Introduction
Spontaneous coronary artery dissection (SCAD) is a relatively rare type of acute coronary syndrome (ACS). SCAD causes ACS in 1.7–4% of cases [1]. SCAD often occurs in patients without conventional cardiovascular risk factors and occurs mainly in females, especially young or middle-aged women [2]. However, SCAD with peripheral artery dissection is rarely reported. Here we report a case of SCAD (involving the left main coronary artery, left anterior descending coronary artery, and left circumflex artery) in a 59-year-old woman who presented with ACS and iliac artery dissection.
Case Report
A 59-year-old woman presented to the outpatient department after experiencing recurrent episodes of chest pain at rest for 2 days. The chest pain radiated to the dorsal part of the shoulder and lasted for 30min. One hour before admission, the patient experienced persistent and severe chest pain after emotional stress. She reported not having had hypertension and other diseases before, and also reported the same for her family. She had gone through menopause and reported not having had abnormal menstruation. Her blood pressure at admission was 219/133 mmHg and her heart rate was 96 beats per minute. The patient had never measured her blood pressure before, so her blood pressure and hypertension history was uncertain. She had not taken any regular antihypertensive treatment before. Physical examination showed equal pulses in all extremities and no heart murmur. The electrocardiogram (ECG) and troponin level were abnormal. The ECG in the outpatient department revealed a sinus rhythm with flat T waves in inferior and anterior leads (Supplementary Figure S1). At admission, the ECG demonstrated ST elevation in leads V1 and V2, inverted T waves in leads V4–V6, and ST depression in leads II, III, and aVF (Supplementary Figure S2). The level of cardiac troponinI was 0.172 μg/L (normal range 0.000–0.08 μg/L) and that of cardiac troponin T was 0.108 μg/L (normal range 0.000–0.100 μg/L). The level of D-dimer was normal (0.1 mg/L).
Because of the changes on the ECG, cardiac biomarkers, and the symptom of persistent chest pain, ACS was considered. Immediate diagnostic coronary angiography showed that the proximal portion and the midportion of left anterior descending coronary artery exhibited diffuse stenosis (about 60–70%) and the distal segment of the left circumflex artery exhibited mild stenosis (about 50–60%). The right coronary artery was normal. According to the results of coronary angiography, SCAD was considered, so intravascular ultrasonography (IVUS) was performed. IVUS revealed an intramural hematoma (IMH) in the ostium of the left main coronary artery extending to the midportion of the left anterior descending coronary artery and the proximal portion and the midportion of the left circumflex artery (Figure 1). As the IMH involved the left main coronary artery and the blood pressure was very high, multislice spiral CT angiography of the aorta was performed and excluded dissection of the ascending aorta, but revealed bilateral iliac artery dissection without a rupture (Figure 2).
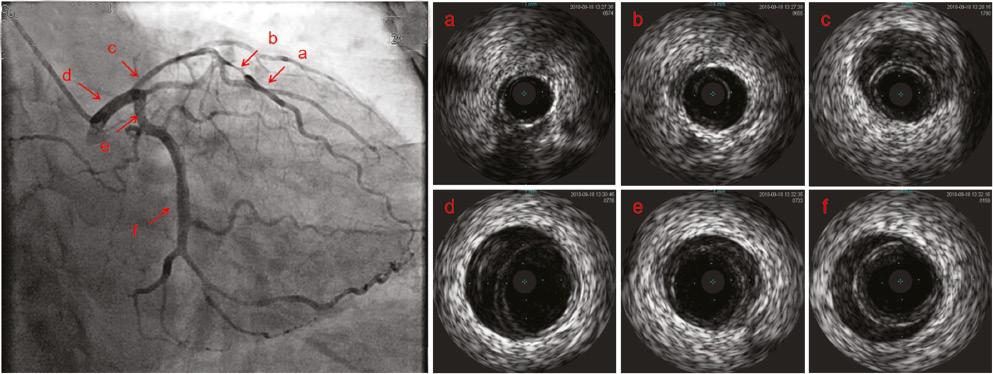
Coronary Angiography Showed That the Proximal Portion and the Midportion of the Left Anterior Descending Coronary Artery Exhibited Diffuse Stenosis (about 60–70%) and the Distal Segment of the Left Circumflex Artery Exhibited Mild Stenosis (about 50–60%).
The right coronary artery was normal. Intravascular ultrasonography showed an intramural hematoma in the ostium of the left main coronary artery (c, d) extending to the midportion of the left anterior descending coronary artery (a, b) and the proximal portion and the midportion of left circumflex artery (e, f).
As her chest pain was relieved and her general condition was stable, percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) was not performed, and conservative medical treatment was selected. She was treated by intravenous pumping of urapidil and esmolol to control her blood pression and heart rate. The initial pumping rate of esmolol was 0.01 mg/kg/min and the initial pumping rate of urapidil was 0.2 mg/min. We adjusted the pumping rate according to the blood pressure and heart rate. The oral antihypertensive drugs metoprolol and nifedipine as controlled-release tablets and losartan-hydrochlorothiazide were prescribed at the same time. The blood pressure was maintained below 130/80 mmHg and the heart rate was maintained below 70 beats per minute.
For treatment of iliac artery dissection, surgical or endovascular treatment of the iliac artery may be not necessary at this moment, because the patient had no symptoms of lower limb ischemia and the pulses in the peripheral arteries were well palpable and equal. At 7 days after admission, there was no recurrence of chest pain. After discharge, she continued to take oral medication. She remained free of chest pain and had experienced no cardiac events at a 12-month telephone follow-up. Also she had no pain at rest, numbness, or intermittent limp of the lower limbs as a result of the iliac artery dissection.
Discussion
As far as we know, this is the first case of ACS with SCAD (involving the left main coronary artery, left anterior descending coronary artery, and left circumflex artery) and iliac artery dissection. Usually, SCAD is a nonatherosclerotic coronary artery dissection, resulting in the formation of an IMH or false lumen in the coronary artery wall. SCAD predominantly occurs in women in their 40s and 50s, but it can happen in earlier or later years. Among women, SCAD is the cause of ACS in as high as 35% of cases [3]. The cause of SCAD is still uncertain. Current studies show that hormonal influences, underlying acquired or genetic arteriopathies, systemic inflammatory diseases, female sex hormones and pregnancy, fibromuscular dysplasia (FMD), precipitants, and triggering factors such as precipitating emotional stress, Valsalva-like maneuvers such as coughing or retching, use of recreational drugs, intense exercise, and molecular and genetic factors such as inherited connective tissue disorders are associated with SCAD [4–6].
Isolated iliac artery dissection is a very rare disease, and there are only a few case reports. In those articles, the possible causes included hypertension, atherosclerosis, FMD, connective tissue disease, trauma, and pregnancy [7]. FMD is a nonatherosclerotic vascular disease that results in occlusion, aneurysm, or dissection of the artery. The gold standard for imaging of FMD is arterial angiography. The characteristic changes of angiography is beaded, tubular or diverticular. The most commonly involved vascular beds are the renal arteries (79.7%) and the extracranial carotid arteries (74.3%) [8, 9]. In this case, arterial angiography and CT angiography did not show the changes mentioned above. The 59-year-old woman with SCAD and iliac artery dissection most possibly experienced uncontrolled hypertension.
The clinical presentation of SCAD is similar to that of ACS. But the angiogram can differentiate SCAD from atherosclerosis. According to the angiogram, SCAD is classified in three types. Type 1 refers to the appearance of a double lumen under contrast medium staining of the arterial wall. Type 2 appears as diffuse (usually >20 mm) and smooth narrowing that can differ in severity and length (depending on the hematoma). Type 3 SCAD mimics atherosclerosis with focal or tubular stenosis and can be confirmed only under intravascular visualization [1, 5, 10, 11]. Type 2 is the most common angiographic manifestation of SCAD. The patient appeared to have type 2 SCAD. Current guidelines recommend further assessment with intravascular imaging, which can aid in the diagnosis of SCAD in angiographically subtle or nondiagnostic cases [5, 12]. Therefore, intracoronary imaging was needed and was required for further management.
The short-term and long-term management of SCAD is not well developed, as there have been no randomized trials assessing the optimal strategy. Currently, conservative treatment strategies are mostly used for SCAD, but active intervention (PCI or CABG) may be required for patients with lesions with high-risk characteristics, such as left main coronary artery lesion or hemodynamic instability. Early PCI is widely advocated in ACS secondary to atherosclerotic heart disease, but there are no randomized data to support PCI in ACS caused by SCAD. Many observational studies have shown an increased risk of coronary complications from PCI performed in SCAD [4, 13–15]. The following factors are correlated: balloon dilation and stent placement may cause worse vessel obstruction, as they can increase the risk of extending dissection of the intima or propagating the IMH upstream and downstream of the vessel; IMH naturally resorbs over time, which can result in subacute and late stent strut malapposition, increasing the risk of stent thrombosis in the future; SCAD most frequently involves the distal coronary segments, which may be too small or too distal for stent implantation; PCI may require long coronary stents as the dissection lengths are often extensive, which can increase the risk of subsequent in-stent restenosis and stent thrombosis [5, 12].
In this case, PCI was difficult and had limitations. Firstly, IVUS showed that the IMH involved left main coronary artery bifurcation. Secondly, the diameters of the left anterior descending coronary artery and the left main coronary artery were different. Thirdly, the diameter of external elastic membrane in the left main coronary artery measured by IVUS was more than 6 mm. The diameter was too large for stent implantation, and it would lead to incomplete stent apposition. CABG is recommended for patients with left main coronary artery disease, multivessel involvement, failure of coronary intervention, or refractory myocardial ischemia. Some studies have shown that most SCAD lesions can heal spontaneously [2, 16]. So CABG may be limited to initially preserving coronary flow to the at-risk myocardium until the SCAD has healed and native coronary flow has been restored [4]. In this case, the woman refused CABG and chose conservative treatment. She was clinically stable and without objective evidence of ongoing ischemia, so conservative therapy may be recommended [5].
Mostly, experts still recommend aspirin for at least 1 year following SCAD in the absence of complications. However, given current uncertainty of the benefits versus risks, decisions for antiplatelet use in SCAD patients should be tailored to each individual case [5, 12]. We did not prescribe aspirin for our patient because she also had iliac artery dissection. Statin therapy is not recommended routinely after SCAD but is reserved for patients meeting guideline-based indications for primary prevention of atherosclerosis [17]. Beta-blockers may help to reduce the risk of recurrence [18].
Statements recommend that patients with acute SCAD be observed in hospital for up to 5 days [5, 12]. This patient was hospitalized for 7 days. Up to the time of writing, she remained free of chest pain and felt well during our telephone follow-up.
Conclusion
Accurate recognition and diagnosis of SCAD in patients with ACS is crucial, as the management and outcomes of SCAD differ substantially from those of atherosclerotic coronary disease. Extracoronary vascular abnormalities are strongly associated with SCAD. Treatment should be individualized. PCI and CABG need to be carefully considered and individually. A conservative approach could be used to treat a clinically stable patient with SCAD involving the left main coronary artery.


