Introduction
There have been more than 60 reported cases suggesting a possible causal relationship between ventricular preexcitation and dilated cardiomyopathy (DCM) in the absence of sustained tachyarrhythmia in the last 10 years [1–9]. Preexcitation of part of the myocardium may induce left ventricular (LV) dyssynchrony, which may mediate LV dysfunction and remodeling. However, until recently, ventricular preexcitation was considered as one of the causes of DCM, and the detailed mechanism is still not entirely clear.
We aimed to examine the clinical, electrocardiographic, echocardiographic, and electrophysiological characteristics and prognosis through ablation in and medication of 12 children presenting with ventricular preexcitation and DCM.
Study Participants
We identified 12 patients in this study. Eleven of them were inpatients and one was an outpatient in our clinic. All patients met the inclusion criteria and agreed to participate in the study, and written informed consent was given by their parents. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee, and it was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University. The inclusion criteria were as follows: older than 2 years for patients undergoing ablation; normal cardiac structure; intracardiac electrophysiological examination and radiofrequency catheter ablation (RFCA) and medication from March 2011 to December 2018. Patients with congenital heart disease, tachycardiomyopathy, atrial conduction to a ventricle via an accessory pathway while in atrial fibrillation, endless supraventricular tachycardia (SVT), intermittent ventricular preexcitation, hyperthyroidism, electrolyte disturbance, liver or kidney dysfunction, or acute infection were excluded. All the children stopped taking antiarrhythmic drugs at least five half-lives before ablation. Periodic 24-hour dynamic electrocardiograms (ECGs) were performed in every patient to confirm the presence of acute episodes of tachyarrhythmia and the absence of persistent SVT or tachycardia lasting more than 12 hours.
Electrocardiogram
Twelve-lead electrocardiography during sinus rhythm was conducted in all patients before and after the RFCA. The QRS duration was measured manually.
Echocardiography
Echocardiography was postponed if a patient had experienced an episode of tachycardia within the previous 72 hours. Transthoracic echocardiography was performed with a Vivid E9 ultrasound system with a 3.5 MHz probe (GE Medical Systems, USA). All patients underwent echocardiographic examination by the same physician before RFCA and 1, 3, 6, 9, 12, 24, and 36 months after ablation. Images and data were analyzed off-line by another physician, with use of Echopac (version 4.0.2; GE Vingmed Ultrasound). Each echocardiographic parameter was calculated as the mean value from three cardiac cycles. LV ejection fraction (LVEF) was calculated by the biplane Simpson method. LV end-diastolic diameter (LVEDD) was measured through the parasternal long-axis view. DCM was defined as an LVEF below 45% combined with an LVEDD Z score greater than 2 [10]. We calculate the LVEDD Z score according to the method adopted by Boston Children’s Hospital (USA) [11, 12]. The basal interventricular septal thickness was measured from the apical four-chamber view at the end of systole. Intraventricular dyssynchrony was quantified as the time delay between the peak septal systolic motion and the left posterior ventricular wall (LVPW) systolic motion (septal to posterior wall motion delay; SPWMD) [13]. Speckle tracking echocardiography was applied in patients 2–12. The two-dimensional LV strain was investigated [14, 15]. The apical four-, two-, and three-chamber long-axis views were collected. The longitudinal strains were analyzed with Echopac version 4.0.2 (GE Vingmed Ultrasound) to demonstrate whether the left ventricle was contracting synchronously. The standard deviation of the time to the peak systolic strain (Ts-SD) over 18 longitudinal segments was measured.
Electrophysiology Studies and Radiofrequency Ablation
All procedures were undertaken with the patients under anesthesia with propofol and analgesia administered intravenously. Standard electrophysiological methods were applied. In eight patients ablation was done with use of a two-dimensional mapping system. A 7Fr deflectable electrode catheter (Biosense Webster, USA) with a 4mm distal electrode was used for RFCA with a radiofrequency current power setting of 30W and a temperature setting of 60°C. In four patients ablation was done with a 7.5Fr saline-irrigated catheter with use of a Carto 3 mapping system (Biosense Webster, USA).
The ablation end points were the loss of antegrade and retrograde conduction of the overt accessory pathways. All the ablations were accomplished with Swartz sheaths to stabilize the catheters. After ablation, the conducting system was reevaluated to determine whether SVT was still inducible, and whether there was residual anterograde or retrograde conduction. The long-term indicators of successful ablation were the absence of recurrence of tachycardia and lack of evidence of a ventricular preexcitation wave on the ECG.
Medication
All patients except patient 12 were treated with routine anti-heart failure chemotherapy, including digoxin, furosemide, aldactone, and captopril, once they had received a diagnosis of DCM. Digoxin was used only when periodic Holter monitoring was performed in every patient and showed no atrial tachyarrhythmia episodes. Three patients received high-dose intravenous immunoglobulin several times, and seven patients received predinsone orally. Patient 3 received three courses of cyclophosphamide pulse therapy. Standard medical treatment of heart failure, such as with digoxin, furosemide, aldactone, and captopril, was continued in all patients after ablation until they achieved recovery of their LV function. Captopril use was stopped when the patient’s LVEDD became normal.
Statistical Analysis
Data were analyzed with SPSS Statistics for Windows (version 17.0; SPSS Inc., Chicago, IL, USA). Continuous variables are expressed as the mean±standard deviation. Differences between the two groups were compared by Student’s ttest. P<0.05 was considered statistically significant.
Results
Patient Characteristics
The clinical characteristics of the 12 patients with DCM and ventricular preexcitation are shown in Table 1. There were nine females and three males. Their mean ages at disease onset and ablation were 3.75±2.85 years and 7.1±3.07 years, respectively. At admission, overt ventricular preexcitation was noted on the 12-lead ECG in all patients. The vectors of the delta wave all suggested right-sided pathways. Two patients (patients 2 and 11) had moderate-to-severe mitral regurgitation and prolapse in addition to DCM and ventricular preexcitation. The longest duration of disease was 5 years. DCM was diagnosed in three patients before they were 1 year old.
Clinical, Electrophysiological, and Echocardiographic Characteristics of the Patients.
| Patient | Sex | Age (years) at | Weight (kg) | Height (cm) | Follow-up after RFCA (months) | QRS duration before/after RFCA (ms) | LVEF (%) | LVEDD Z score before/after RFCA* | Basal IVS thickness before/after RFCA (3 months) (mm) | SPWMD before/after RFCA (1 month) (ms) | Ts-TD before/after RFCA (l month) (ms) | Location of AP** | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diagnosis of DCM | RFCA | Before RFCA | After RFCA | |||||||||||||||
| 24 hours | 1 month | 3 months | 6 months | 9 months | ||||||||||||||
| 1 | F | 6 | 8 | 25 | 131 | 63 | 160/100 | 41 | – | 59 | 63 | 63 | 64 | 3.36/0.42 | 3/6 | 100/54 | – | Anteroseptum |
| 2 | F | 6 | 11 | 29 | 138 | 51 | 220/128 | 41.6 | 42 | 44 | 45.2 | 51 | 60 | 3.75/1.21 | 2.8/4.8 | 280/80 | 243/23 | Anteroseptum |
| 3 | F | 2 | 7 | 20 | 125 | 47 | 170/82 | 42 | 50.9 | 51 | 52 | 62 | 62 | 2.9/1.54 | 4/5.5 | 100/60 | 269/19 | Free wall 8:00 |
| 4 | F | 8 | 11 | 30.5 | 147 | 35† | 180/92 | 42 | 49 | 51.8 | 66 | 67 | 70 | 6.37/2.67 | 5/7 | 140/47 | 63/10 | Free wall 9:00 |
| 5 | F | 0.75 | 4.16 | 17.5 | 115 | 43 | 142/96 | 44 | 46 | 54 | 60 | 60 | 60 | 3.6/1.21 | 3.8/4.4 | 95/50 | 138/23 | Anteroseptum |
| 6 | M | 0.58 | 5 | 32 | 117 | 41 | 136/86 | 45 | 50 | 58 | 60 | 60 | 63 | 3.87/2.13 | 2.7/4.9 | 124/72 | 153/29 | Free wall 8:00 |
| 7 | F | 1 | 11 | 29 | 136 | 37 | 132/83 | 38 | 46 | 52 | 55 | 58 | 61 | 3.47/1.45 | 3.2/5.1 | 140/53 | 160/38 | Free wall 9:00 |
| 8 | F | 3 | 6 | 25 | 120 | 32 | 130/97 | 43 | 51 | 54 | 57 | 62 | 65 | 4.01/1.89 | 2.9/5.5 | 154/64 | 177/24 | Anteroseptum |
| 9 | F | 7 | 9 | 26 | 128 | 38 | 138/91 | 52 | 55 | 60 | 62 | 62 | 65 | 3.1/1.53 | 3.8/6 | 176/87 | 183/29 | Free wall 9:00 |
| 10 | M | 0.66 | 2 | 23 | 101 | 27 | 136/88 | 48 | 53 | 58 | 64 | 65 | 66 | 1.9/1.4 | 3.2/4.9 | 160/59 | 138/32 | Posteroseptum |
| 11 | M | 3 | 3.5 | 17 | 100 | 32 | 130/94 | 39 | 40 | 42 | 48 | 52 | 55 | 9.66/4.64 | 3.1/5.1 | 164/73 | 180/21 | Free wall 9:00 |
| 12 | M | 7.5 | 7.5 | 32.5 | 134 | 16 | 150/98 | 36 | 39 | 48 | 50 | 54 | 54 | 5.4/2.3 | 4.2/6.2 | 160/62 | 178/19 | Free wall 10:00 |
| Mean±SD | – | 3.75±2.85 | 7.1± 3.07 | 27±4.7 | 134.5± 9.5 | 12±7.8 | 152±26.9/ 94.6±12 | 42.63± 4.34 | 49.95± 4.6 | 52.65± 5.79 | 56.85± 6.79 | 59.7±5 | 62± 4.52 | 4.28±2.05/ 1.86±1.05 | 3.47±0.7 45±0.73 | 140.4±149.4/ 63.4±12.3 | 186.5±43.4/ 31.1±18.8 | – |
AP, accessory pathway; DCM, dilated cardiomyopathy, F, female; IVS, interventricular septum; LVEDD, diastolic left ventricular internal dimension; LVEF, left ventricular ejection fraction; M, male; SD, standard deviation; SPWMD, septal to posterior wall motion delay; RFCA, radiofrequency catheter ablation; Ts-SD, standard deviation of the time to peak systolic strain.
*The last follow-up after the ablation.
**Tricuspid annulus is likened to a clock dial at left anterior oblique 45°. Accessory pathways located at 6:00~12:00 are defined as free wall accessory pathways.
†The time for the follow-up of patient 4 was calculated from the first ablation.
Clinical Manifestations
Patient 12 had no symptoms or impaired exercise tolerance before disease onset. He suffered from a sudden cardiac origin of syncope presenting with no heartbeat. Ventricular tachycardia and fibrillation appeared when immediate chest compression was performed. Iron channel disease–related genetic testing showed negative results, and the possibility of inherited arrhythmogenic diseases was slight. The patient undewent successful RFCA when he was in a relatively stable condition. The other 11 patients presented with the clinical signs and symptoms of chronic congestive heart failure, such as fatigue, exertional dyspnea, and impaired exercise tolerance, and were at the low end of physical growth compared with children in the same age group. Infant patients presented with hidrosis and not enough physical strength to suck. Seven patients never experienced paroxysmal SVT. Five patients had paroxysmal SVT, and three of them had paroxysmal SVT just once. Patients experienced palpitation, fatigue, nausea, and vomiting when paroxysmal SVT happened. The clinical manifestation in young children was crying that was hard to comfort. ECG monitoring was performed if the patient could not express symptoms clearly. None of the patients experienced frequent tachycardias before presentation with DCM. Tachycardiomyopathy and other causes were excluded by the main complaint, frequency of SVT, and periodic 24-hour dynamic ECGs.
Echocardiographic Manifestations
Abnormal interventricular septal motion and dyssynchronous ventricular contraction were observed in all patients and were quite impressive (as shown by arrows in Figures 1, 2, and 3A). The LV long-axis four-chamber view showed the basal segment of the interventricular septum (IVS) became thin and moved like an aneurysm (Figure 1), and M-mode tracing from the parasternal long-axis view showed the ventricular septum and LV posterior wall (LVPW) moved in the same direction (Figure 2) in 11 patients (not in patient 11). Echocardiography indicated misplacement of the IVS and LVPW in patient 11 (Figure 4). A significant decrease in the thickness of the IVS was observed (Table 1). SPWMD and Ts-SD over 18 longitudinal segments were 140.4±49.4 ms and 186.5±43.4 ms, respectively, before ablation, and decreased significantly to 63.4±12.3 ms and 31.1±18.8 ms, respectively, after ablation (P<0.05). Longitudinal strain analysis showed paradoxical motion of the basal and middle segments of the interventricular septum. Their strains were positive and opposite those of the other segments (Figure 5A). The ventricular motion became synchronous, and the segmental dyskinesia disappeared after ablation in all patients, as shown in Figures 3B and 5B.
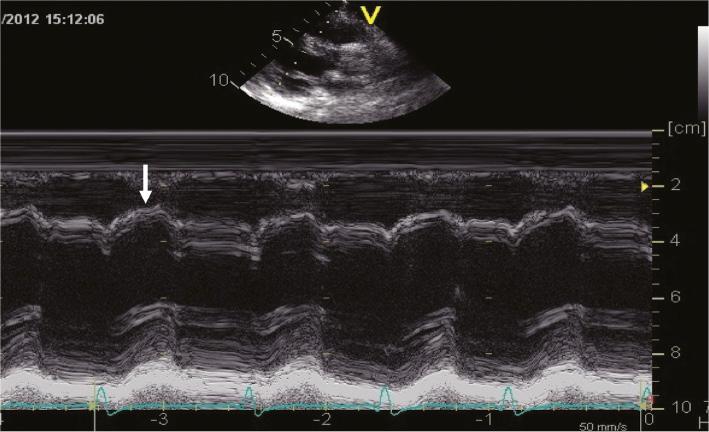
The Interventricular Septum and Posterior Wall of the Left Ventricle Moved in the Same Direction.
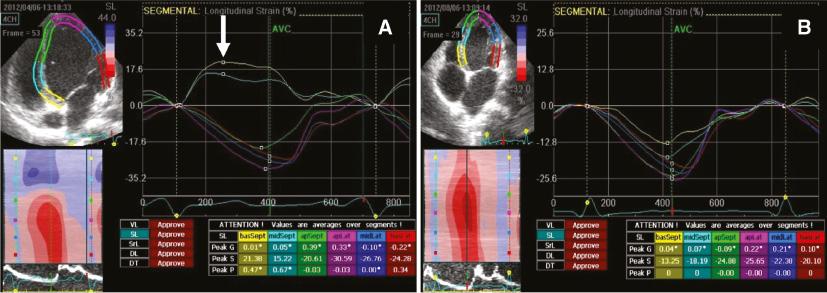
(A) Left Ventricular Longitudinal Strain Curves and M-Mode Tracing From the Apical Four-Chamber View of Patient 3 Before the Ablation Showing the Paradoxical Motion of the Basal and Middle Segments of the Interventricular septum (arrow, positive strain). (B) The Left Ventricular Longitudinal Strain Changed to Normal 4 Months After the Ablation.
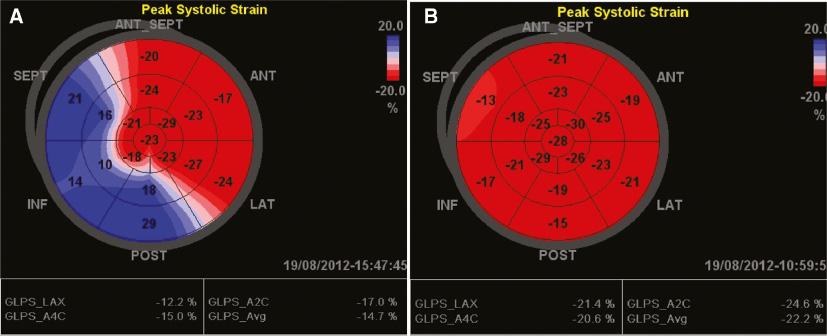
(A) The Bull’s Eye of the Left Ventricular Longitudinal Strain of Patient 3 Before the Ablation Showing the Paradoxical Motion of the Basal and Middle Segments of the Interventricular Septum and the Inferior and Posterior walls (positive strain). (B) The Bull’s Eye of the Left Ventricular Longitudinal Strain Changed to Normal 4 Months After the Ablation.
Electrophysiology Studies and Radiofrequency Ablation
SVT was induced in seven patients. All pathways were successfully ablated. No multiple accessory pathways could be identified. The successful ablation sites were the right-sided anteroseptum in four patients, right-sided posteroseptum in one patient, and right-sided free wall in seven patients. Retrograde conduction of the accessory pathway of patient3 recurred 4 months after the ablation. She experienced six episodes of paroxysmal SVT afterward, sustained for approximately 1 hour each time. Nonetheless, her LV function continued to improve during this period. In addition, she accepted a second ablation (which was successful) 8 months after the first ablation. There were no procedure-related serious complications. ECGs were routinely obtained after the catheter ablation, and they confirmed the loss of the preexcitation pattern in these patients.
Clinical and Echocardiographic Improvement after Catheter Ablation/Medication
Patients 1–11 had improved physical activity, growth, and development after the ablation. No life-threatening malignant arrhythmia recurred in patient 12. The abnormal movement between the interventricular septal wall and the LVPW changed to normal movement. The phenomenon of the basal segment of the IVS moving like an aneurysm disappeared. SPWMD and Ts-SD decreased significantly. All of the patients achieved normal LV systolic function gradually after ablation. Six patients had an increase of LVEF of more than 5% just 24 hours after ablation. Only four patients did not have an LVEF of more than 60% 6 months after ablation. Patients 11 and 12 took 9 and 15 months, respectively, to achieve an increased LVEF of 55% (Table 1). All patients with DCM had decreased LVEDD during follow-up. Eight patients had a normal LVEDD Z score. Mitral regurgitation was alleviated in patients 2 and 11.
LVEF in the 12 patients with DCM was (32.14±7.71)% at disease onset, (42.63±4.34)% before ablation, and (63.82±2.6)% 1 year after ablation. LVEF increased significantly after medication (t=−4.1, P<0.05) and increased to a higher level 1 year after ablation (t=−14, P<0.05). LVEDD Z scores were 2.88±1.2, 4.28±2.04, and 1.86±1.05 at disease onset, before ablation, and 1 year after ablation, respectively. LVEDD increased after medication (t=−2.05, P=0.053) and decreased 1 year after ablation (t=3.57, P=0.002). There were only two patients with LVEDD Z scores higher than 2.
Discussion
In this study, we described better outcomes of ablation in patients with ventricular preexcitation–induced DCM. Ventricular preexcitation–induced DCM diagnosed in patients 1–11 was originally misdiagnosed as idiopathic DCM combined with ventricular preexcitation or Wolff-Parkinson-White syndrome. Routine anti-heart failure chemotherapy did not have satisfactory effects on them. As we knew more about the diagnosis of and therapy for ventricular preexcitation–induced DCM, we performed detailed echocardiographic examinations of the patients and found that the IVS and LVPW moved in the same direction and the basal segment of the IVS moved like an aneurysm. Therefore, we made the diagnosis of ventricular preexcitation–induced DCM. These patients had complete recovery of LV systolic function through successful ablation. Their excellent prognosis confirmed our initial diagnosis. We summarize its characteristics as follows: the patients fulfill the diagnostic criteria for DCM; incessant tachycardia and other causes of DCM can be excluded; the ECG indicates an overt right-sided accessory pathway; LV dyssynchrony can be detected by M-mode echocardiography or tissue Doppler imaging or two-dimensional strain analysis; the prognosis of ventricular preexcitation–induced DCM through ablation is excellent.
With regard to the diagnosis, some clinicians hold different views. They diagnosed such cases as idiopathic DCM combined with ventricular preexcitation. They hold that ventricular preexcitation is just an adverse factor and not the cause of DCM. We considered that LV systolic function might completely recover through 1 year of regular anti-heart failure chemotherapy in mild cases with idiopathic DCM but with less possibility in moderate or severe cases. The patients had rapid recovery of LVEF after the loss of the antegrade conduction of the overt accessory pathways, while the dosages of drugs were not altered. The rapid recovery suggested a causal relationship between the overt right-sided accessory pathway and the development of DCM. Besides the recovery from disease, echocardiography could also provide information and contributed to differential diagnosis. Patients with idiopathic DCM may present with dyssynchronous ventricular wall contraction, but it is rarely seen that the basal segment becomes thin and moves like an aneurysm, which is typical for ventricular preexcitation–induced DCM [16, 9].
Retrograde conduction of the accessory pathway of patient4 recurred 4 months after the ablation. However, her LV systolic function continued to improve and LV dyssynchrony disappeared during this period despite the frequent tachycardia. Therefore, this evidence indicates that abnormal ventricular activation alone, due to anterograde conduction in patients with overt right-sided accessory pathways, would have adverse effects on cardiac function. Since RFCA can eliminate the pathogeny, the prognosis of ventricular preexcitation–induced DCM is much better than that of idiopathic DCM [16, 17, 3]. RFCA may be the first therapeutic choice for ventricular preexcitation–induced DCM [1–9]. If there is an anteroseptal accessory pathway, serious atrioventricular block should be avoided [18]. For infant cases, drugs that can inhibit the anterograde conduction of the accessory pathway such as amiodarone can be administered [19].
All patients with ventricular preexcitation–induced DCM could benefit from regular anti-heart failure chemotherapies, increasing LVEF to a certain extent. However, LVEF was not satisfactory until successful ablation had been performed and LVEDD seemed to increase with chemotherapy alone. Anti-heart failure drugs were still needed for recovery after ablation.
At present, the cause of ventricular preexcitation–induced DCM, LV dysfunction, or LV dilation is thought to be abnormal interventricular septal motion and LV dyssynchrony induced by electromechanical dyssynchrony [20]. The location of the accessory pathway is predictive of this association [5]. Of 63 reported patients with accessory pathway–induced DCM, 50 had right-sided septal accessory pathways and 13 had right-sided free wall accessory pathways [19, 1, 20–22, 2–9]. In our patients, five had septal accessory pathways and seven had free wall accessory pathways. We consider that prominent segmental dyskinesia and dyssynchronous LV contraction can also be induced by right-sided free wall accessory pathways similarly to septal accessory pathways. We suspect the extent of LV dyssynchrony and abnormal ventricular septal motion may be critical determinants in the onset of LV dysfunction and LV remodeling. Septal dyskinesia was reported by Kwon et al. [20] as the only significant risk factor for reduced LVEF, as identified in a multivariate analysis that included septal dyskinesia, age at diagnosis, accessory pathway location, and QRS duration. Septal dyskinesia meant basal septal aneurysmal bulging with paradoxical septal motion and septal wall thinning. Its mechanism is similar to that of LV dysfunction and remodeling induced by LV dyssynchrony in patients with right ventricular pacing or complete left bundle branch block [23, 24].
The pathogenic mechanisms responsible for the development of LV dysfunction and LV remodeling in patients with overt right-sided accessory pathways have not yet been fully elucidated. As timing is crucial for synchronous ventricular contraction and regional myocardial function, a delay in the electrical activation of a myocardial segment may contribute to cardiac pump inefficiency [23]. The preexcited myocardial segment gradually becomes thinner, which is thought to be associated with the decreased workload and hypotrophy [16, 25]. A dyssynchronous septal myocardial segment may function much like an aneurysm, as seen in most of the patients. Why did most of the patients with overt right-sided accessory pathways have no abnormal ventricular wall motion, LV systolic dysfunction, or LV dilation? Further study is needed to understand the detailed mechanism.
The incidence of ventricular preexcitation–induced DCM, LV dysfunction, and LV dilation has probably been underestimated. Many patients thought to have ventricular preexcitation–induced DCM likely originally received a misdiagnosis of idiopathic DCM [25, 3]. Accessory pathway–induced DCM should be suspected in all patients presenting with DCM and overt right-sided accessory pathways with a good prognosis. This diagnosis needs to be considered when other causes of DCM are excluded, particularly tachyarrhythmia-mediated DCM. Electromechanical resynchronization can be the first therapeutic choice for accessory pathway–induced DCM. Newer echocardiographic techniques, such as tissue Doppler imaging and speckle tracking echocardiography, may demonstrate more details of ventricular dyssynchrony and function [26, 8].
Conclusions
We consider that ventricular preexcitation via a right-sided accessory pathway may induce dyssynchronous ventricular contraction leading to LV dysfunction or DCM through LV remodeling. Ventricular preexcitation–induced DCM is an indication for ablation with excellent prognosis. Electromechanical resynchronization can reverse LV dysfunction and remodeling. Ventricular preexcitation–induced DCM should be suspected in all patients presenting with heart failure and overt right-sided acccessory pathways in whom no cause of their DCM can be found. LV dyssynchrony detected by echocardiographic examination supports the diagnosis. The prognosis after ablation can confirm the final diagnosis.



