Introduction
Antiplatelet treatment is the cornerstone of treatment for patients with acute coronary syndrome (ACS) undergoing percutaneous coronary intervention (PCI) [1–3]. Dual antiplatelet therapy with clopidogrel and aspirin is currently the antiplatelet treatment of choice for prevention of major adverse cardiovascular events, stent thrombosis, etc. [4, 5]. However, despite regular administration of clopidogrel, a number of patients continue to have adverse cardiac events [6, 7]. Many in vitro studies have pointed out that individual response to clopidogrel is not unified. Responses to clopidogrel are influenced by interindividual and intraindividual variability [8, 9]. Notably, there is a growing amount of evidence that recurrence of ischemic complications may be due to the poor response to clopidogrel [10]. Patients who are poor responders or who have high on-clopidogrel platelet reactivity (HPR) to adenosine diphosphate (ADP) are at increased risk of post-PCI ischemic events [11]. The VerifyNow P2Y12 assay is a validated optical turbidimetric point-of-care assay explicitly assessing the effect of P2Y12 receptor blockers [12]. It uses prostaglandin E1 in addition to ADP to increase the levels of intraplatelet cyclic adenosine monophosphate, making the test more sensitive and specific for the effects of ADP mediated by the P2Y12 receptor [13]. The results are expressed as P2Y12 reaction units (PRU). A PRU value greater than 208 was used to define patients with HPR [14]. According to the findings, pre-PCI evaluation of the platelet reactivity provides valuable prognostic information and might guide the therapeutic schedule for those patients who do not fall within the desired therapeutic window [15, 16]. In patients with a low response to clopidogrel and higher ischemic risk, more aggressive antiplatelet strategies might be of use in obtaining PRU values that fall within the desired range. Although impaired platelet response to clopidogrel is a strong predictor of unfavorable outcome after PCI, the impact of HPR to a 600 mg loading dose of clopidogrel before PCI in Chinese patients with ACS undergoing PCI is still unknown. The present study aimed to determine whether in patients with ACS the presence of HPR to a 600 mg loading dose of clopidogrel before PCI was associated with worse outcomes after PCI. Furthermore, we wanted to determine whether there is a relationship between PRU values and some characteristics of the patients. We found the PRU cutoff value before PCI predicted the long-term prognosis.
Material and Methods
Participants
We analyzed 134 consecutive patients in whom platelet function testing was performed about 6 hours after a 600 mg loading dose of clopidogrel before PCI between October 1, 2016, and October 31, 2017. The key enrolment criteria were as follows (Figure 1): (1) age older than 18 years; (2) signed informed consent; (3) guideline-appropriate requirement for PCI, typically based on presentation with ACS. Furthermore, the PCI procedure was performed in accordance with current standard guidelines, and the type of stent implanted and the use of pharmacological agents were at the discretion of the operator. The exclusion criteria were as follows (Figure 1): (1) patients with adult congenital heart disease; (2) upstream use of a glycoprotein IIb/IIIa inhibitor or treatment with oral anticoagulant drugs, platelet count less than 125,000/mL, and high bleeding risk (e.g., active internal bleeding or history of hemorrhagic stroke); (3) coronary artery bypass surgery in the previous 3 months.
Platelet Reactivity Testing
Blood was drawn from the radial artery 6 hours after a 600 mg loading dose of clopidogrel before PCI. The first 5 mL of the blood sample was discarded, and a further sample was collected in a 2-mL tube containing 3.2% sodium citrate.
The VerifyNow point-of-care assay (Accumetrics, San Diego, CA, USA) was used to measure platelet reactivity. The device is a turbidimetry-based optical analyzer that measures platelet-induced aggregation in a system containing fibrinogen-coated beads. This instrument measures changes according to light transmission and the rate of aggregation in whole blood. In the cartridge used for the assay, there is a channel in which inhibition of the P2Y12 receptor can be measured; the channel contains ADP as the agonist of platelets and prostaglandin E1 as a suppressor of free calcium to reduce the nonspecific contribution of ADP binding to the P2Y12 receptor. Samples of venous blood anticoagulated with sodium citrate were tested from each patient 6 hours after a 600 mg loading dose of clopidogrel before PCI. The platelet reactivity was expressed as PRUs. A PRU value greater than 208 was used to define patients with HPR.
Clinical Patient Follow-up
For all patients, follow-up was for 6 months or until a fatal event occurred. Follow-up data were collected from patients by telephone interview or from electronic medical records. If telephone contact could not be established, a mailed questionnaire was used. The primary end point included cardiac death, recurrence of unstable angina, and rehospitalization for target lesion revascularization (TLR). The second end point included bleeding events. The methods used were in accordance with the standard procedure of the Fourth Affiliated Hospital of Harbin Medical University, and included data collection and follow-up under approval by the institutional review board. This study complied with the Declaration of Helsinki and was approved by the local ethics committees, with all patients giving written informed consent.
Statistical Analyses
Continuous variables were expressed as the mean and the standard deviation (SD) and were compared with Student’s t test if applicable. The χ 2 test was used for discrete variables. All statistical tests were two-sided with a significance level of less than 0.05. Multivariate linear regression analysis was used to assess the relationship between the PRU values and the number of variables included. We also assessed the relation between HPR and subsequent clinical outcomes using standard receiver operating characteristic (ROC) curve analyses. All statistical analyses with performed with IBM SPSS Statistics, version 23.0 (IBM, Armonk, NY, USA).
Results
Study Population
Between October 1, 2016, and October 31, 2017, 134 patients with ACS with stents successfully implanted at the Fourth Affiliated Hospital of Harbin Medical University were enrolled. In these patients, platelet function testing was performed about 6 hours after a 600 mg loading dose of clopidogrel before PCI. Clopidogrel and aspirin were prescribed at discharge for all patients and were taken throughout the 6 months of follow-up. One hundred thirty-three patients completed 6 months of follow-up and one patient died. Of the 134 participants in our study, 81 were males. The male-to-female ratio was 1.5:1. The mean age was 60.62 years (SD 9.13 years) and was 59.68 years (SD 8.96 years) in the normal platelet reactivity (NPR) group and 62.41 years (SD 9.27 years) in the HPR group. Statistical analysis of the data obtained (Table 1) revealed that there was not a statistical difference in age, sex, body mass index, presence of diabetes mellitus, presence of hypertension, previous myocardial infarction, and the level of creatinine between the NPR group and the HPR group (P>0.05). As shown in Table 1, a more evident difference concerning hemoglobin level and hematocrit was observed between the NPR group and the HPR group (P<0.05).
Clinical Characteristics.
| Overall population (N = 134) | NPR group (n = 88) | HPR group (n = 46) | P | |
|---|---|---|---|---|
| Age (years) | 60.62±9.13 | 59.68±8.96 | 62.41±9.27 | 0.114 |
| Male | 81 (60.4%) | 55 (62.5%) | 26 (56.52%) | 0.455 |
| Body mass index (kg/m2) | 22.4±2.5 | 22.3±2.4 | 22.5±2.7 | 0.409 |
| Diabetes mellitus | 24 (17.9%) | 15 (17.0%) | 9 (6.5%) | 0.315 |
| Hypertension | 82 (61.19%) | 53 (60.22%) | 29 (63.04%) | 0.513 |
| Previous myocardial infarction | 4 (2.99%) | 2(2.27%) | 2 (4.35%) | 0.514 |
| Previous coronary intervention | 3 (2.24%) | 2 (2.27%) | 1 (2.17%) | 0.964 |
| Left ventricular ejection fraction (%) | 55±7 | 55±7 | 55±8 | 0.807 |
| Creatinine (µmol/L) | 73.31±21.73 | 78.01±21.36 | 73.17±23.69 | 0.241 |
| Hemoglobin (g/dL) | 142.05±17.09 | 145.91±16.62 | 134.76±16.52 | 0.000 |
| Hematocrit (%) | 42.42±4.59 | 43.33±4.49 | 40.71±4.57 | 0.002 |
| Clopidogrel regimen, loading dose 600 mg | 134 | 88 | 46 | 1 |
| Clopidogrel regimen, maintenance dose 75 mg | 134 | 88 | 46 | 1 |
| Aspirin | 134 | 88 | 46 | 1 |
| Statin | 134 | 88 | 46 | 1 |
| P2Y12 reaction units | 165.18±75.22 | 123.01±54.73 | 244.3±30.51 | 0.000 |
| Baseline reaction units | 253.96±40.77 | 243.0±40.11 | 274.59±33.72 | 0.000 |
Values are the mean ± the standard deviation or the number and percentage.
HPR, high platelet reactivity; NPR normal platelet reactivity.
Adverse Events
Among the 134 patients analyzed, 46 patients had HPR (34.3%) and 88 did not have HPR (65.7%). One hundred thirty-three patients completed 6 months of follow-up, and the results showed that clinical events were not rare (Table 2). Through the 6 months of follow-up, 14 patients (10.44%) had ischemic cardiac events (cardiac death, unstable angina, and rehospitalization for TLR) and one patient (0.75%) had minor bleeding. Furthermore, we found that there was a significant difference in clinical ischemic events between HPR and NPR patients. Compared with the NPR group, the HPR after a 600 mg loading dose of clopidogrel before PCI was strongly related to cardiac death, recurrence of unstable angina, and rehospitalization for TLR (P=0.029). We performed multivariate analysis to identify the univariable predictors using candidate predictors such as PRU value, age, sex, diabetes, hemoglobin level, aspartate transaminase level, alanine transaminase level, blood urea nitrogen level, creatinine level, and creatinine clearance. The results indicate that hemoglobin level was a consistent predictor of the PRU value. Male sex also correlated with the PRU value. Multivariate analysis identified hemoglobin level and sex as independent predictors of the PRU value (y=456.355−1.736x 1−31.880x 2, P < 0.05). To assess the impact of pre-PCI platelet aggregation on the primary outcome measure, we stratified the study patients according to their platelet aggregation value using cutoff points derived from ROC curve analysis (Figure 2). ROC curve analysis was used to evaluate the ability of the PRU value to distinguish between patients with and patients without ischemic cardiac events, at 6 months. We defined the ischemic cardiac events as death, recurrence of unstable angina, or rehospitalization for TLR. Through the ROC curve analysis, PRU values could significantly discriminate between patients with and patients without cardiac death, unstable angina, and rehospitalization for TLR (area under the curve 0.758, 95% confidence interval 0.62–0.85, P < 0.001). By ROC curve analysis, the optimum PRU cutoff value for definitely probable was 176.
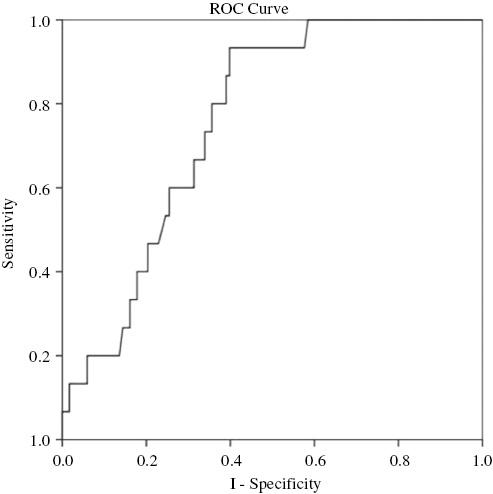
Ischemic Events were Defined as Death, Recurrence of Unstable Angina, or Rehospitalization for Target Lesion Revascularization.
A receiver operating character (ROC) curve analysis was used to evaluate the ability of the P2Y12 reaction unit (PRU) value to distinguish between patients with and patients without ischemic cardiac events at 6 months. Through the ROC curve analysis, PRU values could significantly discriminate between patients with and patients without cardiac death, unstable angina, and rehospitalization for target lesion revascularization (area under the curve 0.758, 95% confidence interval 0.62–0.85, P < 0.001). By ROC curve analysis, the optimum PRU cutoff value for definitely probable was 176.
Distribution and Type of Outcome at 6 Months According to Aspirin and Clopidogrel Antithrombotic Therapy.
| Overall population (N = 134) | NPR group (n = 88) | HPR group (n = 46) | P | |
|---|---|---|---|---|
| Primary outcome: composite of MACE (death, myocardial infarction, or TLR) | 14 | 6 (includes TLR) | 8 (7 had TLR and 1 died) | 0.029 |
| Secondary outcome: minor bleeding | 1 | 1 |
MACE, major adverse cardiovascular events; TLR, target lesion revascularization.
Discussion
The 2016 Chinese guidelines for the use of antiplatelet therapy in cardiovascular disease recommend the use of antiplatelet agents to prevent reinfarction in patients with ACS undergoing PCI.
Clopidogrel exerts potent pharmacological activities against platelets. Notably, there is a growing amount of evidence that recurrence of ischemic complications may be due to clopidogrel resistance. The 2016 Chinese guidelines recommend a 600 mg loading dose of clopidogrel before PCI. The impact of HPR to a 600 mg loading dose of clopidogrel before PCI in Chinese patients with ACS undergoing PCI is still unknown. For these reasons, we designed our observational clinical research.
In our observational clinical research, we included 134 patients with ACS undergoing PCI, and we measured the PRU value by VerifyNow analysis 6 hours after a 600 mg loading dose of clopidogrel before PCI. The results showed that among the 134 patients, 46 patients had HPR (34.3%) and 88 patients did not have HPR (65.7%). Moreover, at a mean follow-up of 6 months (SD 1 month), the rates of cardiac death, recurrence of unstable angina, and rehospitalization for TLR were higher in the HPR group. Multivariate analysis identified hemoglobin level and sex as independent predictors of the PRU value. On ROC curve analysis, a cutoff of 176 PRUs could significantly discriminate between patients with and patients without cardiac death, recurrence of unstable angina, and rehospitalization for TLR.
Variable platelet inhibition in patients receiving clopidogrel was first reported by Jaremo et al. [17] in 2002. Since the first report, HPR has been comprehensively reported. Matetzky et al. [18] reported an association between HPR and the risk of adverse cardiac events following PCI among 60 ACS patients who had taken a 300 mg loading dose of clopidogrel, followed by a daily dosage of 75 mg for 3 months. Geisler et al. [19] reported that the primary end point of myocardial infarction, stroke, and death was significantly increased in the HPR group, who were followed up for 3 months after PCI. HPR was found to affect 15–40% of the patients and has been considered to be associated with poor outcomes after PCI. Recent studies found that pre-PCI evaluation of platelet reactivity provides important prognostic information and may guide the therapeutic approach for those patients who do not fall within the described therapeutic window [15, 16]. However, the impact of HPR to a 600 mg loading dose of clopidogrel before PCI in Chinese patients with ACS is still unknown. Evidence-based data to guide treatment of HPR Asian patients with a 600 mg loading dose of clopidogrel before PCI are limited and tend to rely on studies from Europe, the USA, and Korea [20–22]. Firstly, our study indicated that the incidence of HPR in patients who received a 600 mg loading dose of clopidogrel before PCI was higher in Chinese patients than in European and US patients. Our findings expand observation of the platelet reactivity to about 6 hours after a 600 mg loading dose of clopidogrel before PCI in Asian patients. Secondly, our study directly tested platelet function by VerifyNow analysis about 6 hours after a 600 mg loading dose of clopidogrel before PCI. At a mean follow-up of 6 months (SD 1 month), the rates of cardiac death, recurrence of unstable angina, and rehospitalization for TLR were higher in the HPR group. Our findings confirmed the close correlation between HPR and adverse cardiac events in patients with a 600 mg loading dose of clopidogrel before PCI. Our data suggested that early intervention might improve the prognosis of HPR patients with a 600 mg loading dose of clopidogrel before PCI. Thirdly, we performed platelet function testing by VerifyNow analysis about 6 hours after a 600 mg loading dose of clopidogrel before PCI. The VerifyNow P2Y12 assay developed recently aims to overcome the limitations of conventional optical platelet aggregation [23, 24]. A PRU value greater than 208 was used to define patients with HPR [25]. It has been pointed out that HPR may be a marker of intrinsic platelet characteristics or functionality that promotes plaque formation and calcification [26, 27]. HPR might due to genetic and nongenetic factors, which is challenging for therapy today. Genetic variabilities that might interfere with platelet reactivity to clopidogrel include polymorphisms of the CYP2C19, CYP3A4/CYP3A5, CYP2C9, ABCB1, PON1, CES1 genes and the gene encoding the P2Y12 receptor (P2RY12) [28–34]. We can conclude from the current literature that, except for CYP2C19, most of the genetic polymorphisms investigated have a weak or insignificant effect on clopidogrel exposure and the inhibition of platelets. Clinical factors such as concomitant disease, drug-drug interaction, patient adherence, obesity, and age are well known to influence clopidogrel response [35–37]. However, in our multivariate analysis, we identified hemoglobin level and sex as independent predictors of the PRU value (y=456.355−1.736x 1−31.880x 2, P < 0.05). This finding may have a significant implication for exploring the factors contributing to HPR. Similarly to previous studies, we found that platelet reactivity expressed as a PRU value was an independent predictor of poor prognosis. On ROC curve analysis, the PRU cutoff value that best discriminated adverse cardiac events tended to be slightly lower than that in previous analysis (176 vs. 208). This is probably due to a different study population, the definition of adverse cardiac events, the different clopidogrel loading dose, the different time of VerifyNow testing, and the duration of follow-up.
It is very important to emphasize the counterbalancing effects of hemorrhagic and ischemic complications after PCI. By calculating platelet reactivity, we can accurately evaluate the risk of hemorrhagic and ischemic complications after stent implantation. Furthermore, we can tailor strategies for the use of clopidogrel and maximize clinical benefits. The findings from our study add evidence-based medicine to direct our clinical practice to ACS patients following PCI, especially for Asian patients. We highly suggest calculating platelet reactivity in patients with ACS undergoing PCI. We speculate that early and accurate assessment of platelet reactivity to clopidogrel may improve the prognosis of patients with ACS following PCI. The main direction of our follow-up research will focus on intervention in patients in Heilongjiang province with HPR to clopidogrel.
Limitations
There might be several limitations inherent to our study. First, CYP2C19 genetic testing might be proposed before clopidogrel therapy is started to identify patients likely to show reduced antiplatelet activity. In future studies, we will complete the list of genetic variants of CYP2C19. Second, the mechanisms leading to HPR will be further elucidated; for example, the relationship between hemoglobin level and PRU value. We will solve these problems in our future research. Our finding confirmed the close correlation between HPR and adverse cardiac events in patients receiving a 600 mg loading dose of clopidogrel before PCI. Whether early intervention might improve the prognosis of NPR patients receiving a 600 mg loading dose of clopidogrel before PCI still needs further research.
Conclusions
In our observational study, we found that HPR to a 600 mg loading dose of clopidogrel before PCI was associated with higher rates of cardiac death, recurrence of unstable angina, and rehospitalization for TLR. Furthermore, we performed multivariate analysis and found that HPR was associated with hemoglobin level and sex. Finally, we performed ROC curve analysis and found that the optimum PRU cutoff value for definite clinical adverse ischemic events was 176. Our data suggest that early intervention might improve the prognosis of HPR patients who receive a 600 mg loading dose of clopidogrel before PCI.


