex, owing to the long-term stability this position offers and the ease with which it can be accessed for lead placement. Many recently conducted clinical studies have illustrated that RV pacing is associated with an increase in the risk of atrial fibrillation (AF), among other adverse effects [2]. Conventional RV pacing disrupts the ventricular synchrony that is observed in a normal heart. This may lead to mitral insufficiency, left atrial enlargement, and left ventricular (LV) hypertrophy, which collectively predispose to AF [3].
A contemporary approach to circumvent the detrimental effects associated with RV pacing is the incorporation of pacing algorithms in dual-chamber pacemakers. These algorithms focus on preserving physiological ventricular activation as much as possible, with periodic changes in pacing mode and/or timing cycles in an attempt to minimize ventricular pacing [4]. However, whether minimizing ventricular pacing actually decreases the incidence of AF remains controversial. While certain randomized controlled trials (RCTs) have demonstrated that minimizing ventricular pacing, mediated by novel pacing algorithms, is associated with a reduction in AF incidence [5–9], others have shown no such correlation [2, 10–14]. Therefore, we conducted a systematic review and meta-analysis of RCTs with the aim of comparing the incidence of AF between minimizing ventricular pacing and conventional pacing protocols in patients with permanent pacemakers.
Methods
Search Strategy, Inclusion Criteria, and Quality Analysis
This systematic review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [15]. It has been registered with PROSPERO. PubMed, Embase, and the Cochrane Library were searched up to August 1, 2017, with no language restriction, for studies that investigated the incidence of AF with and without minimizing ventricular pacing. The following search terms were used: [“minimizing right ventricular pacing” or “managed ventricular pacing” or “minimize ventricular pacing” or “ventricular pacing reduction”]. The following inclusion criteria were applied: (1) the design of the study was an RCT in humans; (2) AF incidence was reported for minimizing ventricular pacing and nonminimizing ventricular pacing modes. Quality assessment of RCTs was performed with the Cochrane Collaboration’s tool (Supplementary Figures 1 and 2) [16].
Data Extraction and Statistics
Data from the different studies were entered in a prespecified spreadsheet in Microsoft Excel. All potentially relevant reports were retrieved as complete articles and assessed for compliance with the inclusion criteria. In this meta-analysis, the extracted data elements consisted of (1) publication details (last name of first author, publication year, and location); (2) the follow-up duration; (3) the end points; (4) the quality score; and (5) the characteristics of the population, including sample size, sex, age, and number of participants. Two reviewers (I.L. and M.G.) independently reviewed each included study, and disagreements were resolved by adjudication with input from a third reviewer (G.T.). The end point for this meta-analysis was incident AF. Event rates for AF were extracted for each study. This enabled the calculation of odds ratios, which were then pooled in the meta-analysis.
Heterogeneity between studies was determined with Cochran’s Q, which is the weighted sum of squared differences between individual study effects and the pooled effect across studies, and the I 2 statistic from the standard chi-square test, which is the percentage of the variability in effect estimates resulting from heterogeneity. I 2>50% was considered to reflect significant statistical heterogeneity. A fixed-effects model was used if I 2<50%; otherwise the random-effects model using the inverse variance heterogeneity method was selected. To locate the origin of the heterogeneity, sensitivity analysis excluding one study at a time was performed. Funnel plots, the Begg and Mazumdar rank correlation test, and Egger’s test were used to detect possible publication bias.
Results
Figure 1 shows a flow diagram detailing the search strategy and study selection process. In total, 215 publications were retrieved from PubMed and 115 were retrieved from Embase. A total of 11 studies involving 5705 patients (mean age 71 years [standard deviation 11 years], 61% males) were included. The baseline characteristics of these studies are listed in Table 1. The mean follow-up duration was 24 months. Our meta-analysis shows that use of algorithms for minimizing ventricular pacing significantly reduced the odds of developing AF (odds ratio 0.74; 95% confidence interval 0.55–1.00; P<0.05; Figure 2). Cochran’s Q was greater than the number of degrees of freedom (27 vs. 10), suggesting the true effect size was different among the various studies. Moreover, I 2 was 63%, indicating the presence of moderate heterogeneity. This was potentially due to variations in study protocols, ranging from differences in sample sizes and follow-up periods to the definitions of incident AF used. Sensitivity analysis by a leave-one-out study did not significantly alter the pooled odds ratio (Supplementary Figure 3). Funnel plots plotting standard error or precision against the logarithm of the odds ratio are shown in Supplementary Figures 4 and 5, respectively. Begg and Mazumdar rank correlation suggested no significant publication bias (Kendal’s τ was −0.2; P>0.05). Egger’s test demonstrated no significant asymmetry (intercept−0.8, t=0.5; P>0.05).
Flow Diagram Detailing the Search Strategy and Study Selection Process for This Systematic Review and Meta-Analysis of the Effects of Minimizing Ventricular Pacing in Reducing the Incidence of Atrial Fibrillation (AF).
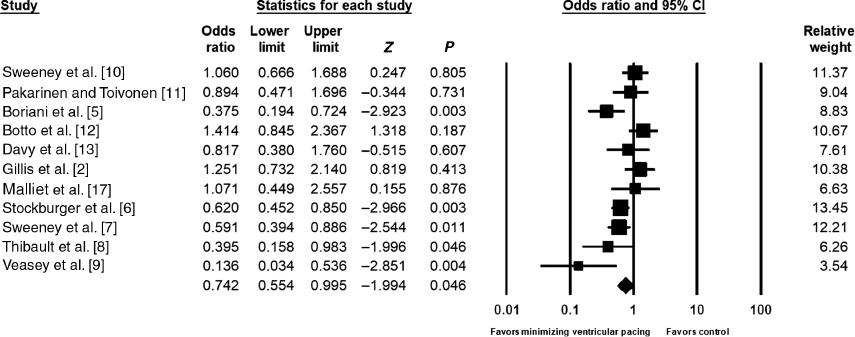
Odds Ratios for Included Studies Examining the Effects of Minimizing Ventricular Pacing Algorithms on the Incidence of Atrial Fibrillation.
Meta-analysis of 11 randomized controlled trials revealed that the use of algorithms for minimizing ventricular pacing significantly reduced the odds of developing atrial fibrillation. CI, confidence interval.
Characteristics of the Studies Included in This Meta-Analysis.
| Study | Sample size (n) | Algorithm for minimizing ventricular pacing (n) | Controls (n) | Average age (years) | No. of males | Name of trial | Comparison | AF definition | Follow-up duration (months) |
|---|---|---|---|---|---|---|---|---|---|
| Sweeney et al. [10] | 1030 | 518 | 512 | 62 | 818 | MVP | Managed ventricular pacing vs. VVI | Persistent AF | 29 |
| Pakarinen and Toivonen [11] | 389 | 195 | 194 | 73 | 180 | – | VIP™ on vs. VIP™ off | AF | 12 |
| Boriani et al. [5] | 768 | 383 | 385 | 74 | 583 | MINERVA | DDDRP + managed ventricular pacing vs. DDDRP | Permanent AF | 24 |
| Botto et al. [12] | 605 | 299 | 306 | 75 | 365 | Prefer for Elective Replacement Managed Ventricular Pacing | Managed ventricular pacing + DDD vs. DDD | Persistent AF | 24 |
| Davy et al. [13] | 422 | 141 | 281 | 73 | 211 | – | SafeR™ vs. DDD | Permanent AF | 12 |
| Gillis et al. [2] | 258 | 129 | 129 | 72 | 98 | – | Managed ventricular pacing vs. DDDRP | At least one episode of AT/AF | 6 |
| Malliet et al. [17] | 97 | 49 | 48 | – | – | – | SafeR™ vs. DDD | AF | 35 |
| Stockburger et al. [6] | 632 | 314 | 318 | 72 | 358 | ANSWER | SafeR™ vs. DDD | AF | 36 |
| Sweeney et al. [7] | 1065 | 530 | 535 | 72 | 520 | SAVE PACe | Dual-chamber minimal ventricular pacing vs. conventional dual-chamber pacing | Persistent AF | 20 |
| Thibault et al. [8] | 373 | 191 | 182 | 71 | 233 | CAN-SAVE R | SafeR™ vs. DDD | AF | 36 |
| Veasey et al. [9] | 66 | 33 | 33 | 75 | 37 | MinVPACE | Managed ventricular pacing vs. DDDRP | AF | 17 |
Advanced pacemakers feature atrial preventive pacing and atrial antitachycardia pacing (DDDRP). AF, atrial fibrillation; AT, atrial tachycardia; DDD: dual-chamber pacing; VVI, backup-only ventricular pacing.
Discussion
This study was a systematic review and meta-analysis of 11 RCTs evaluating the effects of minimizing ventricular pacing on the incidence of AF in 5705 patients. The main finding was a 26% reduction in the risk of incident AF with use of algorithms for minimizing ventricular pacing compared with no use.
Electrical activation of the heart typically occurs through the cardiac conduction system. Many defects that may manifest themselves in this normal pathway are treated by pacemaker implantation, involving the RV apex. Pacing of the right ventricle is nonphysiological, and provides a route through which the depolarization wave can circumvent the standard pattern of myocardial activation. As such, this leads to delayed stimulation of the left ventricle akin to that in left bundle branch block [18], in turn culminating in ventricular dyssynchrony, along with an increased risk of mitral insufficiency, left atrial enlargement, and LV hypertrophy, all of which collectively induce AF [3]. The underlying pathophysiological mechanisms consist of atrial structural remodeling via fibrosis: an electrophysiological substrate that predisposes to arrhythmogenesis by reducing the conduction velocity of action potentials propagating through the atria [19]. The necessity of pacing for patients with bradyarrhythmias coupled with its evident indirect role in AF pathogenesis presents a clinical dilemma, wherein artificial stimulation needs to be delivered at just the optimal dosage so as to maximize treatment while preventing the development of detrimental atrial arrythmias.
Conventional dual-chamber pacemaker strategies, albeit effective in sustaining AV synchrony, involve higher incidences of pacing that, as already mentioned, limit therapeutic benefit by enhancing ventricular dysfunction [20]. One solution proposed to overcome this problem is the use of alternative pacing sites instead of the RV apex, aiming to conduct the electrical wavefront in a more physiological manner throughout the heart [21]. The primary anatomical locations that have been investigated include the RV outflow tract, the intraventricular septum, and the bundle of His. Although some evidence suggests the superiority of such sites over apical pacing in terms of dyssynchrony burden as well as LV function and structure [22, 23], there are also data supporting the contrary [24]. More recently, however, the advent of algorithms for minimizing ventricular pacing provides another possible answer to the issue of unwanted, excessive pacing. These algorithms accomplish this by prolonging the AV interval such that intrinsic AV conduction, if present, is maintained and ventricular stimulation, in turn, is delivered only in the absence of an AV impulse within the elongated period [25]. The feasibility of these algorithms has been heavily scrutinized with the conduction of many RCTs intending to examine the effect of different ventricular pacing reduction modalities compared with conventional dual-chamber pacing on the incidence of AF. A meta-analysis of the data from seven such RCTs failed to demonstrate a significant difference in the occurrence of new-onset AF in these two groups [26]. However, this investigation excluded certain trials because of their crossover study design, leading to exclusion of some key cohorts. In contrast, our meta-analysis included data from four additional trials, with the pooled-effect estimate showing a significant benefit in minimizing ventricular pacing for reducing incident AF. Given this apparent therapeutic advantage, together with its additional benefits, such as increasing device longevity and reducing the need for device replacement and its subsequent complications, the incorporation of algorithms for minimizing ventricular pacing in routine clinical practice should in theory lead to a decrease in AF-related morbidity and mortality.

