Significance Statement: Coronary artery disease continues to be a global health concern. Invasive coronary angiography, as the gold standard, is widely used to detect coronary stenosis but has limitations and risks. Magnetic resonance coronary angiography (MRCA), a noninvasive method, is expected to be an alternative method for diagnosis and assessment of coronary artery disease. The image quality and the depiction of artery length have been substantially improved and the acquisition speed has been substantially increased by use of three-dimensional whole-heart MRCA. Hence, we conducted a meta-analysis to evaluate the diagnostic accuracy of three-dimensional whole-heart MRCA in detecting stenosis of coronary arteries with invasive coronary angiography as the reference standard.
Introduction
Invasive coronary angiography (ICA), as the gold standard, is widely used to detect coronary stenosis with high spatial resolution [1]; however, it has several limitations. Firstly, it is an invasive procedure with radiation exposure. Secondly, the use of iodinated contrast agent may lead to various complications. Thirdly, about half of patients with suspected coronary artery disease (CAD) who underwent elective ICA were found to have no significant stenosis [2–4].
Different scan protocols for magnetic resonance coronary angiography (MRCA), a noninvasive method, have evolved during the past few decades [3, 5–7]. From the two-dimensional breath-hold technique to the three-dimensional (3D) respiratory-gated technique, MRCA was initially performed with a target-volume method. It was time-consuming and operator dependent. Subsequently, the whole-heart approach was developed, which makes distal coronary segments more delineative in a reduced total examination time in comparison with the target-volume approach [7]. Herein, we conduct a meta-analysis to evaluate the diagnostic accuracy of 3D whole-heart MRCA in detecting stenosis of coronary arteries.
Materials and Methods
This meta-analysis generally followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [8].
Data Sources and Searches
We searched the PubMed and Embase databases for all published studies in English evaluating the accuracy of 3D whole-heart MRCA with ICA as the reference standard using different combinations of the following thesaurus terms and synonyms as text words: “magnetic resonance angiography,” “whole-heart,” and “coronary artery disease.” In addition, references of retrieved meta-analyses and systematic reviews were screened. All studies were carefully examined to exclude potential duplicates or overlapping data. Two reviewers selected the studies independently. Differences were discussed to reach an agreement.
Study Eligibility
The inclusion criteria for the studies were as follows: (1) 3D whole-heart MRCA was used as a diagnostic test to determine significant stenosis in patients who were suspected of having CAD; (2) ICA served as the standard reference, and a 50% or greater reduction in diameter was considered significant stenosis; (3) raw data provided or data that enabled the building of a 2×2 contingency table based on sensitivity and specificity. Letters, case reports, editorials, reviews, animal studies, and retrospective studies were excluded. As the PRISMA flow diagram in Figure 1 shows, we first scanned the titles and abstracts, and then reviewed the full text to reassess the remaining potentially eligible articles in depth.
Data Extraction and Quality Assessment
Two investigators independently extracted the data using a standardized data extraction form. Discrepancies were solved by interrater consensus.
The following data were extracted from each included study: first author, year of publication, study population characteristics (sample size; sex; age; heart rate); technical characteristics (scanner manufacturer; sequence; magnetic field strength; coil channels; scan time), and test accuracy results (true-positive/true-negative/false-positive/false-negative values). The quality of included studies was assessed by the tailored Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool to make the checklist items more specific and practical [9].
Data Synthesis and Statistical Analysis
The main analysis was performed at the patient level, as we were concerned about whether patients need further management. To look for possible publication bias, we applied Deeks’s test for funnel plot asymmetry [10], which performs linear regression of log diagnostic odds ratios (DORs) on the inverse root of effective sample sizes. A nonzero slope coefficient is suggestive of significant small study bias (P<0.10). The interrater reliability for quality assessment was assessed by the Cohen kappa test.
Primary data synthesis was performed within the bivariate mixed-effects binary regression modeling framework [11, 12]. On the basis of a 2×2 contingency table for each study, sensitivity, specificity, and positive and negative likelihood ratios (LRs) were computed with 95% confidence intervals (CIs). The results for sensitivity and specificity were presented in a forest plot with both individual study and pooled estimates. Positive and negative LRs were used to evaluate the clinical or patient-relevant utility of whole-heart MRCA by our calculating the posttest probability based on Bayes’s theorem. The derived logit estimates of sensitivity, specificity, and respective variances were used to construct a summary receiver operating characteristic curve [13], presenting the point estimates for each study, the joint receiver operating characteristic curve, and the pooled characteristics, including the 95% CI and the 95% prediction region. The area under the curve (AUC), obtained by trapezoidal integration, serves as a global measure of test performance: low (0.5≤AUC<0.7), moderate (0.7≤AUC<0.9), high (0.9≤AUC≤1) accuracy [14].
The heterogeneity across studies was assessed graphically by forest plots and statistically by I 2, which describes the percentage of the total variation across studies that is attributable to heterogeneity rather than chance [15]: values greater than 50% are considered to correspond to substantial heterogeneity. Meta-regression was applied to evaluate predefined possible sources of heterogeneity, which include age, prevalence of CAD, magnetic field strength, and enhancement. Covariates were manipulated as mean-centered continuous effects or as dichotomous fixed effects. The effect of each covariate on sensitivity was estimated separately from that on specificity.
Furthermore, the influence of each study on the summary estimates was investigated by our sequentially omitting each study to reestimate the pooled sensitivity and specificity.
All the analyses were conducted with STATA 14 (Stata Corporation, College Station, Texas, USA) and Revman 5.3.
Results
Characteristics of Selected Studies
A total of 18 studies, which contained 595 patients who tested positive and 540 patients who tested negative, met our inclusion criteria (Figure 1). Two were multicenter studies [16, 17]. The study from Wagner et al. [18] presents both the performance of MRA with or without contrast agent injection. We only included results with contrast agent injection. It is noteworthy that four studies [19–22] evaluated the incremental value of MRCA as part of a cardiac magnetic resonance (CMR) protocol including myocardial perfusion imaging (MPI) and late gadolinium enhancement (LGE). Table 1 describes the characteristics of the included studies. All the studies with a field strength of 1.5 T used a steady-state free precession sequence, while the studies with a field strength of 3.0 T used a spoiled gradient-echo sequence. A vasodilator was used in 12 studies [16, 18–20, 23–30]. The pooled scan time was 11.10±3.29 min. Finally, there were 16 studies at the artery level available for our synthesizing data [16, 17, 19–26, 28–33], which included 804 positive arteries and 2142 negative arteries.
Characteristics of Included Studies.
| Study, year | No. of enrolled patients | Study population characteristics | Technical characteristics | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male (%) | Age (years) | Heart rate | CAD prevalence (%) | Scanner manufacturer | Sequence | Magnetic field strength (T) | Coil channels | Vasodilatory premedication | Contrast agent | Scan time (min) | ||
| Bettencourt et al. [19], 2013 | 43 | 65 | 61±8 | 65±6 | 56 | Siemens | SSFP | 1.5 | 12 | Yes | Yes | 17.9±4.6 |
| Chen et al. [23], 2010 | 67 | 67 | 60±10 | 65±9 | 55 | Siemens | Spoiled gradient-echo sequence | 3 | 12 | Yes | Yes | 9.6±3.2 |
| Hamdan et al. [16], 2011 | 110 | 70 | 65±8 | 63±8 | 56 | Philips | Spoiled gradient-echo sequence | 3 | 32 | Yes | No | 17.0±4.7 |
| He et al. [32], 2016 | 39 | 77 | 57±10 | 70±7 | 59 | Siemens | Spoiled gradient-echo sequence | 3 | 32 | No | Yes | 7.8±0.8 |
| Heer et al. [20], 2013 | 59 | 61 | 59±13 | 62±8 | 51 | GE | SSFP | 1.5 | 8 | Yes | No | 14.3±6.2 |
| Kato et al. [17], 2010 | 127 | 44 | 67± 9 | 68±12 | 44 | Philips | SSFP | 1.5 | 5 | No | No | 9.5±3.5 |
| Klein et al. [21], 2008 | 46 | 48 | 60±10 | 73±15 | 48 | Philips | SSFP | 1.5 | 5 | No | No | 6.3±1.6 |
| Kunimasa et al. [24], 2009 | 43 | 77 | 65±13 | 66±12 | 77 | Philips | SSFP | 1.5 | 5 | Yes | No | 9.0±3.1 |
| Nagata et al. [25], 2011 | 67 | 58 | 69±13 | 72±10 | 58 | Philips | SSFP | 1.5 | 32 | Yes | No | 6.2±2.8 |
| Piccini et al. [33], 2014 | 31 | 68 | 49±21 | NR | 68 | Siemens | SSFP | 1.5 | 30 | NR | Yes | 7.8±1.9 |
| Pouleur et al. [31], 2008 | 77 | 73 | 61±14 | 69±15 | 22 | Philips | SSFP | 1.5 | 5 | No | No | 20.0±4.0 |
| Sakuma et al. [27], 2005 | 20 | 80 | 65±12 | 70±12 | 60 | Philips | SSFP | 1.5 | 5 | Yes | No | 13.8±3.8 |
| Sakuma et al. [26], 2006 | 113 | 87 | 66±11 | 72±13 | 45 | Philips | SSFP | 1.5 | 5 | Yes | No | 12.9±4.3 |
| Wagner et al. [18], 2011 | 27 | 13 | 55±7 | NR | 67 | Siemens | SSFP | 1.5 | 32 | Yes | Yes | 9.1±2.0 |
| Yang et al. [28], 2009 | 62 | 48 | 61±11 | 67±7 | 55 | Siemens | Spoiled gradient-echo sequence | 3 | 12 | Yes | Yes | 9.0±1.9 |
| Yang et al. [29], 2012 | 101 | 48 | 58±11 | 66±8 | 49 | Siemens | Spoiled gradient-echo sequence | 3 | 32 | Yes | Yes | 7.0±1.8 |
| Yonezawa et al. [30], 2014 | 62 | 74 | 69±13 | 73±10 | 53 | Philips | SSFP | 1.5 | 32 | Yes | No | 6.8±2.6 |
| Zhang et al. [22], 2018 | 46 | 72 | 54±12 | 67±10 | 74 | Siemens | Spoiled gradient-echo sequence | 3 | 32 | NR | Yes | 10.4±3.2 |
Values are given as a number or the mean±standard deviation.
CAD, Coronary artery disease; NR, not reported; SSFP, steady-state free precession.
Data Synthesis and Statistical Analysis
At the patient level, the pooled sensitivity, specificity, positive LR, negative LR and DOR were 0.90 (95% CI 0.87–0.93), 0.79 (95% CI 0.73–0.84), 4.3 (95% CI 3.3–5.7), 0.12 (95% CI 0.09–0.17), and 35 (95% CI 21–59), respectively. Significant heterogeneity (Q=40.88, P<0.001, I 2=58.42%) was found in specificity between studies, while moderate heterogeneity (Q=27.12, P=0.06, I 2=37.32%) was detected in sensitivity (Figure 2). The meta-regression analysis showed that the magnetic field strength (P1<0.001, P2<0.001) and enhancement (P1<0.001, P2=0.02) were significant predictors (Figure 3). The prevalence of CAD (P1=0.98, P2=0.66) and the age of the patients (P1=0.99, P2=0.82) showed no significant influence.
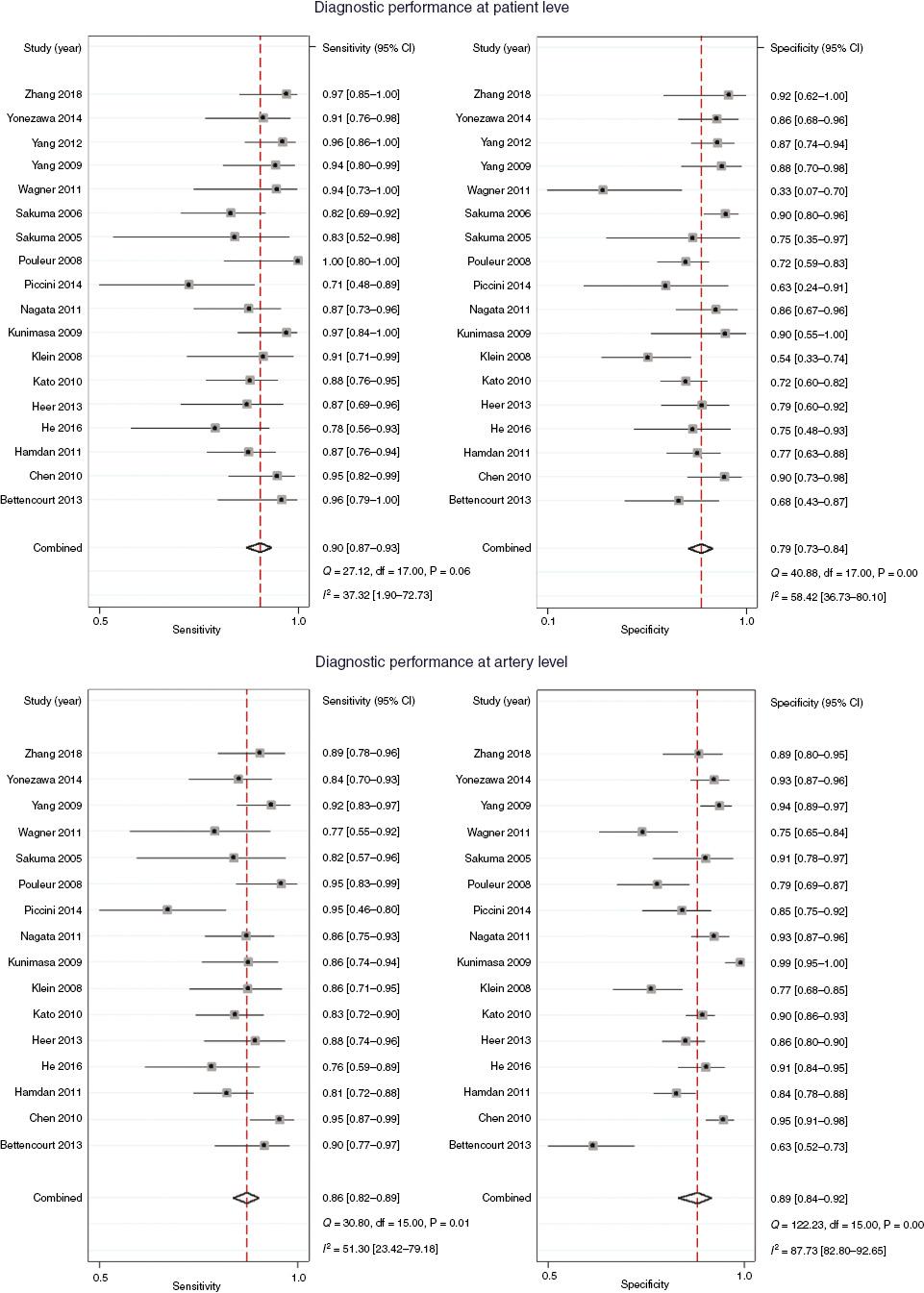
Forest Plots of Sensitivity and Specificity.
CI, Confidence interval; df, degrees of freedom.

Graphic Presentation of Meta-Regression.
CAD, Coronary artery disease; CI, confidence interval; e, enhancement (“yes” means a contrast agent was used; “no” means a contrast agent was not used); t, magnetic field strength.
The probability of CAD after whole-heart MRCA is presented in Figure 4. We assumed that the pretest probabilities of 25%, 50%, and 75% represented low clinical suspicion, the worst-case scenario, and high clinical suspicion, respectively (Figure S1 in the supplementary material). The posttest probability of patients with low suspicion (pretest probability of 25%) was 4% with a negative result. With a pretest probability of 50%, the patients’ posttest probabilities of CAD with positive and negative MRCA results were 81% and 11%, respectively. The posttest probability of patients with high suspicion (pretest probability of 75%) was 93% with a positive result.

Probabilities of Coronary Artery Disease in Different-Hierarchy Patients after Whole-Heart Magnetic Resonance Coronary Angiography.
Whole-heart magnetic resonance coronary angiography performed better in ruling out disease. LR−, Negative likelihood ratio; LR+, positive likelihood ratio; NPV, negative predictive value; PPV, positive predictive value.
The data for different protocols and their combination were available from four studies on a patient basis. If any CMR component was positive, the overall CMR result was considered positive. The pooled estimates are summarized in Table 2. Integrated analysis from MRCA and CMR MPI/LGE increased the overall CMR performance for detection of significant CAD with pooled sensitivity, specificity, positive LR, negative LR, and DOR of 0.96 (95% CI 0.90–0.99), 0.66 (95% CI 0.50–0.79), 2.8 (95% CI 1.8–4.3), 0.06 (95% CI 0.02–0.15), and 50 (95% CI 15–161), respectively.
Diagnostic Performance of Different Protocols and their Combination on a Per-Patient Basis.
| Sensitivity | Specificity | PLR | NLR | DOR | AUC | |
|---|---|---|---|---|---|---|
| MRCA | 0.90 (0.72–0.97) | 0.80 (0.69–0.88) | 4.5 (2.8–7.2) | 0.13 (0.04–0.38) | 35 (10–122) | 0.85 (0.82–0.88) |
| MPI/LGE | 0.83 (0.75–0.89) | 0.82 (0.66–0.91) | 4.5 (2.3–8.7) | 0.21 (0.13–0.32) | 22 (9–54) | 0.84 (0.81–0.87) |
| MRCA plus MPI/LGE | 0.96 (0.90–0.99) | 0.66 (0.50–0.79) | 2.8 (1.8–4.3) | 0.06 (0.02–0.15) | 50 (15–161) | 0.96 (0.94–0.98) |
AUC, Area under the curve; DOR, diagnostic odds ratio; LGE, late gadolinium enhancement; MRCA, magnetic resonance coronary angiography; MPI, myocardial perfusion imaging; NLR, negative likelihood ratio; PLR, positive likelihood ratio.
At the artery level, the pooled sensitivity, specificity, positive LR, negative LR, and DOR were 0.86 (95% CI 0.82–0.89), 0.89 (95% CI 0.84–0.92), 7.5 (95% CI 5.3–10.7), 0.16 (95% CI 0.12–0.21) and 47 (95% CI 28–79), respectively. Heterogeneity was obvious in both sensitivity (Q=30.80, P=0.01, I 2=51.30%) and specificity (Q=122.23, P<0.001, I 2=87.73%) between studies.
The summary receiver operating characteristic curves are illustrated in Figure S2 in the supplementary material, showing the general performance of whole-heart MRCA. The AUC was high at both the patient level and the artery level, with a value of 0.93 (95% CI 0.90–0.95) and 0.92 (95% CI 0.90–0.94), respectively.
No outlier was identified in sensitivity analysis at both the patient level and the artery level as all the new summary estimates were within the 95% CIs of the original estimates. Each individual study did not influence the pooled sensitivity and specificity by more than 0.02 (Figure S3 in the supplementary material).
Publication Bias
Deeks’s test showed no publication bias on any level of analysis (P=0.74 for the per-patient level, P=0.35 for the per-artery level).
Quality Assessment
There was a low risk of bias and a low level of applicability concerns. The details for each module are summarized in Figure S4 in the supplementary material. QUADAS-2 items are given in the supplementary material. The interrater reliability for quality assessment was perfect (κ=0.896).
Discussion
Current European and American guidelines support the use of coronary computed tomography angiography (CCTA) for ruling out CAD [34, 35]. MRCA, another noninvasive method, with improved image quality, increased acquisition speed, and improved depiction of artery length by the whole-heart approach, is expected to be an alternative method for diagnosis and assessment of CAD. In the present meta-analysis, 3D whole-heart MRCA showed good performance, with pooled sensitivity and specificity of 0.90 (95% CI 0.87–0.93) and 0.79 (95% CI 0.73–0.84), respectively, and a high AUC (0.93) at the patient level. In addition, MRCA has incremental value to comprehensive CMR-MPI/LGE protocol for detection of significant coronary stenosis.
In previous studies, CCTA showed a favorable trend toward higher diagnostic performance than MRCA [36]. However, a multicenter study by Hamdan et al. [16] found that CCTA did not have significantly superior performance over MRCA. Moreover, MRCA has several advantages over CCTA. Firstly, MRCA is free of ionizing radiation. Secondly, it is an effective examination for patients with a high calcification score. Thirdly, MRCA can provide a diagnostic image without use of a contrast agent. In addition, β-blocker is not indispensable for MRCA examination.
Multiparametric CMR protocols are a one-stop technique allowing the assessment of cardiac morphology, function, perfusion, and viability as well as coronary artery anatomy. As Table 2 shows, use of MRCA in addition to CMR MPI/LGE increased the overall CMR performance for detection of significant CAD but caused false-positive results. Heer et al. [20] applied a differentiated algorithm such that MRCA use was added in cases of probably normal or probably abnormal perfusion deficits, which increased specificity from 55.6 to 88.9% in comparison with the conventional integration algorithm [20]. Klein et al. [21] showed that MRCA with an excellent image quality combined with CMR MPI/LGE yielded sensitivity of 86% and specificity of 91%. Moreover, MRCA accurately identified stenoses without myocardial ischemia or infarction, which reduced the incidence of false negatives in studies conducted by Zhang et al. [22]. Hence, MRCA has value additive to that of perfusion or LGE imaging for detection of significant coronary stenosis [6, 36, 37].
The technique has developed a lot since MRI was first used to evaluate coronary arteries in the 1980s [37, 38]. Three-dimensional whole-heart MRCA with respiratory motion suppression, T2 preparation, a fat suppression prepulse, and radial k-space sampling has achieved highly promising clinical results and has increasingly been used in clinical routine [5, 39]. In the present meta-analysis, the pooled scan time of included studies was acceptable at 11.10±3.29 min. It showed high sensitivity (0.90) and moderate specificity (0.79) at the patient level, and both the sensitivity (0.86) and the specificity (0.89) were high at the artery level. It was superior to previous meta-analyses that included different scanning approaches along with time. At the patient level, the pooled sensitivity and specificity of previous meta-analyses were 88% and 56% [40], 87.1% and 70.3% [36], 89% and 72% [6], respectively. Moreover, the analysis by Danias et al. [40], with lower pooled estimates, did not include studies conducted by the whole-heart approach. Thus, it can be concluded that the 3D whole-heart approach resulted in better performance than the old scanning protocol.
The meta-regression analysis plots indicated that the studies with a scanner with a field strength of 3.0 T presented better performance. MRI systems with a field strength of 3.0 T, increased signal-to-noise ratio, and increased spatial and temporal resolution can be expected to overcome the shortcomings of systems with a field strength of 1.5 T [5]. In addition, the element channel coils were superior in the 3.0 T group from the included studies; they can partially reduce the noise and therefore increase the signal-to-noise ratio. It was reported that contrast-enhanced MRCA at 3.0 T increased the number of assessable coronary artery segments, especially distal segments, and also improved image quality [18, 19, 29]. However, MRCA at 1.5 T had much broader availability in clinical practice. Twelve of the 18 included studies used a 1.5 T system. Moreover, contrast agent is not required at 1.5 T [39].
Furthermore, we tried to explore whether the examination is suitable for patients with different risk levels. The analysis of the clinical application presented different results for patients with different degrees of pretest probability. As shown in Figure S1, for patients with low suspicion, the examination could be conducted as an exclusionary test. It is also useful for worst-case scenario patients since the posttest probability was significant for both positive and negative results. For patients with high suspicion, it can be considered sufficient to rule in significant stenosis for a positive result with a posttest probability of 91%. Therefore, 3D whole-heart MRCA could be applied to different-hierarchy patients.
In this analysis, the publication years of the included studies ranged from 2005 to 2018. Hence, the MRCA technique was developed slowly with little clinical research in recent years. However, some studies focused on novel motion-compensated and fast imaging technology, which achieved good feasibility in healthy volunteers [41–45]. As the hemodynamic significance of coronary artery stenosis is becoming more important in clinical treatment, stress MPI modalities have been widely used. The additive value of MRCA integration into a comprehensive CMR protocol has more practical significance. To this end, combined MRI may be a noninvasive trend for diagnosis of CAD.
Limitations
Firstly, the included studies did not cover specific patients who have a high calcification score or who had previously undergone coronary artery bypass graft or stent operation, which leads to a limited reference. For these groups of patients it is difficult to estimate the degree of stenosis by another type of noninvasive examination. Secondly, we did not compare the performance of other types of noninvasive examination.


