1. INTRODUCTION
The proper segregation of chromosomes during cell mitosis is essential for maintaining chromosomal stability and preventing cancer development. The centromere and kinetochore are crucial structures involved in this process [1, 2]. Defects in proteins associated with the centromere and carcinogenesis can cause chromosomal instability and disrupt the cell cycle, ultimately leading to cell death [3]. One such protein is the Holliday junction recognition protein (HJURP), which mediates the deposition of histone H3 variant-centromere protein A (CENP-A) at centromeres during the G1 phase of the cell cycle [4]. It has been determined that the TLTY box located within the N-terminal domain of HJURP binds to the centromere-targeting domain of CENP-A [5–7], and the interaction between these two proteins is critical for maintaining chromosomal stability and successful mitosis [8]. The HJURP activation requirement for cancer cell immortality was initially identified in 2007 [9, 10]. Furthermore, overexpression of HJURP has been observed in various tumor tissues and is associated with poor prognosis, thus making HJURP a potential prognostic cancer biomarker [11–14]. Given these findings, HJURP could be a key target for cancer treatment. This review summarizes recent studies that have focused on the mechanisms regulated by HJURP in human cancers and the corresponding effects. The feasibility of HJURP as a promising target for anti-tumor treatment is also discussed.
2. EXPRESSION OF HJURP IN CANCERS
In recent years several studies have demonstrated that HJURP has a significant role in the promotion and progression of cancer. Abnormal upregulation of HJURP has been demonstrated in various human cancers based on public genomics databases and independent patient cohorts. Two pan-cancer analyses were performed to determine the relationship between HJURP and cancer [14, 15]. Specifically, these studies showed that HJURP expression is upregulated in most human cancers. Low levels of DNA methylation may lead to overexpression of HJURP, while high mutation frequencies suggest that HJURP acts as an oncogene and may be involved in drug resistance mechanisms [14, 16, 17]. Additionally, overexpression of HJURP is responsible for poor prognosis and cancer progression. Bioinformatics databases were used to elucidate the molecular mechanisms involved in HJURP. HJURP mainly participates in cell cycle and p53 signaling pathways, which are supported by existing studies [10, 18]. HJURP expression increases during cell cycle progression. G2 is the phase with the highest expression, suggesting a role for HJURP in cell cycle regulation [15]. Furthermore, HJURP has been shown to mediate tumor immune evasion and interacts with immune infiltration and T cell immune exclusion, thus promoting cancer progression and metastasis [15].
To provide insight into the pattern of HJURP expression and the correlation with cancer survival rates, two online tools (the UALCAN Interactive web and Gene Expression Profiling Interactive Analysis [GEPIA]) were used to analyze expression of HJURP mRNA, and Kaplan-Meier survival analysis was performed using data from the Cancer Genome Atlas (TCGA) database [19, 20]. Our analysis revealed that the HJURP mRNA levels were significantly higher in 19 tumor tissue types compared to normal tissue ( Figure 1 ). Moreover, a high level of HJURP was associated with shorter overall survival (OS) and disease-free survival (DFS) in patients with adrenocortical carcinoma (ACC), kidney renal clear cell carcinoma (KIRC), head and neck squamous cell carcinoma (HNSC), kidney chromophobe (KICH), kidney renal papillary cell carcinoma (KIRP), brain lower grade glioma (LGG), liver hepatocellular carcinoma (LIHC), lung adenocarcinoma (LUAD), mesothelioma (MESO), pancreatic adenocarcinoma (PAAD), pheochromocytoma and paraganglioma (PCPG), prostate adenocarcinoma (PRAD), sarcoma (SARC), skin cutaneous melanoma (SKCM), uterine corpus endometrial carcinoma (UCEC) and uveal melanoma (UVM) ( Figure 2 ). We also detected a strong positive correlation between HJURP expression and the pathologic stage of patients with ACC, breast invasive carcinoma (BRCA), kidney chromophobe (KICH), KIRC, KIRP, LIHC, LUAD, ovarian serous cystadenocarcinoma (OV), PAAD, SKCM, testicular germ cell tumors (TGCT) and thyroid carcinoma (THCA) ( Figure 3 ). These results suggest that HJURP has the potential to serve as a biomarker for prognosis and monitoring of disease progression in cancer patients; however, the expression of HJURP and its prognostic implications can vary among different cancer types. For example, high HJURP expression is strongly associated with a better prognosis in thymoma (THYM) and colon adenocarcinoma (COAD), indicating a potential tumor-suppressive role. Therefore, the relationship between HJURP, and tumor occurrence and development is complex and may depend on the specific type of cancer. It is important to note that these findings are based solely on data analysis and may be subject to bias due to small sample sizes. Therefore, further research is warranted to fully understand the precise function of HJURP in different types of cancer.
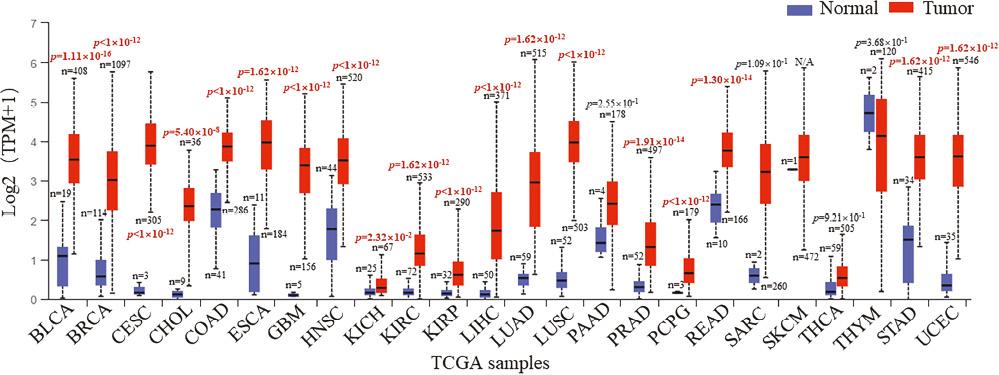
Expression of HJURP in human tumor and normal tissues. Expression of HJURP is higher in tumor tissues than normal tissues for the following types of cancer: BLCA; BRCA; CESC; CCA; COAD; ESCA; GBM; HNSC; KICH; KIRC; KIRP; LIHC; LUAD; LUSC; PCPG; PRAD; READ; STAD; and UCEC.
3. FUNCTIONS AND REGULATION OF HJURP IN CANCERS
3.1 Respiratory system cancer
Lung cancer is a leading cause of cancer-related deaths worldwide. Non-small cell lung cancer (NSCLC) accounts for approximately 85% of cases and the global incidence of NSCLC is increasing [21]. The clinical significance of HJURP has been analyzed in lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC), in which HJURP is highly expressed in tumor tissues and associated with a poor prognosis [22, 23]. Gene set enrichment analysis (GSEA) revealed that HJURP-related genes are mainly enriched in the G1, ataxia telangiectasia mutated (ATM), cell cycle, and extracellular regulated protein kinases (ERK) pathways in LUSC, and associated with basal transcription factors, the cell cycle, homologous recombination, NSCLC, oocyte meiosis, the p53 signaling pathway, cancer pathways, RNA degradation, and spliceosomes in LUAD [22, 23]. Furthermore, HJURP was shown to be remarkably associated with 28 types of tumor-infiltrating lymphocytes, especially activated CD4 cells in patients with LUAD [22]. The active role of immune infiltration in cancer growth and development highlights the potential carcinogenicity of HJURP [24].
HJURP has been shown to have an oncogenic role in NSCLC cells [9]. During the mitotic cell cycle, HJURP localizes to the nucleolus in the G1 phase and interacts with hMSH5, NBS1, and MRE11 in sub-nuclear foci during the S phase. HJURP then relocates to the nucleolus in the late G2 phase and finally localizes to pre-nucleolar bodies during telophase. Further studies have demonstrated that HJURP is a downstream target in the ATM signaling pathway and is involved in DNA double-strand break (DSB) repair via the homologous recombination pathway. Knocking down HJURP significantly inhibits viability of U2OS cells by inducing cell cycle arrest at the G2/M phase. HJURP also protects cancer cells from genomic instability and senescence by deregulating recombination in the rDNA region.
NSCLC is known to have a high rate of metastasis at late stages, leading to a high mortality rate. Clinical data have shown that high HJURP expression is associated with distant metastases and an advanced tumor node metastasis (TNM) stage in NSCLC [25]. In vitro experiments demonstrated that silencing HJURP induces apoptosis and inhibits proliferation, migration, and invasion of NSCLC cells by suppressing the epithelial-mesenchymal transition (EMT) and wingless-related integration site (Wnt)/β-catenin signaling pathway.
3.2 Digestive system cancer
3.2.1 Oral cancer
Oral cancer ranks sixth as the most prevalent form of malignancy globally [26]. Dietary habits are a significant risk factor for oral cancer [26]. Research has shown that HJURP is highly expressed in most oral cancer tissues, and is associated with a reduced overall survival rate [27]. Depleting HJURP has been shown to inhibit oral cancer cell growth by inducing cell cycle arrest and senescence, with the involvement of CENP-A. In oral squamous cell carcinoma (OSCC), p53 is the most commonly mutated gene with a concurrent general mutation in HJURP [28]. Additionally, HJURP is upregulated in p53-null human cancers, including breast cancer, melanoma, and pancreatic cancer [18]. Loss of p53 leads to increased HJURP and CENP-A levels, promoting cell growth and the cell cycle via the DREAM-CDE/CHR pathway. Depleting HJURP activates p53 inversely, leading to cell cycle arrest at the G1 and G2/M phases. In the absence of p53, HJURP deletion leads to the rapid loss of CENP-A from the centrosome, causing centromere dysfunction, aneuploidy, and p53-independent apoptosis. These results indicated that targeting HJURP holds great promise in cancer therapy, particularly in p53-deficient cancers.
3.2.2 Hepatocellular carcinoma (HCC)
Polymorphisms in genes at the molecular level can alter the amino acids encoded by codons and affect gene function, which can lead to the development of cancer [29]. Specifically, a significant association has been demonstrated between the rs3771333 polymorphism in the HJURP gene and susceptibility to hepatitis B virus (HBV)-related HCC [30, 31]. The rs3771333 polymorphism is a non-synonymous single nucleotide polymorphism (SNP) located in exon 8 of the HJURP gene. This polymorphism results in a change from GAA-to-GAC in the codon encoding the 568th amino acid residue, leading to the substitution of glutamic acid by aspartic acid. According to computer predictions, the non-synonymous SNP rs3771333 does not appear to affect protein structure, but is expected to alter binding of the exon splicing enhancer (ESE) SF2/ASF or the number of ESE binding sites [31]. The presence of the rs3771333 A/C or C/C genotypes is associated with a higher risk of HBV-related HCC compared to the A/A genotype among Chinese, which express lower levels of HJURP mRNA and protein in Epstein-Barr virus (EBV)-transformed blood lymphocytes. Given the impact of HJURP on DSB repair and the potential acceleration of HCC onset and progression caused by impaired DSB repair, reducing HJURP expression could potentially contribute to HCC predisposition [32]. In addition to the Oncomine database, analysis of HCC tissues and cells has shown high levels of HJURP expression, which is correlated with poor patient survival rates and several pathologic factors, including patient age, microvascular invasion, tumor size, and stage [33, 34]. Furthermore, HJURP is strongly correlated with tumor number and differentiation and has been shown to play a crucial role in the immunosuppressive tumor microenvironment (TME) [35, 36]. The pro-proliferative effect of HJURP on HCC has been demonstrated in vitro and in vivo [33]. Mechanistic studies have investigated the role of p21 in HJURP-induced cell cycle transition, revealing that HJURP induces p21 ubiquitination and facilitates its translocation from the nucleus to the cytoplasm via the mitogen-activated protein kinase (MAPK)/ERK1/2 and protein kinase B (AKT)/glycogen synthase kinase 3β (GSK3β) signaling pathways. Furthermore, HJURP upregulates sphingosine kinase 1 (SPHK1) and promotes EMT, thereby enhancing cell migration and invasion in vitro and promoting tumor metastasis to the lungs in vivo [34].
3.2.3 Pancreatic cancer
Pancreatic cancer is a highly aggressive and malignant form of digestive system cancer that is often associated with a poor prognosis [37]. The expression of HJURP is significantly higher in pancreatic cancer cells and tissues compared to adjacent normal tissues, and high expression of HJURP predicts a lower survival rate [38]. Furthermore, studies have demonstrated that HJURP promotes pancreatic cancer cell viability, sphere formation, migration, and invasion in vitro, as well as tumorigenesis and metastasis in vivo. The mechanism underlying these effects is related to the ability of HJURP to promote binding of histone 3 lysine 4 dimethylation (H3K4me2) at the promoter region of murine double minute 2 (MDM2), which results in the degradation of p53 and blockage of downstream signaling pathways.
3.2.4 Cholangiocarcinoma (CCA)
The ectopic expression of HJURP and its prognostic significance in various subtypes of CCA, including intrahepatic (iCCA), perihilar (pCCA), and distal (dCCA) CCA, were investigated [13]. HJURP is highly expressed in all subtypes of CCA and serves as an independent prognostic biomarker in iCCA and pCCA, but not dCCA. Additionally, the correlation between HJURP expression and clinicopathologic factors varies between different subtypes of CCA, with iCCA showing the most significant correlation between HJURP and advanced tumor infiltration (T stage) [13]. Given that the efficacy of targeted therapy for CCA is currently limited, the identification of HJURP not only provides valuable prognostic information, but also offers a novel target for CCA treatment [39].
3.2.5 Colorectal cancer (CRC)
CRC is a highly prevalent malignant digestive tract cancer that is responsible for a large number of cancer-related deaths worldwide [40]. Although the expression of HJURP protein is higher in CRC tissues compared to normal tissues, no significant association between HJURP expression and clinicopathologic factors, such as tumor stage and metastasis, has been demonstrated [41]. In comparison to other types of cancer, the prognostic relevance of HJURP in CRC is not as significant, which may be due to the small sample size of studies; however, based on Kaplan-Meier analysis, high expression of HJURP has been shown to be correlated with shorter survival rates in CRC patients. In vitro studies have demonstrated that HJURP promotes cell anchorage-independent growth, migration, and invasion in CRC cell lines. Further research is required to elucidate the molecular mechanisms underlying these carcinogenic effects.
3.2.6 Gastric cancer
Gastric cancer is a prevalent, often fatal tumor that is the third leading cause of cancer-related deaths worldwide. The most common pathologic type of gastric cancer is stomach adenocarcinoma (STAD), which arises from gastric mucosal glands and accounts for approximately 95% of gastric cancer cases [42]. Recent studies have shown that HJURP expression is significantly higher in gastric cancer tissues compared to normal tissues, and this expression is correlated with lymphatic metastasis, TNM stage, and the presence of cancer thrombus [43]. Moreover, microarray and bioinformatic analyses have shown that HJURP is upregulated in STAD and associated with TNM stage and a longer survival rate in patients without distant metastasis. These findings suggest that HJURP could be a valuable early prognostic marker for gastric adenocarcinoma [44, 45].
3.3 Reproductive system cancer
3.3.1 Breast cancer
Breast and ovarian cancer are the most common malignant cancers in women, both of which have common risk factors and gene expressions [46]. According to the Gene Expression Omnibus (GEO) database, the expression of HJURP in breast cancer cell lines is higher than normal breast cells, particularly in invasive ductal carcinomas (IDC) [11]. HJURP was strongly associated with shorter survival time and pathologic factors of breast cancer, including estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, advanced Scarff-Bloom-Richardson (SBR) grade, young age, and Ki67 proliferation indices. Additionally, HJURP is a better independent prognosis marker for patients with ER-positive luminal A breast carcinoma than the currently used proliferation marker, Ki67 [47]. Furthermore, the levels of HJURP and YAP1 expression are significantly higher in triple-negative breast cancer, which is the most aggressive subtype with poor prognosis and limited therapeutic targets, and jointly associated with poor recurrence-free survival [48]. HJURP inhibits YAP1 ubiquitination and regulates the yes-associated protein 1 (YAP1)/N-myc downstream regulated 1 (NDRG1) pathway to promote cell proliferation and decrease response to chemotherapy. Therefore, HJURP may be a candidate prognosis biomarker for breast cancer and a therapeutic target against drug resistance, especially triple-negative breast cancer [49]. Interestingly, although patients with a low level of HJURP generally have longer survival times, clinical data and in vitro experiments suggest that HJURP enhances sensitivity to radiotherapy and effectively prolongs survival time after radiotherapy in breast cancer, possibly due to the role in DSB repair [11].
3.3.2 Ovarian cancer
Ovarian cancer is highly aggressive and one of the most lethal gynecologic tumors [50]. Serous ovarian carcinoma is the most common subtype of recurrent ovarian cancer [50]. HJURP, which is mainly expressed in the nucleus of serous ovarian cancer cells, is elevated in advanced serous ovarian cancer tissue samples. This increase in HJURP is significantly associated with a lower overall survival rate, lymph node metastasis, and clinicopathologic parameters, all of which are indicative of malignant progression [50, 51]. The molecular mechanism underlying HJURP in the progression of ovarian cancer has been established. HJURP targets CENP-A and regulates EMT to facilitate the metastatic capacity of ovarian cancer cells. Silencing HJURP inhibits cell proliferation in vitro and in vivo by inducing apoptosis and cell cycle arrest by regulating proteins related to the cell cycle [51, 52]. GO analysis has identified that differential genes in ovarian cancer are enriched in regulation of the cell cycle [51, 52]. WEE1 and MYC are positively correlated with HJURP according to the GEPIA database and experiments. Silencing HJURP enhances the sensitivity to cisplatin and a Wee1 inhibitor (AZD1775) alone or in combination within ovarian cancer cells by inducing apoptosis partly through the MYC/WEE1 pathway and inhibiting DNA damage repair. These results suggested a promising role of HJURP interference in anti-cancer treatment against chemoresistance.
3.3.3 Prostate cancer
Chen et al. [53] initially reported high expression of HJURP in prostate cancer, which has been shown to be associated with several key indicators, including elevated levels of prostate cancer-specific antigen, a high Gleason score, advanced pathologic stage, metastasis, and biochemical recurrence-free survival. Subsequent clinical data from hospitals have also identified the prognostic value of HJURP in prostate cancer [54]. In vitro and in vivo experiments have confirmed the pro-proliferation effect of HJURP in prostate cancer by inducing cell cycle transition. Further investigations aimed to clarify the molecular mechanisms underlying these effects revealed that HJURP activates CDKN1A ubiquitin-dependent proteasome degradation via the GSK3β/JNK signaling pathway.
3.4 Urinary system cancer
3.4.1 Bladder cancer
Bladder cancer is a prevalent malignant cancer of the urinary tract that involves several biological processes, including the cell cycle pathway and chromatin regulation [55]. A previous study showed that HJURP is markedly upregulated in bladder cancer tissues. Furthermore, knockdown of HJURP has been shown to inhibit bladder cancer cell proliferation [56]. Subsequent mechanistic studies revealed that silencing HJURP induces the generation of reactive oxygen species (ROS), apoptosis, and cell cycle arrest at the G0/G1 phase through the peroxisome proliferator-activated receptor γ (PPARγ)-sirtuin 1 (SIRT1) pathway. High HJURP expression was detected in bladder urothelial carcinoma (BLCA), the most common type of bladder cancer, and was found to be a poor prognostic indicator [57]. Spearman analysis showed that HJURP is upregulated with genes related to the c-Jun N-terminal kinase (JNK)/signal transducer and activator of transcription 3 (STAT3) pathway, cell proliferation, and cell cycle. Further studies verified that HJURP activates the JNK/STAT3 signaling pathway to regulate cell proliferation, cell cycle, and apoptosis in vitro.
3.4.2 Renal cancer
Renal cancer, most often renal cell carcinoma (RCC), is a malignant tumor of the urinary tract, second only to bladder cancer with respect to incidence and mortality [58, 59]. Unlike other cancers, HJURP is downregulated in RCC tissues and cell lines. Overexpression of HJURP reduced the viability and colony formation of RCC cell lines and induced apoptosis, cell cycle arrest at the G0/G1 phase, and oxidative stress through the PPARγ/SIRT1 signaling pathway [60]. In contrast, KIRC is the most common subtype of RCC, and HJURP was identified as a hub gene positively associated with the metastasis, progression, and worse prognosis of KIRC [61, 62]. Furthermore, correlation analysis showed that HJURP is closely related to immune infiltration and senescence processes, implying its critical role in the TME, which requires further investigation [62, 63].
3.5 Central nervous system cancer
Astrocytoma is the most common type of central nervous system cancer, and glioblastoma is the most malignant subtype [64]. HJURP is overexpressed in patients with astrocytomas and glioblastomas of different grades, and is closely associated with poor prognosis and tumor aggressiveness [65, 66]. Consistent with previous findings, knocking down HJURP inhibited the viability and migration of glioblastoma cells by inducing cell cycle arrest, premature senescence, and apoptosis without affecting non-tumor cells [65, 67, 68]. Additionally, silencing HJURP increased the sensitivity of glioblastoma cells to irradiation, leading to higher rates of apoptosis and cell death. Molecular studies have shown that in addition to an interaction with CENP-A, HJURP was positively regulated by Kruppel-like factor 11 (KLF11), thus enhancing the proliferation and migration of glioblastoma cells [67, 68]. Therefore, targeting HJURP could be a potential adjunct therapy for glioblastoma treatment.
3.6 Endocrine system cancer
Anaplastic thyroid cancer is a highly aggressive and deadly subtype of thyroid cancer that currently has limited therapeutic effect. Effective targeted therapy is urgently needed to improve outcomes for patients [69]. HJURP is a potential therapeutic target that is upregulated in anaplastic thyroid cancer. Further investigation is needed to verify its efficacy as a treatment option [70].
3.7 Other cancers
3.7.1 Multiple myeloma
Multiple myeloma is a type of blood cancer that is currently considered incurable; however, there is promising research on targeted drugs that could potentially overcome drug resistance and improve patient prognosis [71]. HJURP has been identified as a super-enhancer (SE)-associated gene that is frequently overexpressed and linked to cancer progression and poor patient outcomes in t(4;14)-positive multiple myeloma [72]. Further research has revealed that transcription of HJURP was regulated by the nuclear receptor-binding SET domain protein 2 (NSD2)/bromodomain-containing protein 4 (BRD4) complex, which activated SE activity upstream of the gene. Notably, knocking down HJURP has been shown to inhibit cell growth by inducing apoptosis, suggesting that SE-driven HJURP could be a potential therapeutic target for t(4;14)-positive multiple myeloma. These findings offer hope for improving treatment options for patients with this type of blood cancer.
3.7.2 Thymic epithelial tumor (TET)
Recent research has shed light on the relationship between HJURP and TETs [73]. HJURP expression is primarily confined to the nucleus, although cytoplasmic localization is more commonly observed in B3- and C-type TETs than in other TET subtypes, and is positively associated with advanced Masaoka-Koga stage. In contrast, nuclear HJURP expression does not exhibit such correlations, suggesting that the subcellular localization of HJURP may have distinct functional implications.
4. SUMMARY AND PERSPECTIVES
Pan-cancer analysis has revealed that HJURP is frequently overexpressed in a significant number of human cancers, and is closely associated with poor prognosis and disease progression. Recent studies have further confirmed that HJURP overexpression promotes cell proliferation, metastasis, and drug resistance, which may be attributed to its involvement in repairing DNA damage to maintain the genomic stability of cancer cells. Silencing HJURP induces apoptosis, cell cycle arrest, and senescence, leading to anti-tumor effects. In addition, inhibiting HJURP expression increases the sensitivity of breast and ovarian cancers to chemotherapy and glioblastomas to radiotherapy [48, 51, 67]. In contrast, HJURP overexpression may help prolong survival after radiotherapy in breast cancer patients, but only to a limited extent compared to patients with lower levels of HJURP expression [11]. The in vivo efficacy of HJURP as an anti-tumor target has only been tested in HCC, and pancreatic, prostate, and ovarian cancers, and should be validated in other cancer types.
Furthermore, the oncogenic properties of HJURP and its underlying regulatory mechanisms have been elucidated. Studies have shown that HJURP is involved in multiple pathways that are related to the cell cycle, drug resistance, cell proliferation, DSB repair, apoptosis, oxidative stress, and EMT in different human cancers ( Table 1, Figure 4 ). Variations in regulatory mechanisms of the identical pathway can occur across different cancer types, such as the PPARγ/SIRT1 signaling pathway in RCC and bladder cancer. Among a number of downstream targets, it is worth noting that the interaction between HJURP and CENP-A has a critical role in cell cycle arrest and cell death. Phosphorylation of HJURP is required for centromeric recruitment and loading of CenH3CENP-A at centromeres by binding to DNA during the regulation of the cell cycle [44]. HJURP, identified as the CENP-A chaperone, contains a cyclin A binding site that regulates specific inhibitory phosphorylation to maintain cell-cycle control of CENP-A assembly [10]. Knockdown of HJURP impairs CENP-A deposition and maintenance at centromeres in cell mitosis, which leads to abnormal chromosome instability and separation defects, and subsequently induces senescence and death of cancer cells [8, 74, 75]. HJURP depletion also represses CENP-A levels that induce cellular senescence through the p53-dependent pathway [76]. Suppression of HJURP can be exploited to induce senescence, and the combination with senolytic agents is now considered a promising anti-cancer therapy [77]. Despite the association with diverse immune signaling pathways and regulation of multiple immune cell infiltration, the precise functions of HJURP on immune regulation remain elusive and need further exploration.
Regulatory factors involved in the HJURP-regulated signaling pathway.
| Regulatory factor | Cell line | Biological function | Ref. |
|---|---|---|---|
| CCND1 | A549, H1299; UM-UC 3, T24, EJ; HCC-LM3, Huh7, SMMC-7721; Hey, A2780; SKOV3, A2780; A498; Capan-2, SW 1990 | Regulation of cell cycle, promotes cell growth, migration, and invasion | [25, 33, 38, 51, 52, 56, 60] |
| p53 | p53-null MEF, p53-null E1A and HRas-V12 transformed MEFs; MPC-1; Capan-2, SW 1990; UM-UC 3, T24, EJ; | Regulation of cell growth, migration, invasion, immune escape, DNA damage repair and cell cycle, involved in MDM2/p53 signaling pathway | [18, 28, 38, 56] |
| CENP-A | UM-UC 3, T24, EJ, Hey, A2780, T98G | Regulation of cell cycle and senescence, involved in chromatin assembly at the centromere | [52, 56, 67] |
| CDK2/4 | UM-UC 3, T24, EJ; HCC-LM3, Huh7, SMMC-7721; Hey, A2780; T24, TCCSUP | Regulation of cell cycle | [33, 52, 56, 57] |
| p21 | HCC-LM3, Huh7, SMMC-7721; SKOV3, A2780; Capan-2, SW 1990; LNCaP, PC3 | Regulation of cell cycle | [33, 38, 51, 54] |
| GSK3β | UM-UC 3, T24, EJ; HCC-LM3, Huh7, SMMC-7721 | Involved in AKT/GSK3β pathway, GSK3β/JNK pathway, regulation of cell cycle | [33, 54, 56] |
| ERK1/2 | UM-UC 3, T24, EJ; LNCap, PC3; HCC-LM3, Huh7, SMMC-7721 | Involved in MAPK/ERK pathway, regulation of cell cycle | [33, 54, 56] |
| Vimentin | A549, H1299; HCC-LM3, Huh7; A2780, HEY, SKOV3 | Regulation of EMT, promotes cell migration and invasion | [25, 34, 51] |
| N-cadherin | A549, H1299; HCC-LM3, Huh7 | Regulation of EMT, promotes cell migration and invasion | [25, 34] |
| CDK6 | SKOV3, A2780; T24, TCCSUP | Regulation of cell cycle | [51, 57] |
| Bax | Capan-2, SW 1990; UM-UC 3, T24, EJ, A2780, SKOV3 | Induces apoptosis | [38, 56] |
| AKT | HCC-LM3, Huh7, SMMC-7721; LNCap, PC3 | AKT/GSK3β pathway, regulation of cell proliferation, cell cycle and apoptosis | [33, 54] |
| MYC | A549, H1299; A2780, SKOV3 | Involved in MYC/WEE1 pathway and Wnt/β-catenin pathway, promotes cell growth, migration, and invasion, regulation of DNA damage repair and cell cycle | [25, 51] |
| SOD2 | UM-UC 3, T24, EJ; A498 | Involved in antioxidant response and ROS metabolism | [56, 60] |
| Catalase | UM-UC 3, T24, EJ; A498 | Involved in antioxidant response and ROS metabolism | [56, 60] |
| PPARγ, SIRT1 | UM-UC 3, T24, EJ; A498 | Involved in PPARγ/SIRT1 signaling pathway, antioxidant reaction, apoptosis, and cell cycle | [56, 60] |
| CCNA1/2 | UM-UC 3, T24, EJ | Regulation of cell cycle | [56] |
| Cyclin E | HCC-LM3, Huh7, SMMC-7721 | Regulation of cell cycle | [33] |
| CCNE1, p27 | SKOV3, A2780 | Regulation of cell cycle | [51] |
| hMSH5, NBS1 and MRE11 | A549, LC319 | Regulation of cell cycle, and involved in the repair of DNA damage | [9] |
| ATM | A549, U2OS | Regulation of cell cycle, and involved in the repair of DNA damage | [9] |
| E-cadherin, SPHK1 | HCC-LM3, Huh7 | Regulation of EMT, and promotes cell migration and invasion | [34] |
| Slug | A2780, HEY, SKOV3 | Regulation of EMT, and promotes cell migration and invasion | [51] |
| β-catenin | A549, H1299 | Involved in the Wnt/β-catenin signaling pathway, promotes cell growth, migration, and invasion, and regulation of the cell cycle | [25] |
| MDM2 | Capan-2, SW 1990 | Involved in the MDM2/p53 signaling pathway, and regulation of DNA damage repair and the cell cycle | [38] |
| Bcl-2 | A2780, SKOV3 | Inhibits apoptosis | [51] |
| Bcl-xl | UM-UC 3, T24, EJ | Inhibits apoptosis | [56] |
| Caspase3/7/9 | UM-UC 3, T24, EJ | Induces apoptosis | [56] |
| PARP | H929, KMS28BM | Induces apoptosis | [72] |
| WEE1 | A2780, SKOV3 | Involved in the MYC/WEE1 signaling pathway, regulation of DNA damage repair and the cell cycle | [51] |
| γH2A.X | A2780, SKOV3 | Regulation of DNA damage repair | [51] |
| Rb and Cdc2 | NIH/3T3 mouse fibroblasts | Regulation of the cell cycle | [18] |
| Histone H4 | p53-null HRas-V12 transformed MEFs | Forms a dimer with CENP-A that binds to HJURP | [18] |
| Hmox-1 | UM-UC 3, T24, EJ | Regulation of ROS metabolism | [56] |
| FOXO3a | A498 | Downstream of the PPARγ/SIRT1 signaling pathway | [60] |
| JNK | LNCap, PC3 | Involved in the GSK3β/JNK signaling pathway, regulation of cell proliferation, the cell cycle, and apoptosis | [54] |
| JNK1, JNK2 | T24, TCCSUP | Involved in the JNK/STAT3 signaling pathway, regulation of cell proliferation, the cell cycle, and apoptosis | [57] |
| SKP2 | HCC-LM3, SMMC-7721 | Regulation of p21 stability by ubiquitination | [33] |
| YAP1, NDRG1 | MDA-MB-231, BT549 | Involved in the YAP1/NDRG1 signaling pathway, and promotes cell cancer proliferation | [48] |
| STAT3 | T24, TCCSUP | Involved in JNK/STAT3 signaling pathway, regulation of cell proliferation, the cell cycle, and apoptosis | [57] |
| MKI67, PCNA | T24, TCCSUP | Promotes cell proliferation | [57] |
| KLF11 | U87, U251 | Regulation of HJURP | [68] |
| NSD2 | H929, KMS28BM | Regulation of HJURP transcription | [72] |
| BRD4 | H929, KMS28BM | Activation of HJURP super-enhancer | [72] |
The biological functions of HJURP vary depending on the type of cancer and subcellular location. HJURP overexpression is generally associated with poor clinical outcomes and promotes tumorigenesis in most cancers, while contrary roles of HJURP have been observed according to database analysis in some cancers and it has been confirmed to act as a cancer suppressor in RCC, albeit with differences among RCC subtypes [60, 62]. Likewise, downregulation of HJURP expression caused by HJURP polymorphism may lead to susceptibility to HCC [29]. These findings suggest that HJURP may have a dual role in cancer and more evidence is needed to support this hypothesis.
Localization of HJURP expression has been a contentious topic among various studies, with different cancer cell types displaying varying molecular functions. For example, in U2OS cells, serous ovarian cancer cells, and t(4;14)-positive multiple myeloma cells, HJURP has been observed to localize in the nucleus [8, 9, 12, 72]. HJURP has been further found to co-localize with ER and microtubules in U2OS cells [15]; however, in various cancer cells, including HCC, and prostate and breast cancers, HJURP is mainly expressed in the cytoplasm [34, 48, 53, 54]. HJURP has been located in the cytoplasm and/or cell membrane in CRC [41], while in gliomas HJURP immunoreactivity has been observed in both the nucleus and cytoplasm, increasing with the grade of gliomas [64]. HJURP nuclear expression was more positive in epithelial and lymphoid cells, while cytoplasmic HJURP was positive in B3- and C-type TETs and associated with nuclear and cytoplasmic CENP-A [73]. The observed variations in the localization of HJURP imply its involvement in diverse functions because HJURP is functionally associated with CENP-A, which leads to poor prognosis in human cancers when expressed in the nucleus, but a better prognosis in a small subgroup of patients with TETs when expressed in the cytoplasm [73, 78]. Further investigation is needed to advance our understanding of HJURP complex mechanisms in cancer.
In conclusion, our study highlights the critical role of HJURP in cancer progression and suggests that HJURP may serve as a potential biomarker for predicting patient outcomes and as a novel therapeutic target. Therefore, regulating HJURP levels or identifying HJURP inhibitors may be promising strategies for cancer treatment.





