Introduction
Management and treatment of heart failure has evolved dramatically over the past 40 years. Medications such as angiotensin-converting enzyme inhibitors [1, 2], angiotensin receptor blockers [3, 4], beta blockers [5, 6], and aldosterone receptor antagonists [7, 8] have led to significant improvements in both symptom control and survival in patients with heart failure (HF). Additionally, devices such as implantable cardioverter defibrillators (ICDs) are now recommended for primary prevention of sudden cardiac death in selected patients with ischemic and nonischemic cardiomyopathy [9]. Some HF patients benefit from simultaneous pacing of both ventricles (biventricular pacing) or pacing of one ventricle in patients with bundle branch block, an approach known as cardiac resynchronization therapy (CRT) [10, 11]. CRT can be achieved with a device designed only for pacing or can be incorporated into a combination device with an ICD. CRT is now recommended across a spectrum of patients with HF due to systolic dysfunction in association with QRS delay. The rationale for CRT is based on the observation that the presence of a bundle branch block or other intraventricular conduction delay can worsen HF due to systolic dysfunction by causing ventricular dyssynchrony.
Pathophysiology
Cardiomyopathy can result in structural abnormalities of ventricular myocardium that can affect both electrical activation of the ventricles and mechanical contraction [12, 13].
Electrical Dyssynchrony
Under normal conditions, the myocardium is activated by a uniform, high-velocity electrical waveform that propagates through the His-Purkinje system, resulting in synchronized depolarization of the ventricles. In the diseased myocardium, altered electrochemical substrate and impaired conduction fibers can change the velocity and uniformity of electrical propagation, resulting in areas of delayed activation. This delay manifests itself as lengthening of the QRS complex on the surface 12-lead electrocardiogram. Because the QRS complex represents the summation vector of electrical forces generated by the ventricular myocardium during the course of ventricular systole, a prolonged QRS duration suggests electrical dyssynchrony [14]. Although the prevalence of a prolonged QRS duration (>120 ms) is approximately 20% in the general HF population, it is approximately 35% among patients with symptomatic HF [15].
Mechanical Dyssynchrony
Mechanical dyssynchrony is the result of electrical dyssynchrony and can be intraventricular dyssynchrony within the left ventricle seen commonly in patients with left bundle branch block (LBBB) because of a delay between the relatively early-activated interventricular septum and late-activated posterolateral wall, interventricular dyssynchrony between the left and right ventricles that results from sequential activation of the ventricles because of LBBB or right bundle branch block (RBBB), or atrioventricular (AV) dyssynchrony secondary to prolonged or absent AV nodal conduction, potentially coupled with His-Purkinje system dysfunction [14]. Mechanical dyssynchrony can prolong the periods of isovolumic contraction and isovolumic relaxation and consequently decrease cardiac pumping efficiency. Additionally, a dyssynchronous dilated left ventricle can result in mitral regurgitation because of lack of leaflet coaptation and papillary muscle dysfunction [16]. Although a prolonged QRS duration is the best marker for dyssynchrony, some evidence suggests that mechanical dyssynchrony can be present in the absence of QRS duration prolongation [17]. Because QRS morphology and duration are influenced only when a significant amount of the myocardium is involved, regional discrepancies represented by small vectors are often not evident on the surface electrocardiogram. Therefore, small areas of impaired contractility can produce mechanical dyssynchrony without any detectable electrical conduction disturbance. However, as outlined later, there are no clinical data to suggest that treating mechanical dyssynchrony in the setting of a narrow QRS morphology with currently available CRT devices is beneficial, and may even be associated with adverse outcomes. Long-standing cardiac dyssynchrony leads to remodeling that manifests itself clinically as dilation of the left ventricle, worsening systolic and diastolic function, and progressive HF.
History of CRT
In 1979, temporary biventricular pacing was used to assess tachyarrhythmias due to intraventricular reentry [18]. A decade later, in 1989, Grines et al. [19] described how LBBB reduced the diastolic filling time and the septal contribution to left ventricular (LV) ejection. By the 1990s, a link had emerged between electrical dyssynchrony and impairment of LV function, and it became apparent that LV pacing was more hemodynamically favorable than right ventricular (RV) pacing. The concept of “biventricular pacing,” primarily aimed at HF treatment was developed by Morton Mower, who conceived a method of pacing both ventricles after a predetermined AV interval by connecting two electrodes in series, one in the right ventricle and the other around the free wall of the left ventricle [20]. Proof of this concept came in 1993 when Bakker et al. [21] treated 12 patients with end-stage congestive HF, sinus rhythm, and complete LBBB with biventricular stimulation and showed an improvement in functional capacity, improved systolic and diastolic LV function, and a decrease in mitral regurgitation during 2- and 3-year follow-up. Soon thereafter, Cazeau and colleagues described a four-chamber pacing system that reduced pulmonary capillary wedge pressure and increased cardiac output in a patient with New York Heart Association (NYHA) class IV HF [22] and later described a transvenous CRT implantation method [23]. These initial studies paved the way for the pivotal clinical trials of CRT described herein and have led to their widespread use.
Evidence of Clinical Benefit
Several landmark prospective multicenter studies (Table 1) on the effectiveness of biventricular pacing – namely, Pacing Therapies for Congestive Heart Failure (PATH-CHF) [24], Multisite Stimulation in Cardiomyopathies (MUSTIC) [25], Multicenter InSync Randomized Clinical Evaluation (MIRACLE) [26], Multicenter InSync ICD Randomized Clinical Evaluation II (MIRACLE-ICD) [27], Cardiac Resynchronization Therapy for the Treatment of HF in Patients with Intraventricular Conduction Delay and Malignant Ventricular Tachyarrhythmias (CONTAK-CD) [28], Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure (COMPANION) [10], and Cardiac Resynchronization-Heart Failure (CARE-HF) [11] – were performed between 2000 and 2005. These studies consistently showed that CRT safely improved the patient’s quality of life, NYHA functional class, exercise capacity, LV ejection fraction (LVEF; absolute improvement of 5–15%) and, in COMPANION and CARE-HF, reduced mortality.
Pivotal cardiac resynchronization therapy trials (2001–2005).
| Trial name | Year | Design | Patients and characteristics | Results |
|---|---|---|---|---|
| MUSTIC | 2001 | Randomized, crossover, 2 groups | 41 and 47, NYHA class III HF, normal sinus rhythm and atrial fibrillation | Improved 6-min walk, NYHA class, and QOL, better in nonresponder patients |
| PATH-CHF | 2002 | Single-blind, randomized, crossover, controlled | 42, idiopathic cardiomyopathy, ischemic cardiomyopathy, NYHA class III/IV CHF | Improvement in 6-min walk, maximum oxygen consumption |
| MIRACLE | 2002 | Prospective, randomized, double blind, parallel controlled | 453, idiopathic cardiomyopathy, ischemic cardiomyopathy NYHA III/IV CHF, IVCD | Improved exercise capacity, NYHA class, QOL, and LV function |
| MIRACLE-ICD | 2003 | Prospective, randomized, double blind, parallel controlled | 560, idiopathic cardiomyopathy, ischemic cardiomyopathy, NYHA class III/IV CHF, IVCD, ICD indication | Improved exercise capacity, NYHA class, QOL, and LV function |
| CONTAK CD | 2003 | Prospective, randomized, crossover, parallel controlled | 581, idiopathic cardiomyopathy, ischemic cardiomyopathy, NYHA class III/IV HF, IVCD, ICD indication | Improved exercise capacity, NYHA class, and QOL, trend toward improved mortality |
| COMPANION | 2004 | Multicenter, prospective, randomized, controlled | 1520, idiopathic cardiomyopathy, ischemic cardiomyopathy, NYHA class III/IV HF, IVCD, OMT vs. OMT plus CRT-P vs. OMT plus CRT-D | Reduction in all-cause mortality and all-cause hospitalization |
| CARE-HF | 2005 | Multicenter, prospective, randomized, controlled | 813, idiopathic cardiomyopathy, ischemic cardiomyopathy NYHA class III/IV HF, IVCD | Reduction in all-cause mortality and all-cause hospitalization |
CHF, congestive heart failure; CRT-D, cardiac resynchronization therapy with additional defibrillator capabilities; CRT-P, cardiac resynchronization therapy without additional defibrillator capabilities; HF, heart failure; ICD, implantable cardioverter defibrillator; IVCD, intraventricular conduction delay; LV, left ventricular; NYHA, New York Heart Association; OMT, optimal medical therapy; QOL; quality of life.
Later clinical trials – Resynchronization Reverses Remodeling in Systolic Left Ventricular Dysfunction (REVERSE) [29], Multicenter Automatic Defibrillator Implantation Trial-Cardiac Resynchronization Therapy (MADIT-CRT) [30], and Resynchronization for Ambulatory Heart Failure Trial (RAFT) [31] – focused on whether CRT was beneficial in HF patients with reduced LVEF, a wide QRS complex, and milder symptoms (NYHA class I or II). In these studies, patients were randomized to receive an ICD or an ICD with CRT capabilities (CRT-D), and CRT-D was associated with significant reverse remodeling [29], lower rates of hospitalization [30], and improved survival [31].
As would be expected, the magnitude of mortality benefit (Figure 1) conferred by CRT was much higher in the earlier clinical trials COMPANION and CARE-HF (which compared CRT with optimal medical management in patients with severe HF) than in the later trials MADIT-CRT and RAFT (which compared CRT with ICD in patients with less severe HF).
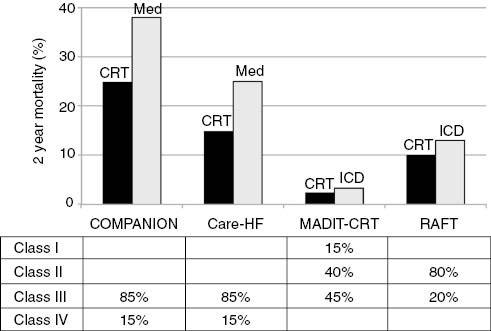
Summary of 2-year Mortality Rates for Cardiac Resynchronization Therapy (CRT) Studies that had Mortality End Points and Relative Percentages of Different New York Heart Association Heart Failure Classification Patients in the Study Populations.
Mortality rates are higher in the COMPANION and CARE-HF trials since patients with severer heart failure were enrolled. Mortality reduction was also more dramatic since CRT (with implantable cardioverter defibrillator [ICD] capabilities in COMPANION and without ICD capabilities in CARE-HF) was compared with best medical therapy (Med) without additional ICD implant.
Mechanism of Benefit
Although several randomized controlled trials have shown improved outcomes with CRT in appropriately selected patients with systolic HF who have an intraventricular conduction delay or LBBB, the molecular basis for these mechanical changes is not well understood. One experimental model suggests that CRT reduces regional and global molecular remodeling, generating more homogeneous activation of stress kinases and reducing apoptosis [32]. Potential mechanisms of benefit include improved contractile function and reverse ventricular remodeling manifested as reductions in LV chamber size and measures of mitral regurgitation. Other hemodynamic and clinical benefits during short-term or long-term biventricular or LV pacing include an increase in cardiac index and a reduction in pulmonary capillary wedge pressure, when compared with normal sinus rhythm or RV pacing [22, 33–35], the ability to tolerate more aggressive medical therapy and neurohormonal blockade, particularly with improved tolerance of beta blockers [36], improved diastolic function among responders to CRT [37], and an improvement in heart rate variability [9].
Device Implantation
Initially, an epicardial lead for LV pacing or a transvenous lead that was not specifically designed and tested for long-term LV pacing was used in clinical trials [28, 38]. Daubert et al. [23] described transvenous coronary sinus lead placement for long-term LV pacing, and this wholly transvenous approach has simplified the implantation procedure and reduced operative risk. However, implantation via the coronary sinus may result in perforation or dissection and other complications, and should be performed only by experienced operators. The most common complication with transvenous CRT implantation is the inability to implant the LV pacing lead successfully in the coronary vein. Other infrequently encountered complications include coronary sinus or coronary vein trauma, pneumothorax, diaphragmatic/phrenic nerve pacing, and infection [10, 11, 39, 40]. Although a theoretical concern existed about a proarrhythmic effect due to alterations in depolarization and repolarization sequences [41], randomized controlled trials have not suggested any excess risk of sudden death or noncardiac death in CRT device recipients.
Follow-up
Once implanted, CRT devices need careful follow-up and at times in-office device reprogramming, which may be more frequent than in patients with standard pacemakers or ICDs. However, one report from the Insync ICD Italian Registry indicated a marked reduction in the number of interrogations requiring reprogramming between the first 6 months of follow-up and subsequent periods, as pacing and CRT delivery parameters were usually optimized soon after implantation and were maintained unmodified thereafter [42]. Therefore, remote follow-up appears to be an acceptable alternative in these patients, especially with the use of algorithms that automatically adjust device settings such as LV pacing output on the basis of daily threshold measurements [43].
Nonresponders
About a third of patients do not achieve the expected clinical benefits after CRT device implantation [44, 45]. Several theories, including inconsistent pacing, myocardial scar burden, lack of mechanical dyssynchrony, and suboptimal lead positioning, have been proposed to explain CRT nonresponse and are bring actively investigated (Figure 2). Although the cause of CRT nonresponse is an area of active investigation, one difficulty in assessing a prognostic response to CRT appears to be the lack of an adequate surrogate. LV reverse remodeling [46], peak Vo2, and natriuretic peptides [47] have been considered but have significant limitations that render them inadequate as potential surrogates.
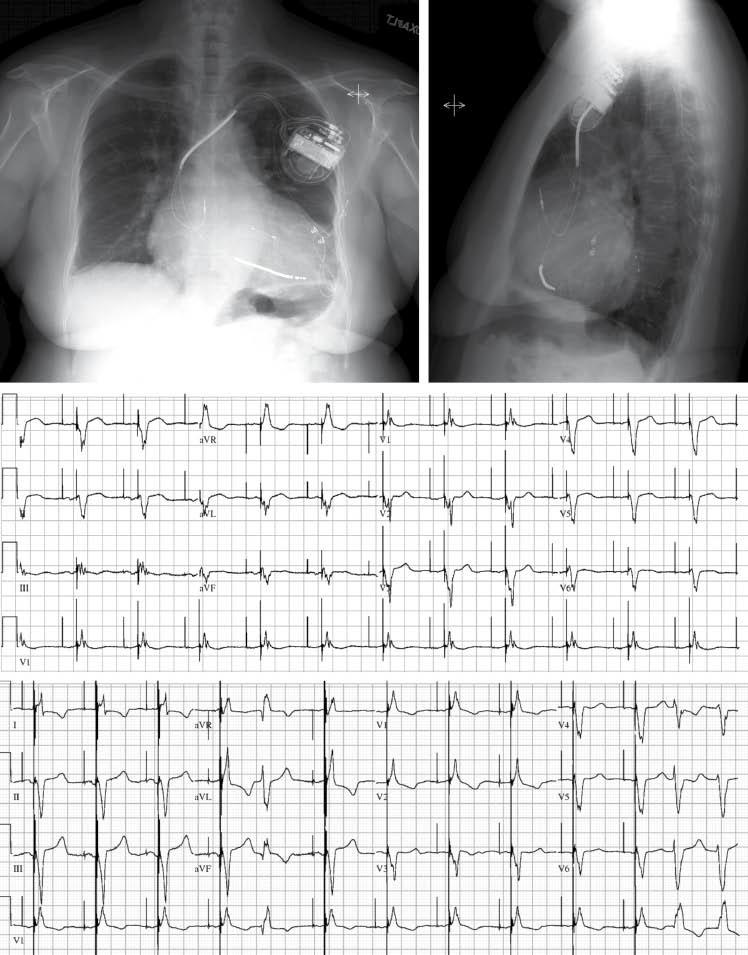
Top row: Chest radiography in a patient who initially received a cardiac resynchronization therapy (CRT) device with additional defibrillator capabilities that used epicardial leads placed apically (posteroanterior view) and anteriorly (best seen in the lateral view). Because of continued symptoms despite CRT, the patient underwent device revision with placement of an endovascular lead placed in a lateral vein via the coronary sinus (arrowheads). The patient’s heart failure symptoms improved after device revisions. Second row: The 12-lead electrocardiogram obtained with the epicardial electrodes is characterized by a predominantly negative QRS complex in lead aVL (suggesting an anterolateral location) Third row: The 12-lead electrocardiogram obtained with the endocardial electrode has a later precordial transition (positive QRS complex in V2), which suggests a more basal pacing site.
Controversies, Areas of Ongoing Research
Patient Selection
CRT in Atrial Fibrillation
An important subset of HF patients are those with atrial fibrillation (AF), who make up 25%–30% of HF patients, and are overrepresented among HF patients with more advanced symptoms. In HF patients with AF, CRT appears to be less effective than in patients in sinus rhythm, which may be due to competition between ventricular depolarization generated by the CRT device and intrinsic ventricular depolarization from the heart’s own electrical conduction system. RAFT [48] included more patients with permanent AF than all other published studies combined, and failed to demonstrate a clear improvement in any clinical or surrogate outcome by CRT in patients with permanent AF, despite a trend toward reduction of HF hospitalization rates. It has been hypothesized that the reduced benefit might be attributed to suboptimal delivery of CRT since only a third of the patients received more than 95% ventricular pacing. Because a CRT device paces only when the patient’s intrinsic rate is lower than the programmed rate, in patients with AF who are prone to rapid ventricular response, their tachycardia may limit the percentage of paced beats and thereby lead to decreased effectiveness of the CRT. Ablation of the AV node to increase the percentage of pacing in AF is likely to increase CRT response, and this strategy is being prospectively evaluated in the Cardiac Resynchronization Therapy and AV Nodal Ablation Trial in Atrial Fibrillation Patients (CAAN-AF) study.
CRT in Narrow QRS Complex Patients
Yu et al. [49] found that LV systolic and diastolic mechanical dyssynchrony is common even in patients with HF with narrow QRS complexes (<120 ms) and proposed that the QRS complex duration is not a determinant of systolic asynchrony, but rather that the assessment of intraventricular synchronicity is probably more important than QRS duration. This early premise formed the foundation for subsequent research evaluating whether CRT is beneficial in patients with a QRS duration less than 120 ms. Although preliminary data from small single-center studies were encouraging [50–52], two contemporary multicenter randomized controlled studies, The Evaluation of Resynchronization Therapy for Heart Failure (LESSER-EARTH) trial [53] and the Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study [54], failed to show a mortality benefit from addition of CRT to an ICD in patients with HF, reduced LVEF and a narrow QRS complex (≤120 ms for LESSER-EARTH and ≤130 ms for EchoCRT). In the LESSER-EARTH trial, CRT did not improve clinical outcomes or induce LV reverse remodeling and adversely affected exercise tolerance [53]. Unlike the LESSER-EARTH trial, patients enrolled in the EchoCRT study had echocardiographic evidence for cardiac dyssynchrony. However, even with this additional enrollment criterion, the EchoCRT study was stopped prematurely for futility on the basis of the recommendation of the data and safety monitoring board because patients who were randomized to the CRT-D group not only did not have reduced rates of death or hospitalization for HF but were also found to have increased mortality [54]. At this point CRT should not be considered in patients with a narrow QRS complex unless they will require substantial ventricular pacing due to AV block (see later).
CRT in RBBB
The use of CRT in patients with a wide QRS complex without LBBB is not well established. A meta-analysis of randomized CRT trials with a total of 5356 patients, of which 1233 patients had non-LBBB conduction abnormalities and were randomly assigned to receive CRT or no CRT, found that there was no reduction in the rate of clinical events in the non-LBBB patient category [55]. Post hoc analysis of MADIT-CRT has shown that subgroups with non-LBBB QRS morphology do not derive significant benefit from CRT [56]. This analysis was designed to determine whether QRS morphology identifies patients who benefit from CRT-D and whether it influenced the risk of primary and secondary end points in patients enrolled in MADIT-CRT. The combined end point of HF event or death was the primary end point of the trial. Death, HF event, ventricular tachycardia, and ventricular fibrillation were secondary end points. Among 1817 patients with available sinus rhythm electrocardiograms at the baseline, there were 1281 (70%) with LBBB, 228 (13%) with RBBB, and 308 (17%) with nonspecific intraventricular conduction disturbances. The latter two groups were defined as non-LBBB groups. Hazard ratios for the primary end point for comparisons of CRT-D patients versus patients who received only an ICD were significantly lower in LBBB patients (0.47; P <0.001) than in non-LBBB patients (1.24; P = 0.257). The risk of ventricular tachycardia, ventricular fibrillation, or death was decreased significantly in CRT-D patients with LBBB but not in non-LBBB patients. Echocardiographic parameters showed significantly greater reduction in LV volumes and improvement in ejection fraction with CRT-D in LBBB patients than in non-LBBB patients (absolute increase in LVEF 12% vs. 9% respectively; P < 0.001). The study authors concluded that no clinical benefit was observed in patients with a non-LBBB QRS pattern.
The recently completed Pacing Affects Cardiovascular Endpoints in Patients with Right Bundle-Branch Block (PACE-RBBB) trial prospectively investigated whether resynchronization by RV pacing alone is equivalent to biventricular pacing in patients with HF and RBBB. The study was completed in August 2014 but no results have been presented.
CRT in AV Block Regardless of LVEF
The effects of CRT have also been evaluated in patients who require pacemaker implantation for AV block irrespective of LVEF. The Biventricular Pacing for Atrioventricular Block to Prevent Cardiac Desynchronization (BIOPACE) study [57] and the Biventricular versus RV Pacing in Patients with Left Ventricular Dysfunction and Atrioventricular Block (BLOCK-HF) study [58] focused on this subgroup of patients. Prior studies, including the Dual Chamber and VVI Implantable Defibrillator (DAVID) [59] and Mode Selection Trial (MOST) [60], showed that RV pacing worsens long-term ventricular function and outcomes. The BLOCK-HF study randomized 691 patients with AV block requiring pacing support with an LVEF of 50% or less to CRT or RV pacing. After an average follow-up of 3 years, CRT significantly reduced the combined primary end point of mortality, HF-related urgent care, and increase in LV end-systolic volume by 26% compared with RV pacing, primarily by reducing LV end-systolic volume. The improvement, however, came at the cost of a higher number of adverse events (83 patients versus 30 patients for CRT and RV pacing, respectively), mostly due to LV lead implant and postimplant issues. In a preliminary presentation of the BIOPACE trial presented at the European Society for Cardiology Congress 2014, after enrollment of 1810 patients with a mean follow-up of 5.6 years, biventricular pacing failed to significantly improve the primary combined outcome of time to death or first HF hospitalization when compared with RV pacing, although a nonsignificant trend in favor of biventricular pacing over RV pacing was detected (hazard ratio 0.87; 95% confidence interval 0.75–1.01; P = 0.08) (http://www.escardio.org/The-ESC/Press-Office/Press-releases/Last-5-years/Biventricular-pacing-disappoints-in-BIOPACE-trial). Implant failure was more common in the CRT group (14.8%) than in the RV pacing group (0%).
Procedural
Site(s) of Pacing
The currently used technique of LV lead placement via the coronary sinus for CRT implantation has remained mostly unchanged since its first description in 1998 by Daubert et al. [23]. A good angiographic result requires the LV lead to be in the posterolateral position with acceptable pacing parameters and no diaphragmatic stimulation (Figure 2). Apical position of the LV lead has been found to have worse outcomes [61]. However, the response to CRT is variable even when the LV lead is in a “good” position. This led to the concept of targeting LV segments with the most delayed activation in order to improve response. The Speckle Tracking Assisted Resynchronization Therapy for Electrode Region (STARTER) trial [62] showed that deploying LV leads in late-activated segments by echocardiographic guidance reduced the risk of death or HF hospitalization (hazard ratio 0.48; 95% confidence interval 0.28–0.82; P = 0.006). However, exact concordance between late-activated segments and LV lead position was achieved in only 30% of patients. Furthermore, segments with likely scar were regarded as missing data. Therefore, it remains unclear whether the benefits of echocardiographic guidance were due to avoidance of scar or targeting of late-activated segments. In the Targeted Left Ventricular Lead Placement to Guide Cardiac Resynchronization Therapy (TARGET) trial, Khan et al. [63] also showed significantly improved response (70% vs. 55%), clinical status, and lower rates of combined death and HF-related hospitalizations with the use of speckle-tracking echocardiography to the target LV lead placement .
Viability of the paced LV segment could also influence the CRT outcome. Leyva et al. [64] used cardiac magnetic resonance imaging to avoid scar and demonstrated improvement in the response to CRT. Similarly, in the TARGET trial, patients with an LV lead placed in a region of scar had poorer outcomes and a higher rate of HF hospitalizations [63]. The benefits of targeting a specific location for lead placement still needs further evaluation, and with current technology will be constrained by venous anatomy, but it seems reasonable to place the electrodes in a late-activating site and to avoid regions of dense scar if technically feasible.
Endocardial LV Pacing
Placing leads in the venous tributaries of the heart leads to initial epicardial depolarization, and investigators have proposed using LV endocardial pacing to maximize the benefit of CRT, but this strategy has not translated into a clinical benefit in early studies [65]. The Wireless Stimulation Endocardially for CRT (WiSE-CRT) study demonstrated the feasibility of providing endocardial stimulation for CRT with a leadless ultrasound-based technology [66]. Transeptal/transmitral and transapical endocardial LV pacing approaches remain experimental, but their use can be considered in unique clinical situations.
Multisite LV Pacing
Leclercq et al. [67] evaluated the role of multipolar LV leads in CRT. Potential advantages of multisite LV pacing include avoidance of diaphragmatic stimulation and availability of multiple pacing vectors. This technology is already being widely adopted and will largely replace leads that use only one or two electrodes. There are some isolated case reports that suggest the potential utility of simultaneous pacing from several spatially separate LV locations (e.g., the anterolateral and posterolateral walls).
Device Optimization
The finding that LV function varies according to AV delays [34] has led to several approaches for device optimization. Echocardiography is commonly used to identify the AV delay yielding optimal LV filling. One frequently used Doppler parameter for echocardiography-guided optimization is the aortic velocity-time integral [48, 68], In one single-blind randomized trial the impact of AV delay optimization based on the aortic velocity-time integral was evaluated and showed improvement in NYHA functional class in optimized versus control patients [37]. Many single-center studies have shown improvement in cardiac output by tailored Doppler echocardiography – guided AV delay optimization [11, 25, 34].
In addition to programming the AV delay, all currently available CRT devices allow individual programming of the interval between LV and RV pacing stimuli (VV) and which stimulus will be delivered first. Device-based AV/VV proprietary algorithms have been developed by all manufacturers. Device-based interval optimization by the QuickOpt® algorithm was inferior to echocardiographic optimization in the Frequent Optimization Study Using the QuickOpt Method (FREEDOM) [69]. In another study, AV optimization by the Smart-AV® algorithm did not lead to LV reverse remodeling compared with nominal settings in the Comparison of AV Optimization Methods Used in Cardiac Resynchronization Therapy (Smart-AV) study [70]. On the other hand, the Adaptive Cardiac Resynchronization Therapy study showed that an algorithm that provides automatic selection between synchronized LV or biventricular pacing, as well as AV and VV optimization, was comparable to echocardiographic optimization [71]. However, it remains to be determined whether this is attributable to AV/VV optimization or to the pacing mode.
In summary, although regarded as the gold standard, echocardiographic optimization has not been shown to improve outcomes in large multicenter randomized trials, but appears as effective as nominal device settings and at this point should still be considered superior to the current device-based algorithms for optimizing the timing of pacing stimuli [70].
Cost-effectiveness
The incremental cost-effectiveness ratio (ICER), or the additional cost of a quality-adjusted life-year (QALY) saved, is a widely accepted measure of the cost of medical interventions [72]. An acceptable ICER is less than $50,000 in the United States.
Although CRT devices are expensive, the costs are offset in part by savings from reduced rates of hospitalizations for HF as suggested by the acceptable cost-effectiveness ratios for CR, in the cost-effectiveness analyses from the COMPANION and CARE-HF trials [73, 74]. Data from the COMPANION trial were extrapolated to develop a cost-effectiveness estimate for CRT alone (CRT-P) and CRT-D. The cost-effectiveness ratio projected over 7 years was estimated to be $19,600 per QALY for CRT-P and $43,000 per QALY for CRT-D [74]. Similarly, the analysis of CARE-HF patients found that over a median follow-up of 29 months, the cost-effectiveness ratio was €19,319 per QALY [74]. For mild HF, a REVERSE trial analysis showed that over a 10-year time frame, CRT “on” was associated with an ICER of €14,278 per QALY saved [75]. Similarly, a MADIT-CRT analysis yielded an ICER for CRT-D of $58,330 compared with ICD implantation [76]. When the ICER was calculated for a longer time period and for inclusion of a preimplantation LBBB, it decreased substantially to less than $10,000 [76]. Therefore, one must exercise caution in interpreting these numbers as the analyses are based on extrapolated results and the results vary substantially according to changes in input variables such as the magnitude of reduction in HF admission rates, cost of device implantation, and frequency of device complications. However, taken together, these analyses demonstrate that CRT-P and CRT-D are at least as cost-effective as many other medical interventions for treating HF.
Current Guideline Recommendations
Recommendations for CRT first appeared in the 2008 American College of Cardiology/American Heart Association/Heart Rhythm Society guidelines for device-based therapy of cardiac rhythm abnormalities [77], and more recently in the 2012 American College of Cardiology Foundation/American Heart Association/Heart Rhythm Society focused update incorporated into the 2008 guidelines (2012 focused update) [78] and the 2013 American College of Cardiology Foundation/American Heart Association guideline for the management of HF [9]. Importantly, the current guidelines for CRT are consistent in their recommendations (Figure 3). In addition, all guidelines recommend CRT with the expectation that appropriate medical therapy is already being provided, usually described as “guideline-directed medical therapy.”
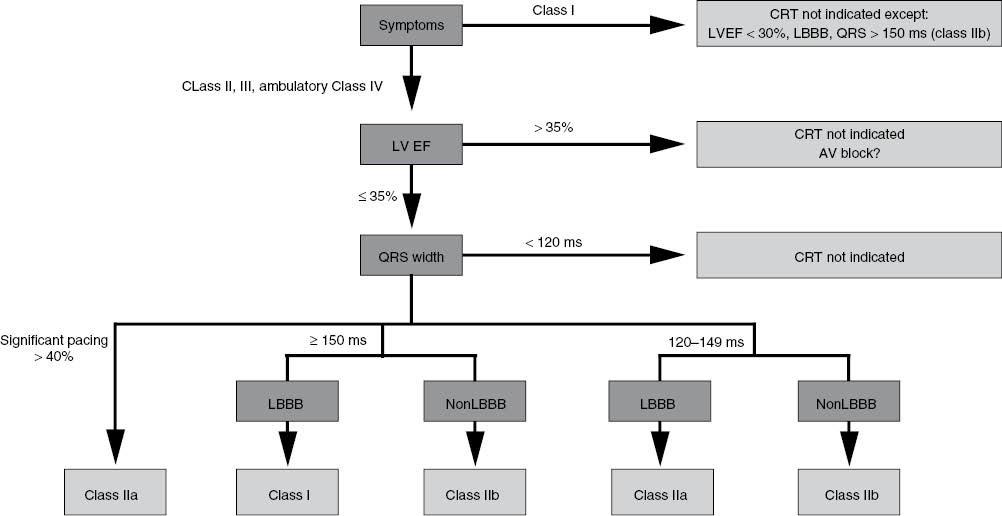
Flowchart Showing Current Recommendations for Cardiac Resynchronization Therapy Based on the 2012 Update of the 2008 American College of Cardiology/American Heart Association/Heart Rhythm Society Guidelines for Device-Based Therapy of Cardiac Rhythm Disorders and the 2013 American College of Cardiology Foundation/American Heart Association Heart Failure Guidelines.
For CRT recommendations, the 2012 focused update included data from the REVERSE trial, MADIT-CRT, and RAFT, which studied the use of CRT in patients with less severe symptoms, to make recommendations. In addition the 2012 focused update also considered the growing body of evidence suggesting that CRT is less effective in patients with narrower QRS durations (<150 ms) or non-LBBB QRS patterns. In the 2008 guidelines, the only class I recommendation for CRT was for patients with relatively severe symptoms (NYHA functional class III or IV HF) accompanied by sinus rhythm, LVEF of 35% or less, and a QRS duration of 120 ms or more with no specific consideration of QRS morphology. For the 2012 focused update (and the 2013 HF guidelines), this class I indication was expanded to patients with NYHA class II HF, sending the clear message that CRT “is indicated” for a population with milder symptoms. At the same time, the 2012 focused update refined this single class I recommendation by confining it only to patients with LBBB and a QRS duration of 150 ms or more (Figure 3). Patients with LBBB and a QRS duration of only 120–149 ms and those with a non-LBBB pattern and a QRS duration of 150 ms ir more, included in class I in 2008, were downgraded to class IIa, and those patients with a non-LBBB pattern with a QRS duration between 120 and 149 ms were downgraded further to class IIb. For patients with LBBB (regardless of QRS duration), patients with NYHA class II symptoms are now included as candidates for CRT (class I for a QRS duration greater than 150 ms and class IIa for a QRS duration of 120–149 ms). For patients with non-LBBB block, if the QRS duration was less than 150 ms, the indication was not expanded beyond patients with NYHA class III/ambulatory class IV symptoms, and CRT is “not recommended” (a class III recommendation) for patients with NYHA class II symptoms. The evolution in recommendations was based on the consistent results of studies demonstrating that non-LBBB conduction abnormalities are associated with failure to benefit from CRT [55, 79–81].
Conclusion and Take-Home Message
CRT remains one of the most innovative treatments of HF. It is a clinically beneficial and cost-effective treatment for patients with mild to severe HF and a wide QRS complex. Ongoing and future research will continue to investigate ways to refine implantation, optimize device function, and better elucidate the cause of nonresponse to CRT with the goal of ultimately reducing nonresponder rates and identifying other groups of patients who may benefit from this therapy.

